Clarithromycin
These highlights do not include all the information needed to use CLARITHROMYCIN TABLETS safely and effectively. See full prescribing information for CLARITHROMYCIN TABLETS. CLARITHROMYCIN tablets, for oral use Initial U.S. Approval: 1991
25debd85-0840-4f8d-9898-027772061dd2
HUMAN PRESCRIPTION DRUG LABEL
Feb 4, 2023
A-S Medication Solutions
DUNS: 830016429
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Clarithromycin
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (17)
Drug Labeling Information
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Clarithromycin

CLINICAL STUDIES SECTION
14 CLINICAL STUDIES
14.1 Mycobacterial Infections
Prophylaxis of Mycobacterial Infections
A randomized, double-blind clinical trial (trial 3) compared clarithromycin 500 mg twice a day to placebo in patients with CDC-defined AIDS and CD4 counts less than 100 cells/μL. This trial accrued 682 patients from November 1992 to January 1994, with a median CD4 cell count at entry of 30 cells/mcL. Median duration of clarithromycin was 10.6 months vs. 8.2 months for placebo. More patients in the placebo arm than the clarithromycin arm discontinued prematurely from the trial (75.6% and 67.4%, respectively). However, if premature discontinuations due to Mycobacterium avium complex (MAC) or death are excluded, approximately equal percentages of patients on each arm (54.8%) on clarithromycin and 52.5% on placebo) discontinued study drug early for other reasons. The trial was designed to evaluate the following endpoints:
1. MAC bacteremia, defined as at least one positive culture for Mycobacterium avium complex bacteria from blood or another normally sterile site
2. Survival
3. Clinically significant disseminated MAC disease, defined as MAC bacteremia accompanied by signs or symptoms of serious MAC infection, including fever, night sweats, weight loss, anemia, or elevations in liver function tests
MAC Bacteremia
In patients randomized to clarithromycin, the risk of MAC bacteremia was reduced by 69% compared to placebo. The difference between groups was statistically significant (p < 0.001). On an intent-to-treat basis, the one- year cumulative incidence of MAC bacteremia was 5.0% for patients randomized to clarithromycin and 19.4% for patients randomized to placebo. While only 19 of the 341 patients randomized to clarithromycin developed MAC, 11 of these cases were resistant to clarithromycin. The patients with resistant MAC bacteremia had a median baseline CD4 count of 10 cells/mm3 (range 2 cells/mm3 to 25 cells/mm3). Information regarding the clinical course and response to treatment of the patients with resistant MAC bacteremia is limited. The 8 patients who received clarithromycin and developed susceptible MAC bacteremia had a median baseline CD4 count of 25 cells/mm3 (range 10 cells/mm3 to 80 cells/mm3). Comparatively, 53 of the 341 placebo patients developed MAC; none of these isolates were resistant to clarithromycin. The median baseline CD4 count was 15 cells/mm3 (range 2 cells/mm3 to 130 cells/mm3) for placebo patients that developed MAC.
Survival
A statistically significant survival benefit of clarithromycin compared to placebo was observed (see Figure 3 and Table 13). Since the analysis at 18 months includes patients no longer receiving prophylaxis the survival benefit of clarithromycin may be underestimated.
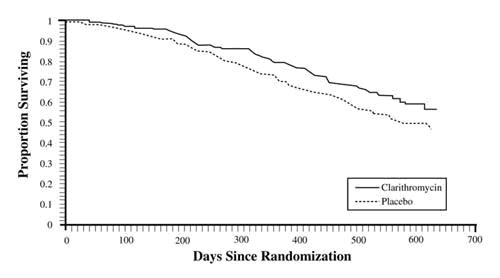
Figure 3. Survival of All Randomized AIDS Patients Over Time in Trial 3
Table 13. Mortality Rates at 18 months in Trial 3
|
Mortality Rates |
Reduction in Mortality Rates on Clarithromycin | ||
|
Placebo |
Clarithromycin | ||
|
6 month |
9.4% |
6.5% |
31% |
|
12 month |
29.7% |
20.5% |
31% |
|
18 month |
46.4% |
37.5% |
20% |
Clinically Significant Disseminated MAC Disease
In association with the decreased incidence of MAC bacteremia, patients in the group randomized to clarithromycin showed reductions in the signs and symptoms of disseminated MAC disease, including fever, night sweats, weight loss, and anemia.
