Lurasidone Hydrochloride
These highlights do not include all the information needed to use LURASIDONE HYDROCHLORIDE TABLETS safely and effectively. See Full Prescribing Information for LURASIDONE HYDROCHLORIDE TABLETS. LURASIDONE HYDROCHLORIDE tablets, for oral use Initial U.S. Approval: 2010
27f70e15-ee22-47ae-ba87-20e2b4a1a4d6
HUMAN PRESCRIPTION DRUG LABEL
Jan 25, 2023
Alembic Pharmaceuticals Limited
DUNS: 650574663
Products 5
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Lurasidone Hydrochloride
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (8)
Lurasidone Hydrochloride
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (8)
Lurasidone Hydrochloride
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (8)
Lurasidone Hydrochloride
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (10)
Lurasidone Hydrochloride
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (8)
Drug Labeling Information
WARNINGS AND PRECAUTIONS SECTION
5 WARNINGS AND PRECAUTIONS
5.1 Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6- to 1.7-times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug- treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Lurasidone hydrochloride is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning, Warnings and Precautions (5.3)].
5.2 Suicidal Thoughts and Behaviors in Pediatric and Young Adult Patients
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients, and over 4,400 pediatric patients, the incidence of suicidal thoughts and behaviors in pediatric and young adult patients was greater in antidepressant-treated patients than in placebo-treated patients. The drug- placebo differences in the number of cases of suicidal thoughts and behaviors per 1000 patients treated are provided in Table 2.
No suicides occurred in any of the pediatric studies. There were suicides in the adult studies, but the number was not sufficient to reach any conclusion about antidepressant drug effect on suicide.
Table 2: Risk Differences of the Number of Cases of Suicidal Thoughts or Behaviors in the Pooled Placebo-Controlled Trials of Antidepressants in Pediatric and Adult Patients
|
Age Range |
Drug-Placebo Difference in Number of Patients of Suicidal Thoughts or Behaviors per 1000 Patients Treated |
|---|---|
|
Increases Compared to Placebo | |
|
<18 |
14 additional patients |
|
18 to 24 |
5 additional patients |
|
Decreases Compared to Placebo | |
|
25 to 64 |
1 fewer patient |
|
≥65 |
6 fewer patients |
It is unknown whether the risk of suicidal thoughts and behaviors in pediatric and young adult patients extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance studies in adults with MDD that antidepressants delay the recurrence of depression.
Monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing lurasidone hydrochloride, in patients whose depression is persistently worse, or who are experiencing emergent suicidal thoughts or behaviors.
5.3 Cerebrovascular Adverse Reactions, Including Stroke in Elderly Patients
with Dementia-Related Psychosis
In placebo-controlled trials with risperidone, aripiprazole, and olanzapine in elderly subjects with dementia, there was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks), including fatalities, compared to placebo-treated subjects. Lurasidone hydrochloride is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning, Warnings and Precautions (5.1)].
5.4 Neuroleptic Malignant Syndrome
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with administration of antipsychotic drugs, including lurasidone hydrochloride. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability. Additional signs may include elevated creatine phosphokinase, myoglobinuria (rhabdomyolysis), and acute renal failure.
If NMS is suspected, immediately discontinue lurasidone hydrochloride and provide intensive symptomatic treatment and monitoring.
5.5 Tardive Dyskinesia
Tardive dyskinesia is a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements that can develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to rely upon prevalence estimates to predict, at the inception of antipsychotic treatment, which patients are likely to develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
The risk of developing tardive dyskinesia and the likelihood that it will become irreversible are believed to increase as the duration of treatment and the total cumulative dose of antipsychotic drugs administered to the patient increase. However, the syndrome can develop, although much less commonly, after relatively brief treatment periods at low doses or may even arise after discontinuation of treatment.
The syndrome may remit, partially or completely, if antipsychotic treatment is withdrawn. Antipsychotic treatment, itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome and thereby may possibly mask the underlying process. The effect that symptomatic suppression has upon the long-term course of the syndrome is unknown.
Given these considerations, lurasidone hydrochloride should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that (1) is known to respond to antipsychotic drugs, and (2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinesia appear in a patient on lurasidone hydrochloride, drug discontinuation should be considered. However, some patients may require treatment with lurasidone hydrochloride despite the presence of the syndrome.
5.6 Metabolic Changes
Atypical antipsychotic drugs have been associated with metabolic changes that may increase cardiovascular/cerebrovascular risk. These metabolic changes include hyperglycemia, dyslipidemia, and body weight gain. While all of the drugs in the class have been shown to produce some metabolic changes, each drug has its own specific risk profile.
Hyperglycemia and Diabetes Mellitus
Hyperglycemia, in some cases extreme and associated with ketoacidosis or hyperosmolar coma or death, has been reported in patients treated with atypical antipsychotics. Assessment of the relationship between atypical antipsychotic use and glucose abnormalities is complicated by the possibility of an increased background risk of diabetes mellitus in patients with schizophrenia and the increasing incidence of diabetes mellitus in the general population. Given these confounders, the relationship between atypical antipsychotic use and hyperglycemia-related adverse events is not completely understood. However, epidemiological studies suggest an increased risk of hyperglycemia-related adverse events in patients treated with the atypical antipsychotics.
Patients with an established diagnosis of diabetes mellitus who are started on
atypical antipsychotics should be monitored regularly for worsening of glucose
control. Patients with risk factors for diabetes mellitus (e.g., obesity,
family history of diabetes) who are starting treatment with atypical
antipsychotics should undergo fasting blood glucose testing at the beginning
of treatment and periodically during treatment. Any patient treated with
atypical antipsychotics should be monitored for symptoms of hyperglycemia
including polydipsia, polyuria, polyphagia, and weakness. Patients who develop
symptoms of hyperglycemia during treatment with atypical antipsychotics should
undergo fasting blood glucose testing. In some cases, hyperglycemia has
resolved when the atypical antipsychotic was discontinued; however, some
patients required continuation of anti-diabetic treatment despite
discontinuation of the suspect drug.
Bipolar Depression
Adults
Monotherapy
Data from the adult short-term, flexible-dose, placebo-controlled monotherapy bipolar depression study is presented in Table 4.