Treatment of Mycobacterial Infections
Dose-Ranging Monotherapy Trials in Adult AIDS Patients with MAC
Two randomized clinical trials (Trials 1 and 2) compared different dosages of clarithromycin in patients with CDC-defined AIDS and CD4 counts less than100 cells/mcL. These trials accrued patients from May 1991 to March 1992. Trial 500 was a randomized, double-blind trial; trial 577 was an open-label compassionate use trial. Both trials used 500 mg and 1000 mg twice daily dosing of clarithromycin; trial 1 also had a 2000 mg twice daily clarithromycin group. Trial 1 enrolled 154 adult patients and trial 2 enrolled 469 adult patients. The majority of patients had CD4 cell counts less than 50 cells/mcL at study entry. The trials were designed to evaluate the following end points:
1. Change in MAC bacteremia or blood cultures negative for M. avium.
2. Change in clinical signs and symptoms of MAC infection including one or more of the following: fever, night sweats, weight loss, diarrhea, splenomegaly, and hepatomegaly.
The results for trial 1 are described below. The trial 2 results were similar to the results of trial 1.
MAC Bacteremia
Decreases in MAC bacteremia or negative blood cultures were seen in the majority of patients in all clarithromycin dosage groups. The mean reductions in MAC colony forming units (CFU) from baseline after 4 weeks of therapy in the 1000 mg (n=32) twice daily and 2000 mg (n=26) twice daily regimen was 2.3 Log CFU compared to 1.5 Log CFU in the clarithromycin 500 mg twice daily (n=35) regimen. A separate trial with a four-drug regimen2 (ciprofloxacin, ethambutol, rifampicin, and clofazimine) had a mean reduction of 1.4 Log CFU.
Clinical outcomes evaluated with the different dosing regimens of clarithromycin monotherapy are shown in Table 14. The 1000 mg and 2000 mg twice daily doses showed significantly better control of bacteremia during the first four weeks of therapy. No significant differences were seen beyond that point. All of the isolates had MIC less than 8 mcg/mL at pre-treatment. Relapse was almost always accompanied by an increase in MIC.
Table 14. Outcome with the Different Dosing Regimens of Clarithromycin
|
Outcome |
Clarithromycin 500 mg twice daily |
Clarithromycin 1000 mg twice daily |
Clarithromycin 2000 mg twice daily |
|
One or more negative blood cultures at any time during acute therapy |
61% (30/49) |
59% (29/49) |
52% (25/48) |
|
Two or more negative blood cultures during acute therapy sustained through study day 84 |
25% (12/49) |
25% (12/49) |
8% (4/48) |
|
Death or discontinuation by day 84 |
23% (11/49) |
37% (18/49) |
56% (27/48) |
|
Relapse by day 84 |
14% (7/49) |
12% (6/49) |
13% (6/48) |
|
Median time to first negative culture (in days) |
54 |
41 |
29 |
|
Median time to first decrease of at least 1 log CFU (in days) |
29 |
16 |
15 |
|
Median time to first positive culture or study discontinuation following the first negative culture (in days) |
43 |
59 |
43 |
Clinically Significant Disseminated MAC Disease
Among patients experiencing night sweats prior to therapy, 84% showed resolution or improvement at some point during the 12 weeks of clarithromycin at 500 mg to 2000 mg twice daily doses. Similarly, 77% of patients reported resolution or improvement in fevers at some point. Response rates for clinical signs of MAC are given in Table 15 below.
The median duration of response, defined as improvement or resolution of clinical signs and symptoms, was 2 weeks to 6 weeks.
Since the trial was not designed to determine the benefit of monotherapy beyond 12 weeks, the duration of response may be underestimated for the 25% to 33% of patients who continued to show clinical response after 12 weeks.