Table 4: Change in Fasting Glucose in the Adult Monotherapy Bipolar Depression Study
|
****Lurasidone Hydrochloride | |||
|---|---|---|---|
|
Placebo |
20 to 60 mg/day |
80 to 120 mg/day | |
|
Mean Change from Baseline (mg/dL) | |||
|
n=148 |
n=140 |
n=143 | |
|
Serum Glucose |
+1.8 |
-0.8 |
+1.8 |
|
Proportion of Patients with Shifts to ≥ 126 mg/dL | |||
|
Serum Glucose |
4.3% |
2.2% |
6.4% |
Patients were randomized to flexibly dosed lurasidone hydrochloride 20 to 60 mg/day, lurasidone hydrochloride 80 to 120 mg/day, or placebo
In the uncontrolled, open-label, longer-term bipolar depression study, patients who received lurasidone hydrochloride as monotherapy in the short- term study and continued in the longer-term study, had a mean change in glucose of +1.2 mg/dL at week 24 (n=129).
Adjunctive Therapy with Lithium or Valproate
Data from the adult short-term, flexible-dosed, placebo-controlled adjunctive therapy bipolar depression studies are presented in Table 5.
Table 5: Change in Fasting Glucose in the Adult Adjunctive Therapy Bipolar Depression Studies
|
Placebo |
Lurasidone Hydrochloride | |
|---|---|---|
|
Mean Change from Baseline (mg/dL) | ||
|
n=302 |
n=319 | |
|
Serum Glucose |
-0.9 |
+1.2 |
|
Proportion of Patients with Shifts to ≥ 126 mg/dL | ||
|
Serum Glucose |
1% |
1.3% |
Patients were randomized to flexibly dosed lurasidone hydrochloride 20 to 120 mg/day or placebo as adjunctive therapy with lithium or valproate.
In the uncontrolled, open-label, longer-term bipolar depression study, patients who received lurasidone hydrochloride as adjunctive therapy with either lithium or valproate in the short-term study and continued in the longer-term study, had a mean change in glucose of +1.7 mg/dL at week 24 (n=88).
Pediatric Patients (10 to 17 years)
In studies of pediatric patients 10 to 17 years and adults with bipolar depression, changes in fasting glucose were similar. In the 6-week, placebo- controlled study of pediatric patients with bipolar depression, mean change in fasting glucose was +1.6 mg/dL for lurasidone hydrochloride 20 to 80 mg/day (n=145) and -0.5 mg/dL for placebo (n=145).
Pediatric Patients (6 to 17 years)
In a 104-week, open-label study in pediatric patients with bipolar depression, autistic disorder, or another indication, 7 % of patients with a normal baseline fasting glucose experienced a shift to high at endpoint while taking lurasidone.
Dyslipidemia
Undesirable alterations in lipids have been observed in patients treated with
atypical antipsychotics.
Bipolar Depression
Adults
Monotherapy
Data from the adult short-term, flexible-dosed, placebo-controlled, monotherapy bipolar depression study are presented in Table 7.
Table 7: Change in Fasting Lipids in the Adult Monotherapy Bipolar Depression Study
|
Lurasidone Hydrochloride | |||
|---|---|---|---|
|
Placebo |
20 to 60 mg/day |
80 to 120 mg/day | |
|
Mean Change from Baseline (mg/dL) | |||
|
n=147 |
n=140 |
n=144 | |
|
Total cholesterol |
-3.2 |
+1.2 |
-4.6 |
|
Triglycerides |
+6 |
+5.6 |
+0.4 |
|
Proportion of Patients with Shifts | |||
|
Total cholesterol |
4.2% |
4.4% |
4.4% |
|
Triglycerides |
4.8% |
10.1% |
9.8% |
Patients were randomized to flexibly dosed lurasidone hydrochloride 20 to 60 mg/day, lurasidone hydrochloride 80 to 120 mg/day, or placebo
In the uncontrolled, open-label, longer-term bipolar depression study, patients who received lurasidone hydrochloride as monotherapy in the short- term and continued in the longer-term study had a mean change in total cholesterol and triglycerides of -0.5 mg/dL (n=130) and -1 mg/dL (n=130) at week 24, respectively.
Adjunctive Therapy with Lithium or Valproate
Data from the adult short-term, flexible-dosed, placebo-controlled, adjunctive therapy bipolar depression studies are presented in Table 8.
Table 8: Change in Fasting Lipids in the Adult Adjunctive Therapy Bipolar Depression Studies
|
Placebo |
LurasidoneHydrochloride | |
|---|---|---|
|
Mean Change from Baseline (mg/dL) | ||
|
n=303 |
n=321 | |
|
Total cholesterol |
-2.9 |
-3.1 |
|
Triglycerides |
-4.6 |
+4.6 |
|
Proportion of Patients with Shifts | ||
|
Total cholesterol |
5.7% |
5.4% |
|
Triglycerides |
8.6% |
10.8% |
Patients were randomized to flexibly dosed lurasidone hydrochloride 20 to 120 mg/day or placebo as adjunctive therapy with lithium or valproate.
In the uncontrolled, open-label, longer-term bipolar depression study, patients who received lurasidone hydrochloride, as adjunctive therapy with either lithium or valproate in the short-term study and continued in the longer-term study, had a mean change in total cholesterol and triglycerides of -0.9 (n=88) and +5.3 (n=88) mg/dL at week 24, respectively.
Pediatric Patients (10 to 17 years)
In the 6-week, placebo-controlled bipolar depression study with pediatric patients 10 to 17 years, mean change in fasting cholesterol was -6.3 mg/dL for lurasidone hydrochloride 20 to 80 mg/day (n=144) and 1.4 mg/dL for placebo (n=145), and mean change in fasting triglyceride was -7.6 mg/dL for lurasidone hydrochloride 20 to 80 mg/day (n=144) and +5.9 mg/dL for placebo (n=145).
Pediatric Patients (6 to 17 years)
In a 104-week, open-label study of pediatric patients with bipolar depression, autistic disorder, or another indication, shifts in baseline fasting cholesterol from normal to high at endpoint were reported in 12% (total cholesterol), 3% (LDL cholesterol), and shifts in baseline from normal to low were reported in 27% (HDL cholesterol) of patients taking lurasidone. Of patients with normal baseline fasting triglycerides, 12% experienced shifts to high.
Weight Gain
Weight gain has been observed with atypical antipsychotic use. Clinical monitoring of weight is recommended.
Bipolar Depression
Adults
Monotherapy
Data from the adult short-term, flexible-dosed, placebo-controlled monotherapy bipolar depression study are presented in Table 11. The mean change in weight gain was +0.29 kg for lurasidone hydrochloride-treated patients compared to -0.04 kg for placebo-treated patients. The proportion of patients with a ≥7% increase in body weight (at Endpoint) was 2.4% for lurasidone hydrochloride- treated patients and 0.7% for placebo-treated patients.
Table 11: Mean Change in Weight (kg) from Baseline in the Adult Monotherapy Bipolar Depression Study
|
Lurasidone Hydrochloride | |||
|
Placebo |
20 to 60 mg/day |
80 to 120 mg/day | |
|
All Patients |
-0.04 |
+0.56 |
+0.02 |
Patients were randomized to flexibly dosed lurasidone hydrochloride 20 to 60 mg/day, lurasidone hydrochloride 80 to 120 mg/day, or placebo
In the uncontrolled, open-label, longer-term bipolar depression study, patients who received lurasidone hydrochloride as monotherapy in the short- term and continued in the longer-term study had a mean change in weight of -0.02 kg at week 24 (n=130).
Adjunctive Therapy with Lithium or Valproate
Data from the adult short-term, flexible-dosed, placebo-controlled adjunctive therapy bipolar depression studies are presented in Table 12. The mean change in weight gain was +0.11 kg for lurasidone hydrochloride-treated patients compared to +0.16 kg for placebo-treated patients. The proportion of patients with a ≥7% increase in body weight (at Endpoint) was 3.1% for lurasidone hydrochloride-treated patients and 0.3% for placebo-treated patients.
Table 12: Mean Change in Weight (kg) from Baseline in the Adult Adjunctive Therapy Bipolar Depression Studies
|
Lurasidone Hydrochloride | ||
|
Placebo |
20 to 120 mg/day(n=327) | |
|
All Patients |
+0.16 |
+0.11 |
Patients were randomized to flexibly dosed lurasidone hydrochloride 20 to 120 mg/day or placebo as adjunctive therapy with lithium or valproate.
In the uncontrolled, open-label, longer-term bipolar depression study, patients who were treated with lurasidone hydrochloride, as adjunctive therapy with either lithium or valproate in the short-term and continued in the longer-term study, had a mean change in weight of +1.28 kg at week 24 (n=86).
Pediatric Patients (10 to 17 years)
Data from the 6-week, placebo-controlled bipolar depression study in patients 10 to 17 years are presented in Table 13. The mean change in weight gain was +0.7 kg for lurasidone hydrochloride-treated patients compared to +0.5 kg for placebo-treated patients. The proportion of patients with a ≥7% increase in body weight (at Endpoint) was 4% for lurasidone hydrochloride-treated patients and 5.3% for placebo-treated patients.
Table 13: Mean Change in Weight (kg) from Baseline in the Bipolar Depression Study in Pediatric Patients (10 to 17 years)
|
Lurasidone Hydrochloride | ||
|
Placebo |
20 to 80 mg/day | |
|
All Patients |
+0.5 |
+0.7 |
Pediatric Patients (6 to 17 years)
In a long-term, open-label study that enrolled pediatric patients with bipolar depression, autistic disorder or another indication, from three short-term, placebo-controlled trials, 54% (378/701) received lurasidone for 104 weeks. The mean increase in weight from open-label baseline to Week 104 was 5.85 kg. To adjust for normal growth, z-scores were derived (measured in standard deviations [SD]), which normalize for the natural growth of children and adolescents by comparisons to age-and sex-matched population standards. A z-score change <0.5 SD is considered not clinically significant. In this trial, the mean change in z-score from open-label baseline to Week 104 was -0.06 SD for body weight and -0.13 SD for body mass index (BMI), indicating minimal deviation from the normal curve for weight gain.
5.7 Hyperprolactinemia
As with other drugs that antagonize dopamine D2 receptors, lurasidone hydrochloride elevates prolactin levels.
Hyperprolactinemia may suppress hypothalamic GnRH, resulting in reduced pituitary gonadotrophin secretion. This, in turn, may inhibit reproductive function by impairing gonadal steroidogenesis in both female and male patients. Galactorrhea, amenorrhea, gynecomastia, and impotence have been reported with prolactin-elevating compounds. Long-standing hyperprolactinemia, when associated with hypogonadism, may lead to decreased bone density in both female and male patients [see Adverse Reactions (6)].
Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin-dependent in vitro, a factor of potential importance if the prescription of these drugs is considered in a patient with previously detected breast cancer. As is common with compounds which increase prolactin release, an increase in mammary gland neoplasia was observed in a carcinogenicity study conducted with lurasidone in rats and mice [see Nonclinical Toxicology (13)]. Neither clinical studies nor epidemiologic studies conducted to date have shown an association between chronic administration of this class of drugs and tumorigenesis in humans, but the available evidence is too limited to be conclusive.
Bipolar Depression
Adults
Monotherapy
The median change from baseline to endpoint in prolactin levels, in the adult
short-term, flexible-dosed, placebo-controlled monotherapy bipolar depression
study, was +1.7 ng/mL and +3.5 ng/mL with lurasidone hydrochloride 20 to 60
mg/day and 80 to 120 mg/day, respectively compared to +0.3 ng/mL with placebo-
treated patients. The median change from baseline to endpoint for males was
+1.5 ng/mL and for females was +3.1 ng/mL. Median changes for prolactin by
dose range are shown in Table 16.
Table 16: Median Change in Prolactin (ng/mL) from Baseline in the Adult Monotherapy Bipolar Depression Study
|
Lurasidone Hydrochloride | |||
|
Placebo |
20 to 60 mg/day |
80 to 120 mg/day | |
|
All Patients |
+0.3 |
+1.7 |
+3.5 |
|
(n=147) |
(n=140) |
(n=144) | |
|
Females |
0 |
+1.8 |
+5.3 |
|
(n=82) |
(n=78) |
(n=88) | |
|
Males |
+0.4 |
+1.2 |
+1.9 |
|
(n=65) |
(n=62) |
(n=56) |
Patients were randomized to flexibly dosed lurasidone hydrochloride 20 to 60 mg/day, lurasidone hydrochloride 80 to 120 mg/day, or placebo
The proportion of patients with prolactin elevations ≥5x upper limit of normal (ULN) was 0.4% for lurasidone hydrochloride-treated patients and 0% for placebo-treated patients. The proportion of female patients with prolactin elevations ≥5x ULN was 0.6% for lurasidone hydrochloride-treated patients and 0% for placebo-treated female patients. The proportion of male patients with prolactin elevations ≥5x ULN was 0% and 0% for placebo-treated male patients.