Table 15. Response Rates for Clinical Signs of MAC During 6 Weeks to 12 Weeks of Treatment
|
Resolution of Fever |
Resolution of Night Sweats | ||||
|
Clarithromycin twice daily dose (mg) |
% ever afebrile |
% afebrile |
Clarithromycin twice daily dose (mg) |
% ever resolving |
% resolving |
|
500 |
67% |
23% |
500 |
85% |
42% |
|
1000 |
67% |
12% |
1000 |
70% |
33% |
|
2000 |
62% |
22% |
2000 |
72% |
36% |
|
Weight Gain Greater Than 3% |
Hemoglobin Increase Greater Than 1 gm | ||||
|
Clarithromycin twice daily dose (mg) |
% ever gaining |
% gaining |
Clarithromycin twice daily dose (mg) |
% ever increasing |
% increasing |
|
500 |
33% |
14% |
500 |
58% |
26% |
|
1000 |
26% |
17% |
1000 |
37% |
6% |
|
2000 |
26% |
12% |
2000 |
62% |
18% |
Survival
Median survival time from trial entry (trial 1) was 249 days at the 500 mg twice daily dose compared to 215 days with the 1000 mg twice daily dose. However, during the first 12 weeks of therapy, there were 2 deaths in 53 patients in the 500 mg twice daily group versus 13 deaths in 51 patients in the 1000 mg twice daily group. The reason for this apparent mortality difference is not known. Survival in the two groups was similar beyond 12 weeks. The median survival times for these dosages were similar to recent historical controls with MAC when treated with combination therapies.2
Median survival time from entry in trial 2 was 199 days for the 500 mg twice a day dose and 179 days for the 1000 mg twice a day dose. During the first four weeks of therapy, while patients were maintained on their originally assigned dose, there were 11 deaths in 255 patients taking 500 mg twice daily and 18 deaths in 214 patients taking 1000 mg twice daily.
Dosage-Ranging Monotherapy Trials in Pediatric AIDS Patients with MAC
Trial 4 was a pediatric trial of 3.75 mg/kg, 7.5 mg/kg, and 15 mg/kg of clarithromycin twice daily in patients with CDC-defined AIDS and CD4 counts less than 100 cells/mcL. The trial enrolled 25 patients between the ages of 1 to 20. The trial evaluated the same endpoints as in the adult trials 1 and 2. Results with the 7.5 mg/kg twice daily dose in the pediatric trial were comparable to those for the 500 mg twice daily regimen in the adult trials.
Combination Therapy in AIDS Patients with Disseminated MAC
Trial 5 compared the safety and efficacy of clarithromycin in combination with ethambutol versus clarithromycin in combination with ethambutol and clofazimine for the treatment of disseminated MAC (dMAC) infection. This 24-week trial enrolled 106 patients with AIDS and dMAC, with 55 patients randomized to receive clarithromycin and ethambutol, and 51 patients randomized to receive clarithromycin, ethambutol, and clofazime. Baseline characteristics between treatment arms were similar with the exception of median CFU counts being at least 1 log higher in the clarithromycin, ethambutol, and clofazime arm.
Compared to prior experience with clarithromycin monotherapy, the two-drug regimen of clarithromycin and ethambutol extended the time to microbiologic relapse, largely through suppressing the emergence of clarithromycin resistant strains. However, the addition of clofazimine to the regimen added no additional microbiologic or clinical benefit. Tolerability of both multidrug regimens was comparable with the most common adverse events being gastrointestinal in nature. Patients receiving the clofazimine-containing regimen had reduced survival rates; however, their baseline mycobacterial colony counts were higher. The results of this trial support the addition of ethambutol to clarithromycin for the treatment of initial dMAC infections but do not support adding clofazimine as a third agent.
14.2 Otitis Media
Otitis Media Trial of Clarithromycin vs. Oral Cephalosporin
In a controlled clinical trial of pediatric patients with acute otitis media performed in the United States, where significant rates of beta-lactamase producing organisms were found, clarithromycin was compared to an oral cephalosporin. In this trial, strict evaluability criteria were used to determine clinical response. For the 223 patients who were evaluated for clinical efficacy, the clinical success rate (i.e., cure plus improvement) at the post-therapy visit was 88% for clarithromycin and 91% for the cephalosporin.
In a smaller number of patients, microbiologic determinations were made at the pre-treatment visit. The presumptive bacterial eradication/clinical cure outcomes (i.e., clinical success) are shown in Table 16.