In the uncontrolled, open-label, longer-term bipolar depression study, patients who were treated with lurasidone hydrochloride tablet as monotherapy in the short-term and continued in the longer-term study, had a median change in prolactin of -1.15 ng/mL at week 24 (n=130).
Adjunctive Therapy with Lithium or Valproate
The median change from baseline to endpoint in prolactin levels, in the adult
short-term, flexible-dosed, placebo-controlled adjunctive therapy bipolar
depression studies was +2.8 ng/mL with lurasidone hydrochloride 20 to 120
mg/day compared to 0 ng/mL with placebo-treated patients. The median change
from baseline to endpoint for males was +2.4ng/mL and for females was +3.2
ng/mL. Median changes for prolactin across the dose range are shown in Table
17.
Table 17: Median Change in Prolactin (ng/mL) from Baseline in the Adult Adjunctive Therapy Bipolar Depression Studies
|
Placebo |
Lurasidone Hydrochloride 20 to 120 mg/day | |
|
All Patients |
0 |
+2.8 |
|
(n=301) |
(n=321) | |
|
Females |
+0.4 |
+3.2 |
|
(n=156) |
(n=162) | |
|
Males |
-0.1 |
+2.4 |
|
(n=145) |
(n=159) |
Patients were randomized to flexibly dosed lurasidone hydrochloride 20 to 120 mg/day or placebo as adjunctive therapy with lithium or valproate.
The proportion of patients with prolactin elevations ≥5x upper limit of normal (ULN) was 0% for lurasidone hydrochloride-treated patients and 0% for placebo- treated patients. The proportion of female patients with prolactin elevations ≥5x ULN was 0% for lurasidone hydrochloride-treated patients and 0% for placebo-treated female patients. The proportion of male patients with prolactin elevations ≥5x ULN was 0% and 0% for placebo-treated male patients.
In the uncontrolled, open-label, longer-term bipolar depression study, patients who were treated with lurasidone hydrochloride, as adjunctive therapy with either lithium or valproate, in the short-term and continued in the longer-term study, had a median change in prolactin of -2.9 ng/mL at week 24 (n=88).
Pediatric Patients (10 to 17 years)
In the 6-week, placebo-controlled bipolar depression study with pediatric
patients 10 to 17 years, the median change from baseline to endpoint in
prolactin levels for lurasidone hydrochloride-treated patients was +1.1 ng/mL
and was +0.5 ng/mL for placebo-treated patients. For lurasidone hydrochloride-
treated patients, the median change from baseline to endpoint for males was
+0.85 ng/mL and for females was +2.5 ng/mL. Median changes for prolactin are
shown in Table 18.
Table 18: Median Change in Prolactin (ng/mL) from Baseline in the Bipolar Depression Study in Pediatric Patients (10 to 17 years)
|
|
Placebo |
Lurasidone Hydrochloride 20 to 80 mg/day |
|
All Patients |
+0.5 |
+1.1 |
|
(n=157) |
(n=165) | |
|
Females |
+0.55 |
+2.5 |
|
(n=78) |
(n=83) | |
|
Males |
+0.5 |
+0.85 |
|
(n=79) |
(n=82) |
The proportion of patients with prolactin elevations ≥5x ULN was 0% for lurasidone hydrochloride-treated patients and 0.6% for placebo-treated patients. The proportion of female patients with prolactin elevations ≥5x ULN was 0% for lurasidone hydrochloride-treated patients and 1.3% for placebo- treated female patients. No male patients in the placebo or lurasidone hydrochloride treatment groups had prolactin elevations ≥5x ULN.
Pediatric Patients (6 to 17 years)
In a 104-week, open-label study of pediatric patients with bipolar depression, autistic disorder, or another indication, the median changes from baseline to endpoint in serum prolactin levels were -0.2 ng/mL (all patients), -0.3 ng/mL (females), and -0.05 ng/mL (males). The proportions of patients with a markedly high prolactin level (≥5 times the upper limit of normal) at any time during open-label treatment were 2% (all patients), 3% (females), and 1% (males).
Adverse events among females in this trial that are potentially prolactin- related include galactorrhea (0.6%). Among male patients in this study, decreased libido was reported in one patient (0.2%) and there were no reports of impotence, gynecomastia, or galactorrhea.
5.8 Leukopenia, Neutropenia and Agranulocytosis
Leukopenia/neutropenia has been reported during treatment with antipsychotic agents. Agranulocytosis (including fatal cases) has been reported with other agents in the class.
Possible risk factors for leukopenia/neutropenia include pre-existing low white blood cell count (WBC) and history of drug-induced leukopenia/neutropenia. Patients with a pre-existing low WBC or a history of drug-induced leukopenia/neutropenia should have their complete blood count (CBC) monitored frequently during the first few months of therapy and lurasidone hydrochloride should be discontinued at the first sign of decline in WBC, in the absence of other causative factors.
Patients with neutropenia should be carefully monitored for fever or other symptoms or signs of infection and treated promptly if such symptoms or signs occur. Patients with severe neutropenia (absolute neutrophil count <1000/mm3) should discontinue lurasidone hydrochloride and have their WBC followed until recovery.
5.9 Orthostatic Hypotension and Syncope
Lurasidone hydrochloride may cause orthostatic hypotension and syncope, perhaps due to its α1-adrenergic receptor antagonism. Associated adverse reactions can include dizziness, light headedness, tachycardia, and bradycardia. Generally, these risks are greatest at the beginning of treatment and during dose escalation. Patients at increased risk of these adverse reactions or at increased risk of developing complications from hypotension include those with dehydration, hypovolemia, treatment with antihypertensive medication, history of cardiovascular disease (e.g., heart failure, myocardial infarction, ischemia, or conduction abnormalities), history of cerebrovascular disease, as well as patients who are antipsychotic-naïve. In such patients, consider using a lower starting dose and slower titration, and monitor orthostatic vital signs.
Orthostatic hypotension, as assessed by vital sign measurement, was defined by the following vital sign changes: ≥20 mm Hg decrease in systolic blood pressure and ≥10 bpm increase in pulse from sitting to standing or supine to standing position.
Bipolar Depression
Adults
Monotherapy
In the adult short-term, flexible-dose, placebo-controlled monotherapy bipolar
depression study, there were no reported adverse events of orthostatic
hypotension and syncope.
Orthostatic hypotension, as assessed by vital signs, occurred with a frequency of 0.6% with lurasidone hydrochloride 20 to 60 mg and 0.6% with lurasidone hydrochloride 80 to 120 mg compared to 0% with placebo.
Adjunctive Therapy with Lithium or Valproate
In the adult short-term, flexible-dose, placebo-controlled adjunctive therapy bipolar depression therapy studies, there were no reported adverse events of orthostatic hypotension and syncope. Orthostatic hypotension, as assessed by vital signs, occurred with a frequency of 1.1% with lurasidone hydrochloride 20 to 120 mg compared to 0.9% with placebo.
Pediatric Patients (10 to 17 years)
In the 6-week, placebo-controlled bipolar depression study in pediatric patients 10 to 17 years, there were no reported adverse events of orthostatic hypotension or syncope.
Orthostatic hypotension, as assessed by vital signs, occurred with a frequency of 1.1% with lurasidone hydrochloride 20 to 80 mg/day, compared to 0.6% with placebo.
5.10 Falls
Lurasidone hydrochloride may cause somnolence, postural hypotension, motor and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
5.11 Seizures
As with other antipsychotic drugs, lurasidone hydrochloride should be used cautiously in patients with a history of seizures or with conditions that lower the seizure threshold, e.g., Alzheimer’s dementia. Conditions that lower the seizure threshold may be more prevalent in patients 65 years or older.
Bipolar Depression
Monotherapy
In the adult and pediatric 6-week, flexible-dose, placebo-controlled monotherapy bipolar depression studies, no patients experienced seizures/convulsions.
Adjunctive Therapy with Lithium or Valproate
In the adult short-term, flexible-dose, placebo-controlled adjunctive therapy bipolar depression studies, no patient experienced seizures/convulsions.
5.12 Potential for Cognitive and Motor Impairment
Lurasidone hydrochloride, like other antipsychotics, has the potential to impair judgment, thinking or motor skills. Caution patients about operating hazardous machinery, including motor vehicles, until they are reasonably certain that therapy with lurasidone hydrochloride does not affect them adversely.
In clinical studies with lurasidone hydrochloride, somnolence included:
hypersomnia, hypersomnolence, sedation and somnolence.
Bipolar Depression
Adults
Monotherapy
In the adult short-term, flexible-dosed, placebo-controlled monotherapy
bipolar depression study, somnolence was reported by 7.3% (12/164) and 13.8%
(23/167) with lurasidone hydrochloride 20 to 60 mg and 80 to 120 mg,
respectively compared to 6.5% (11/168) of placebo patients.
Adjunctive Therapy with Lithium or Valproate
In the adult short-term, flexible-dosed, placebo-controlled adjunctive therapy bipolar depression studies, somnolence was reported by 11.4% (41/360) of patients treated with lurasidone hydrochloride 20 to 120 mg compared to 5.1% (17/334) of placebo patients.
Pediatric Patients (10 to 17 years)
In the 6-week, placebo-controlled bipolar depression study in pediatric
patients 10 to 17 years, somnolence was reported by 11.4% (20/175) of patients
treated with lurasidone hydrochloride 20 to 80 mg/day compared to 5.8%
(10/172) of placebo treated patients.
5.13 Body Temperature Dysregulation
Disruption of the body’s ability to reduce core body temperature has been attributed to antipsychotic agents. Appropriate care is advised when prescribing lurasidone hydrochloride for patients who will be experiencing conditions that may contribute to an elevation in core body temperature, e.g., exercising strenuously, exposure to extreme heat, receiving concomitant medication with anticholinergic activity, or being subject to dehydration.
5.14 Activation of Mania/Hypomania
Antidepressant treatment can increase the risk of developing a manic or hypomanic episode, particularly in patients with bipolar disorder. Monitor patients for the emergence of such episodes.
In the adult bipolar depression monotherapy and adjunctive therapy (with lithium or valproate) studies, less than 1% of subjects in the lurasidone hydrochloride and placebo groups developed manic or hypomanic episodes.
5.15 Dysphagia
Esophageal dysmotility and aspiration have been associated with antipsychotic drug use. Aspiration pneumonia is a common cause of morbidity and mortality in elderly patients, in particular those with advanced Alzheimer’s dementia. Lurasidone hydrochloride and other antipsychotic drugs should be used cautiously in patients at risk for aspiration pneumonia.
5.16 Neurological Adverse Reactions in Patients with Parkinson’s Disease or
Dementia with Lewy Bodies
Patients with Parkinson’s disease or Dementia with Lewy Bodies are reported to have an increased sensitivity to antipsychotic medication. Manifestations of this increased sensitivity include confusion, obtundation, postural instability with frequent falls, extrapyramidal symptoms, and clinical features consistent with the neuroleptic malignant syndrome.
- Cerebrovascular Adverse Reactions in Elderly Patients with Dementia- Related Psychosis: Increased incidence of cerebrovascular adverse events (e.g., stroke, transient ischemic attack) (5.3).
- Neuroleptic Malignant Syndrome: Manage with immediate discontinuation and close monitoring (5.4).
- Tardive Dyskinesia: Discontinue if clinically appropriate (5.5).
- Metabolic Changes: Monitor for hyperglycemia/diabetes mellitus, dyslipidemia and weight gain (5.6).
- Hyperprolactinemia: Prolactin elevations may occur (5.7).
- Leukopenia, Neutropenia, and Agranulocytosis: Perform complete blood counts (CBC) in patients with a pre-existing low white blood cell count (WBC) or a history of leukopenia or neutropenia. Consider discontinuing lurasidone hydrochloride tablets if a clinically significant decline in WBC occurs in the absence of other causative factors (5.8).