Table 16. Clinical Success Rates of Otitis Media Treatment by Pathogen
|
Pathogen |
Clinical Success Rates | |
|
Clarithromycin |
Oral Cephalosporin | |
|
S. pneumoniae |
13/15 (87%) |
4/5 |
|
H. influenzaea |
10/14 (71%) |
3/4 |
|
M. catarrhalis |
4/5 |
1/1 |
|
S. pyogenes |
3/3 |
0/1 |
|
All Pathogens Combined |
30/37 (81%) |
8/11 (73%) |
|
a None of the H. influenzae isolated pre-treatment was resistant to clarithromycin; 6% were resistant to the control agent. |
Otitis Media Trials of Clarithromycin vs. Antimicrobial/Beta-lactamase Inhibitor
In two other controlled clinical trials of acute otitis media performed in the United States, where significant rates of beta-lactamase producing organisms were found, clarithromycin was compared to an oral antimicrobial agent that contained a specific beta-lactamase inhibitor. In these trials, strict evaluability criteria were used to determine the clinical responses. In the 233 patients who were evaluated for clinical efficacy, the combined clinical success rate (i.e., cure and improvement) at the post-therapy visit was 91% for both clarithromycin and the control.
For the patients who had microbiologic determinations at the pre-treatment visit, the presumptive bacterial eradication/clinical cure outcomes (i.e., clinical success) are shown in Table 17.
Table 17. Clinical Success Rates of Acute Otitis Media Treatment by Pathogen
|
Clinical Success Rates | ||
|
PATHOGEN |
Clarithromycin |
Antimicrobial/Beta-lactamase Inhibitor |
|
S. pneumoniae |
43/51 (84%) |
55/56 (98%) |
|
H. influenzaea |
36/45 (80%) |
31/33 (94%) |
|
M. catarrhalis |
9/10 (90%) |
6/6 |
|
S. pyogenes |
3/3 |
5/5 |
|
All Pathogens Combined |
91/109 (83%) |
97/100 (97%) |
|
a Of the H. influenzae isolated pre-treatment, 3% were resistant to clarithromycin and 10% were resistant to the control agent. |
14.3 H. pylori Eradication to Decrease the Risk of Duodenal Ulcer
Recurrence
Clarithromycin + Lansoprazole and Amoxicillin
Two U.S. randomized, double-blind clinical trials (trial 6 and trial 7) in patients with H. pylori and duodenal ulcer disease (defined as an active ulcer or history of an active ulcer within one year) evaluated the efficacy of clarithromycin 500 mg twice daily in combination with lansoprazole 30 mg twice daily and amoxicillin 1 gm twice daily as 14-day triple therapy for eradication of H. pylori.
H. pylori eradication was defined as two negative tests (culture and histology) at 4 weeks to 6 weeks following the end of treatment.
The combination of clarithromycin plus lansoprazole and amoxicillin as triple therapy was effective in eradication of H. pylori (see results in Table 18). Eradication of H. pylori has been shown to reduce the risk of duodenal ulcer recurrence.
A randomized, double-blind clinical trial (trial 8) performed in the U.S. in patients with H. pylori and duodenal ulcer disease (defined as an active ulcer or history of an ulcer within one year) compared the efficacy of clarithromycin in combination with lansoprazole and amoxicillin as triple therapy for 10 days and 14 days. This trial established that the 10-day triple therapy was equivalent to the 14-day triple therapy in eradicating H. pylori (see results in Table 18).
|
Table 18.H. pylori Eradication Rates-Triple Therapy (clarithromycin/lansoprazole/amoxicillin) Percent of Patients Cured [95% Confidence Interval] (number of patients) | |||
|
Trial |
Duration |
Triple Therapy |
Triple Therapy |
|
Trial 6 |
14 days |
92c [80 to 97.7] |
86c [73.3 to 93.5] |
|
Trial 7 |
14 days |
86d [75.7 to 93.6] |
83d [72 to 90.8] |
|
Trial 8e |
14 days |
85 [77 to 91] |
82 [73.9 to 88.1] |
|
10 days |
84 [76 to 89.8] |
81 [73.9 to 87.6] | |
|
a Based on evaluable patients with confirmed duodenal ulcer (active or within
one year) and H. pylori infection at baseline defined as at least two of three
positive endoscopic tests from CLOtest (Delta West LTD., Bentley, Australia),
histology, and/or culture. Patients were included in the analysis if they
completed the trial. Additionally, if patients were dropped out of the trial
due to an adverse reaction related to the drug, they were included in the
analysis as evaluable failures of therapy. |
Clarithromycin + Omeprazole and Amoxicillin Therapy
Three U.S., randomized, double-blind clinical trials in patients with H. pylori infection and duodenal ulcer disease (n = 558) compared clarithromycin plus omeprazole and amoxicillin to clarithromycin plus amoxicillin. Two trials (trials 9 and 10) were conducted in patients with an active duodenal ulcer, and the third trial (trial 11) was conducted in patients with a duodenal ulcer in the past 5 years, but without an ulcer present at the time of enrollment. The dosage regimen in the trials was clarithromycin 500 mg twice a day plus omeprazole 20 mg twice a day plus amoxicillin 1 gram twice a day for 10 days. In trials 9 and 10, patients who took the omeprazole regimen also received an additional 18 days of omeprazole 20 mg once a day. Endpoints studied were eradication of H. pylori and duodenal ulcer healing (trials 9 and 10 only). H. pylori status was determined by CLOtest®, histology, and culture in all three trials. For a given patient, H. pylori was considered eradicated if at least two of these tests were negative, and none was positive. The combination of clarithromycin plus omeprazole and amoxicillin was effective in eradicating H. pylori (see results in Table 19).