- Orthostatic Hypotension and Syncope: Monitor heart rate and blood pressure and warn patients with known cardiovascular or cerebrovascular disease, and risk of dehydration or syncope (5.9).
DRUG ABUSE AND DEPENDENCE SECTION
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
Lurasidone hydrochloride is not a controlled substance.
9.2 Abuse
Lurasidone hydrochloride has not been systematically studied in humans for its potential for abuse or physical dependence or its ability to induce tolerance. While clinical studies with lurasidone hydrochloride did not reveal any tendency for drug-seeking behavior, these observations were not systematic and it is not possible to predict the extent to which a CNS-active drug will be misused, diverted and/or abused once it is marketed. Patients should be evaluated carefully for a history of drug abuse, and such patients should be observed carefully for signs of lurasidone hydrochloride misuse or abuse (e.g., development of tolerance, drug-seeking behavior, increases in dose).
OVERDOSAGE SECTION
10 OVERDOSAGE
10.1 Human Experience
In premarketing clinical studies, accidental or intentional overdosage of lurasidone hydrochloride was identified in one patient who ingested an estimated 560 mg of lurasidone hydrochloride. This patient recovered without sequelae. This patient resumed lurasidone hydrochloride treatment for an additional two months.
10.2 Management of Overdosage
No specific antidotes for lurasidone hydrochloride are known. In managing overdose, provide supportive care, including close medical supervision and monitoring, and consider the possibility of multiple drug involvement. If an overdose occurs, consult a Certified Poison Control Center (1-800-222-1222 or www.poison.org).
Cardiovascular monitoring should commence immediately, including continuous electrocardiographic monitoring for possible arrhythmias. If antiarrhythmic therapy is administered, disopyramide, procainamide, and quinidine carry a theoretical hazard of additive QT-prolonging effects when administered in patients with an acute overdose of lurasidone hydrochloride. Similarly, the alpha-blocking properties of bretylium might be additive to those of lurasidone hydrochloride, resulting in problematic hypotension.
Hypotension and circulatory collapse should be treated with appropriate measures. Epinephrine and dopamine should not be used, or other sympathomimetics with beta-agonist activity, since beta stimulation may worsen hypotension in the setting of lurasidone hydrochloride-induced alpha blockade. In case of severe extrapyramidal symptoms, anticholinergic medication should be administered.
Gastric lavage (after intubation if patient is unconscious) and administration of activated charcoal together with a laxative should be considered.
The possibility of obtundation, seizures, or dystonic reaction of the head and neck following overdose may create a risk of aspiration with induced emesis.
DESCRIPTION SECTION
11 DESCRIPTION
Lurasidone hydrochloride is an atypical antipsychotic belonging to the chemical class of benzisothiazol derivatives.
Its chemical name is (3aR,4S,7R,7aS)-2-{(1R,2R)-2-[4-(1,2-benzisothiazol-3-yl)piperazin-1 ylmethyl] cyclohexylmethyl}hexahydro-4,7-methano-2H-isoindole-1,3-dione hydrochloride. Its molecular formula is C28H36N4O2S·HCl and its molecular weight is 529.14.
The chemical structure is:
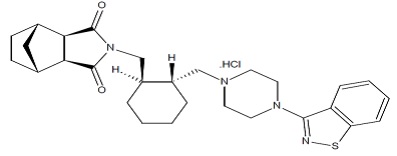
Lurasidone hydrochloride is a white to cream powder. It is very slightly soluble to insoluble in water, sparingly soluble in methanol. Practically insoluble in toluene.
Lurasidone hydrochloride tablets are intended for oral administration only. Each tablet contains 20 mg, 40 mg, 60 mg, 80 mg or 120 mg of lurasidone hydrochloride.
Inactive ingredients are mannitol, corn starch, croscarmellose sodium, hypromellose and magnesium stearate. The tablets are film-coated with coating material containing hypromellose, titanium dioxide and polyethylene glycol 8000. Additionally, the 80 mg tablet contains iron oxide yellow and FD&C Blue No. 2 Aluminum Lake.
CLINICAL STUDIES SECTION
14 CLINICAL STUDIES
14.2 Depressive Episodes Associated with Bipolar I Disorder
Adults
Monotherapy
The efficacy of lurasidone hydrochloride, as monotherapy, was established in a 6-week, multicenter, randomized, double-blind, placebo-controlled study of adult patients (mean age of 41.5 years, range 18 to 74) who met DSM-IV-TR criteria for major depressive episodes associated with bipolar I disorder, with or without rapid cycling, and without psychotic features (N=485). Patients were randomized to one of two flexible-dose ranges of lurasidone hydrochloride (20 to 60 mg/day, or 80 to 120 mg/day) or placebo.
The primary rating instrument used to assess depressive symptoms in this study was the Montgomery-Asberg Depression Rating Scale (MADRS), a 10-item clinician-rated scale with total scores ranging from 0 (no depressive features) to 60 (maximum score). The primary endpoint was the change from baseline in MADRS score at Week 6. The key secondary instrument was the Clinical Global Impression-Bipolar-Severity of Illness scale (CGI-BP-S), a clinician-rated scale that measures the subject’s current illness state on a 7-point scale, where a higher score is associated with greater illness severity.
For both dose groups, lurasidone hydrochloride was superior to placebo in reduction of MADRS and CGI-BP-S scores at Week 6. The primary efficacy results are provided in Table 37. The high dose range (80 to 120 mg per day) did not provide additional efficacy on average, compared to the low dose range (20 to 60 mg per day).
Adjunctive Therapy with Lithium or Valproate
The efficacy of lurasidone hydrochloride, as an adjunctive therapy with lithium or valproate, was established in a 6-week, multicenter, randomized, double-blind, placebo-controlled study of adult patients (mean age of 41.7 years, range 18 to 72) who met DSM-IV-TR criteria for major depressive episodes associated with bipolar I disorder, with or without rapid cycling, and without psychotic features (N=340). Patients who remained symptomatic after treatment with lithium or valproate were randomized to flexibly dosed lurasidone hydrochloride 20 to 120 mg/day or placebo.
The primary rating instrument used to assess depressive symptoms in this study was the MADRS. The primary endpoint was the change from baseline in MADRS score at Week 6. The key secondary instrument was the CGI-BP-S scale.