|
Table 19.H. pylori Eradication Rates: % of Patients Cured [95% Confidence Interval] | ||||
|
Clarithromycin + omeprazole + amoxicillin |
Clarithromycin + amoxicillin | |||
|
Per-Protocol****a |
Intent-to-Treat****b |
Per-Protocol****a |
Intent-to-Treat****b | |
|
Trial 9 |
c77 [64, 86] |
69 [57, 79] |
43 [31, 56] |
37 [27, 48] |
|
Trial 10 |
c78 [67, 88] |
73 [61, 82] |
41 [29, 54] |
36 [26, 47] |
|
Trial 11 |
c90 [80, 96] |
83 [74, 91] |
33 [24, 44] |
32 [23, 42] |
|
a Patients were included in the analysis if they had confirmed duodenal ulcer
disease (active ulcer trials 9 and 10; history of ulcer within 5 years, trial
11) and H. pylori infection at baseline defined as at least two of three
positive endoscopic tests from CLOtest®, histology, and/or culture. Patients
were included in the analysis if they completed the trial. Additionally, if
patients dropped out of the trial due to an adverse reaction related to the
study drug, they were included in the analysis as failures of therapy. The
impact of eradication on ulcer recurrence has not been assessed in patients
with a past history of ulcer. |
Clarithromycin + Omeprazole Therapy
Four randomized, double-blind, multi-center trials (trials 12, 13, 14, and 15) evaluated clarithromycin 500 mg three times a day plus omeprazole 40 mg once a day for 14 days, followed by omeprazole 20 mg once a day (trials 12, 13, and 15) or by omeprazole 40 mg once a day (trial 14) for an additional 14 days in patients with active duodenal ulcer associated with H. pylori. Trials 12 and 13 were conducted in the U.S. and Canada and enrolled 242 and 256 patients, respectively. H. pylori infection and duodenal ulcer were confirmed in 219 patients in trial 12 and 228 patients in trial 13. These trials compared the combination regimen to omeprazole and clarithromycin monotherapies. Trials 14 and 15 were conducted in Europe and enrolled 154 and 215 patients, respectively. H. pylori infection and duodenal ulcer were confirmed in 148 patients in trial 14 and 208 patients in trial 15. These trials compared the combination regimen to omeprazole monotherapy. The results for the efficacy analyses for these trials are described in Tables 20, 21, and 22.
Duodenal Ulcer Healing
The combination of clarithromycin and omeprazole was as effective as omeprazole alone for healing duodenal ulcer (see Table 20).
|
Table 20. End-of-Treatment Ulcer Healing Rates Percent of Patients Healed (n/N) | |||
|
Trial |
Clarithromycin + Omeprazole |
Omeprazole |
Clarithromycin |
|
U.S. Trials | |||
|
Trial 13 |
94% (58/62)a |
88% (60/68) |
71% (49/69) |
|
Trial 12 |
88% (56/64)a |
85% (55/65) |
64% (44/69) |
|
Non-U.S. Trials | |||
|
Trial 15 |
99% (84/85) |
95% (82/86) |
N/A |
|
Trial 14b |
100% (64/64) |
99% (71/72) |
N/A |
|
a p < 0.05 for clarithromycin + omeprazole versus clarithromycin monotherapy. |
Eradication of H. pylori Associated with Duodenal Ulcer
The combination of clarithromycin and omeprazole was effective in eradicating H. pylori (see Table 21). H. pylori eradication was defined as no positive test (culture or histology) at 4 weeks following the end of treatment, and two negative tests were required to be considered eradicated. In the per-protocol analysis, the following patients were excluded: dropouts, patients with major protocol violations, patients with missing H. pylori tests post-treatment, and patients that were not assessed for H. pylori eradication at 4 weeks after the end of treatment because they were found to have an unhealed ulcer at the end of treatment.