Lurasidone hydrochloride was superior to placebo in reduction of MADRS and CGI-BP-S scores at Week 6, as an adjunctive therapy with lithium or valproate (Table 37).
Table 37: Primary Efficacy Results for Adult Studies in Depressive Episodes Associated with Bipolar I Disorder (MADRS Scores)
|
Primary Efficacy Measure: MADRS | ||||
|
Study |
Treatment Group |
Mean Baseline Score (SD) |
LS Mean Change from Baseline (SE) |
Placebo**‐subtracted Differencea****(95% CI)** |
|
Monotherapy Study |
Lurasidone hydrochloride (20 to 60 mg/day)* |
30.3 (5) |
‐15.4 (0.8) |
‐4.6 (‐6.9, ‐2.3) |
|
Lurasidone hydrochloride (80 to 120 mg/day)* |
30.6 (4.9) |
‐15.4 (0.8) |
‐4.6 (‐6.9, ‐2.3) | |
|
Placebo |
30.5 (5) |
‐10.7 (0.8) |
‐‐ | |
|
Adjunctive Therapy study |
Lurasidone hydrochloride (20 to 120 mg/day)* + lithium or valproate |
30.6 (5.3) |
‐17.1 (0.9) |
‐3.6 (‐6, ‐1.1) |
|
Placebo + lithium or valproate |
30.8 (4.8) |
‐13.5 (0.9) |
‐‐ |
SD: standard deviation; SE: standard error; LS Mean: least‐squares mean; CI: confidence interval, unadjusted for multiple comparisons.
aDifference (drug minus placebo) in least‐squares mean change from baseline.
- Treatment group statistically significantly superior to placebo.
Pediatric Patients (10 to 17 years)
The efficacy of lurasidone hydrochloride was established in a 6-week, multicenter, randomized, double-blind, placebo-controlled study of pediatric patients (10 to 17 years) who met DSM-5 criteria for a major depressive episode associated with bipolar I disorder, with or without rapid cycling, and without psychotic features (N=343). Patients were randomized to flexibly dosed lurasidone hydrochloride 20 to 80 mg/day or placebo. At the end of the clinical study, most patients (67%) received 20 mg/day or 40 mg/day.
The primary rating scale used to assess depressive symptoms in this study was the Children’s Depression Rating Scale, Revised (CDRS-R) total score. The CDRS-R is a 17-item clinician- rated scale with total scores ranging from 17 to 113. The primary endpoint was the change from baseline in CDRS-R score at Week 6. The key secondary endpoint was the change from baseline in CGI-BP-S depression score.
Lurasidone hydrochloride was superior to placebo in reduction of CDRS-R total score and CGI-BP-S depression score at Week 6. The primary efficacy results are provided in Table 38.
Table 38: Primary Efficacy Results for the Study in Depressive Episodes Associated with Bipolar I Disorder (CDRS-R Total Score) in Pediatric Patients (10 to 17 years)
|
Treatment Group |
Primary Efficacy Measure: CDRS-R | ||
|
Mean Baseline Score (SD) |
LS Mean Change from Baseline (SE) |
Placebo-subtracted Difference****a******(95% CI)** | |
|
Lurasidone hydrochloride (20 to 80 mg/day)* |
59.2 (8.24) |
-21 (1.06) |
-5.7 (-8.4,-3) |
|
Placebo |
58.6 (8.26) |
-15.3 (1.08) |
-- |
SD: standard deviation; SE: standard error; LS Mean: least-squares mean; CI: confidence interval, unadjusted for multiple comparisons.
aDifference (drug minus placebo) in least-squares mean change from baseline.
- Treatment group statistically significantly superior to placebo.
HOW SUPPLIED SECTION
16 HOW SUPPLIED/STORAGE AND HANDLING
Lurasidone hydrochloride tablets 20 mg are white to off white, round shaped, biconvex, film-coated tablets debossed with ‘515’ on one side and ‘L’ on other side.
Lurasidone hydrochloride tablets 40 mg are white to off white, round shaped, biconvex, film-coated tablets debossed with ‘516’ on one side and ‘L’ on other side.
Lurasidone hydrochloride tablets 60 mg are white to off white, capsule shaped, biconvex, film-coated tablets debossed with ‘L 551’ on one side and plain on other side.
Lurasidone hydrochloride tablets 80 mg are pale green, oval shaped, biconvex, film-coated tablets debossed with ‘L 517’ on one side and plain on other side.
Lurasidone hydrochloride tablets 120 mg are white to off white, oval shaped, biconvex, film-coated tablets debossed with ‘L 518’ on one side and plain on other side.
Tablets are supplied in the following strengths and package configurations (Table 39).
Table 39: Package Configuration for Lurasidone Hydrochloride Tablets
|
Tablet Strength |
Package Configuration |
NDC Code |
|
20 mg |
Bottles of 30 |
46708-494-30 |
|
Bottles of 90 |
46708-494-90 | |
|
Bottles of 500 |
46708-494-71 | |
|
Bottles of 1000 |
46708-494-91 | |
|
Carton of 100 (10 x 10) unit dose tablets |
46708-494-10 | |
|
40 mg |
Bottles of 30 |
46708-495-30 |
|
Bottles of 90 |
46708-495-90 | |
|
Bottles of 500 |
46708-495-71 | |
|
Bottles of 1000 |
46708-495-91 | |
|
Carton of 100 (10 x 10) unit dose tablets |
46708-495-10 | |
|
60 mg |
Bottles of 30 |
46708-496-30 |
|
Bottles of 90 |
46708-496-90 | |
|
Bottles of 500 |
46708-496-71 | |
|
Bottles of 1000 |
46708-496-91 | |
|
Carton of 100 (10 x 10) unit dose tablets |
46708-496-10 | |
|
80 mg |
Bottles of 30 |
46708-497-30 |
|
Bottles of 90 |
46708-497-90 | |
|
Bottles of 500 |
46708-497-71 | |
|
Bottles of 1000 |
46708-497-91 | |
|
Carton of 100 (10 x 10) unit dose tablets |
46708-497-10 | |
|
120 mg |
Bottles of 30 |
46708-498-30 |
|
Bottles of 90 |
46708-498-90 | |
|
Bottles of 500 |
46708-498-71 | |
|
Bottles of 1000 |
46708-498-91 | |
|
Carton of 100 (10 x 10) unit dose tablets |
46708-498-10 |
Storage
Store lurasidone hydrochloride tablets at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
DOSAGE & ADMINISTRATION SECTION
2 DOSAGE AND ADMINISTRATION
2.2 Depressive Episodes Associated with Bipolar I Disorder
Adults
The recommended starting dose of lurasidone hydrochloride tablet is 20 mg given once daily as monotherapy or as adjunctive therapy with lithium or valproate. Initial dose titration is not required. Lurasidone hydrochloride tablet has been shown to be effective in a dose range of 20 mg per day to 120 mg per day as monotherapy or as adjunctive therapy with lithium or valproate [see Clinical Studies (14.2)]. The maximum recommended dose, as monotherapy or as adjunctive therapy with lithium or valproate, is 120 mg per day. In the monotherapy study, the higher dose range (80 mg to 120 mg per day) did not provide additional efficacy, on average, compared to the lower dose range (20 to 60 mg per day) [see Clinical Studies (14.2)].