|
Table 21.H. pylori Eradication Rates (Per-Protocol Analysis) at 4 to 6 weeks Percent of Patients Cured (n/N) | |||
|
Trial |
Clarithromycin + Omeprazole |
Omeprazole |
Clarithromycin |
|
U.S. Trials | |||
|
Trial 13 |
64% (39/61)a,b |
0% (0/59) |
39% (17/44) |
|
Trial 12 |
74% (39/53)a,b |
0% (0/54) |
31% (13/42) |
|
Non-U.S. Trials | |||
|
Trial 15 |
74% (64/86)b |
1% (1/90) |
N/A |
|
Trial 14 |
83% (50/60)b |
1% (1/74) |
N/A |
|
a Statistically significantly higher than clarithromycin monotherapy (p <
0.05). |
Duodenal Ulcer Recurrence
Ulcer recurrence at 6-months and at 12 months following the end of treatment was assessed for patients in whom ulcers were healed post-treatment (see the results in Table 22). Thus, in patients with duodenal ulcer associated with H. pylori infection, eradication of H. pylori reduced ulcer recurrence.
|
Table 22. Duodenal Ulcer Recurrence at 6 months and 12 months in Patients with Healed Ulcers | ||
|
H. pylori****Negative at 4 to 6 Weeks |
H. pylori****Positive at 4 to 6 Weeks | |
|
U.S. Trials Recurrence at 6 Months | ||
|
** Trial 100** | ||
|
Clarithromycin + Omeprazole |
6% (2/34) |
56% (9/16) |
|
Omeprazole |
(0/0) |
71% (35/49) |
|
Clarithromycin |
12% (2/17) |
32% (7/22) |
|
** Trial 067** | ||
|
Clarithromycin + Omeprazole |
38% (11/29) |
50% (6/12) |
|
Omeprazole |
(0/0) |
67% (31/46) |
|
Clarithromycin |
18% (2/11) |
52% (14/27) |
|
** Non-U.S. Trials Recurrence at 6 Months** | ||
|
** Trial 058** | ||
|
Clarithromycin + Omeprazole |
6% (3/53) |
24% (4/17) |
|
Omeprazole |
0% (0/3) |
55% (39/71) |
|
** Trial 812b** | ||
|
Clarithromycin + Omeprazole |
5% (2/42) |
0% (0/7) |
|
Omeprazole |
0% (0/1) |
54% (32/59) |
|
Non-U.S. Trials Recurrence at 12 Months in Trial 14 | ||
|
Clarithromycin + Omeprazole |
3% (1/40) |
0% (0/6) |
|
Omeprazole |
0% (0/1) |
67% (29/43) |
DESCRIPTION SECTION
11 DESCRIPTION
Clarithromycin is a semi-synthetic macrolide antimicrobial for oral use. Chemically, it is 6-0-methylerythromycin. The molecular formula is C38H69NO13, and the molecular weight is 747.96. The structural formula is:

Figure 1: Structure of Clarithromycin
Clarithromycin USP is a white or almost white, crystalline powder. It is soluble in acetone, slightly soluble in methanol, ethanol, and acetonitrile, and practically insoluble in water.
Clarithromycin tablets, USP are available as immediate-release tablets.
Each film-coated tablet contains 250 mg or 500 mg of clarithromycin USP and the following inactive ingredients: microcrystalline cellulose, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, povidone, hypromellose, titanium dioxide, hydroxypropyl cellulose, iron oxide yellow, propylene glycol, vanillin, and sorbic acid.
HOW SUPPLIED SECTION
16 HOW SUPPLIED/STORAGE AND HANDLING
Product: 50090-1830
NDC: 50090-1830-0 20 TABLET, FILM COATED in a BOTTLE
NDC: 50090-1830-1 30 TABLET, FILM COATED in a BOTTLE
NDC: 50090-1830-2 14 TABLET, FILM COATED in a BOTTLE
NDC: 50090-1830-3 28 TABLET, FILM COATED in a BOTTLE