Pediatric Patients (10 to 17 years)
The recommended starting dose of lurasidone hydrochloride tablet is 20 mg given once daily as monotherapy. Initial dose titration is not required. The dose may be increased after one week based on clinical response. Lurasidone hydrochloride tablet has been shown to be effective in a dose range of 20 mg per day to 80 mg per day as monotherapy. At the end of the clinical study, most of the patients (67%) received 20 mg or 40 mg once daily [see Clinical Studies (14.2)]. The maximum recommended dose is 80 mg per day.
The efficacy of lurasidone hydrochloride tablet in the treatment of mania associated with bipolar disorder has not been established.
2.3 Administration Information
Lurasidone hydrochloride tablet should be taken with food (at least 350 calories). Administration with food substantially increases the absorption of lurasidone hydrochloride. Administration with food increases the AUC approximately 2-fold and increases the Cmax approximately 3-fold. In the clinical studies, lurasidone hydrochloride tablet was administered with food [see Clinical Pharmacology (12.3)].
The effectiveness of lurasidone hydrochloride tablet for longer-term use, that is, for more than 6 weeks, has not been established in controlled studies. Therefore, the physician who elects to use lurasidone hydrochloride tablet for extended periods should periodically re-evaluate the long-term usefulness of the drug for the individual patient [see Dosage and Administration (2.1 and 2.2)].
2.4 Dose Modifications for Renal Impairment
Dose adjustment is recommended in moderate (creatinine clearance: 30 to <50 mL/min) and severe renal impairment (creatinine clearance <30 mL/min) patients. The recommended starting dose is 20 mg per day. The dose in these patients should not exceed 80 mg per day [see Use in Specific Populations (8.6)].
2.5 Dose Modifications for Hepatic Impairment
Dose adjustment is recommended in moderate (Child-Pugh Score = 7 to 9) and severe hepatic impairment (Child-Pugh Score = 10 to 15) patients. The recommended starting dose is 20 mg per day. The dose in moderate hepatic impairment patients should not exceed 80 mg per day and the dose in severe hepatic impairment patients should not exceed 40 per mg/day [see Use in Specific Populations (8.7)].
2.6 Dose Modifications Due to Drug Interactions of CYP3A4 Inhibitors and
CYP3A4 Inducers
Concomitant Use with CYP3A4 Inhibitors
Lurasidone hydrochloride tablets should not be used concomitantly with a strong CYP3A4 inhibitor (e.g., ketoconazole, clarithromycin, ritonavir, voriconazole, mibefradil, etc.) [see Contraindications (4)].
If lurasidone hydrochloride tablets are being prescribed and a moderate CYP3A4 inhibitor (e.g. diltiazem, atazanavir, erythromycin, fluconazole, verapamil etc.) is added to the therapy, the lurasidone hydrochloride tablet dose should be reduced to half of the original dose level. Similarly, if a moderate CYP3A4 inhibitor is being prescribed and lurasidone hydrochloride tablet is added to the therapy, the recommended starting dose of lurasidone hydrochloride tablet is 20 mg per day, and the maximum recommended dose of lurasidone hydrochloride tablet is 80 mg per day [see Contraindications (4), Drug Interactions (7.1)].
Grapefruit and grapefruit juice should be avoided in patients taking lurasidone hydrochloride tablets, since these may inhibit CYP3A4 and alter lurasidone hydrochloride concentrations [see Drug Interactions (7.1)].
Concomitant Use with CYP3A4 Inducers
Lurasidone hydrochloride tablets should not be used concomitantly with a strong CYP3A4 inducer (e.g., rifampin, avasimibe, St. John’s wort, phenytoin, carbamazepine, etc.) [see Contraindications (4); Drug Interactions (7.1)]. If lurasidone hydrochloride tablet is used concomitantly with a moderate CYP3A4 inducer, it may be necessary to increase the lurasidone hydrochloride tablet dose after chronic treatment (7 days or more) with the CYP3A4 inducer.
Lurasidone hydrochloride tablets should be taken with food (at least 350 calories). Administration with food substantially increases the absorption of lurasidone hydrochloride (2.3, 12.3). (2)
|
Indication |
Starting Dose |
Recommended Dose |
|
Bipolar Depression – adults (2.2) |
20 mg per day |
20 mg to 120 mg per day |
|
Bipolar Depression – pediatric patients (10 to 17 years) (2.2) |
20 mg per day |
20 mg to 80 mg per day |
- Moderate and Severe Renal Impairment: Recommended starting dose is 20 mg per day, and the maximum recommended dose is 80 mg per day (2.4, 8.6).
- Moderate and Severe Hepatic Impairment: Recommended starting dose is 20 mg per day. The maximum recommended dose is 80 mg per day in moderate hepatic impairment and 40 mg per day in severe hepatic impairment (2.5, 8.7).
- Concomitant Use of a Moderate CYP3A4 inhibitor (e**.**g., diltiazem):
- Lurasidone hydrochloride dose should be reduced to half of the original dose level. Recommended starting dose is 20 mg per day. Maximum recommended dose is 80 mg per day (2.6, 7.1).
- Concomitant Use of a Moderate CYP3A4 Inducer:
It may be necessary to increase the dose of lurasidone hydrochloride (2.6, 7.1). (2)
