ZAVZPRET
These highlights do not include all the information needed to use ZAVZPRET safely and effectively. See full prescribing information for ZAVZPRET. ZAVZPRET™ (zavegepant) nasal spray Initial U.S. Approval: 2023
9c4a7aec-daef-4961-ba77-92f4b58be780
HUMAN PRESCRIPTION DRUG LABEL
Aug 11, 2025
Pfizer Laboratories Div Pfizer Inc
DUNS: 134489525
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
zavegepant
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (6)
Drug Labeling Information
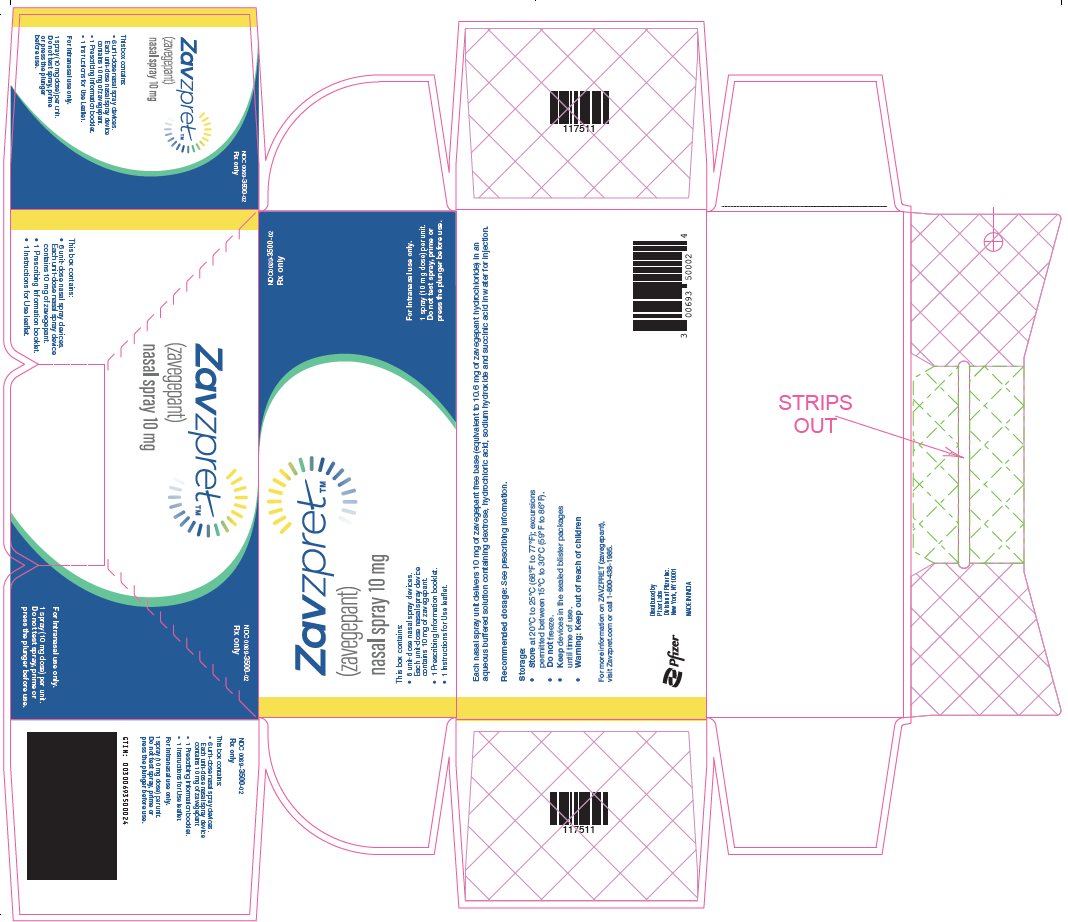
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL – Nasal Spray Carton
NDC 0069-3500-02
Rx only
ZavzpretTM
(zavegepant)
nasal spray 10 mg
This box contains:
• 6 unit-dose nasal spray devices.
Each unit-dose nasal spray device
contains 10 mg of zavegepant.
• 1 Prescribing Information booklet.
• 1 Instructions for Use leaflet.
For Intranasal use only.
1 spray (10 mg dose) per unit.
Do not test spray, prime or
press the plunger before use.
CONTRAINDICATIONS SECTION
4 CONTRAINDICATIONS
ZAVZPRET is contraindicated in patients with a history of hypersensitivity reaction to zavegepant or any of the components of ZAVZPRET. Reactions have included anaphylaxis [see Warnings and Precautions (5.1)].
Patients with a history of hypersensitivity reaction to zavegepant or to any of the components of ZAVZPRET. (4)
WARNINGS AND PRECAUTIONS SECTION
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Serious hypersensitivity reactions, including anaphylaxis, facial swelling, and urticaria, have occurred in patients treated with ZAVZPRET. If a hypersensitivity reaction occurs, discontinue ZAVZPRET and initiate appropriate therapy [see Contraindications (4) and Adverse Reactions (6.1, 6.2)].
5.2 Hypertension
Development of hypertension and worsening of pre-existing hypertension have been reported following the use of CGRP antagonists in the postmarketing setting.
Some of the patients receiving a CGRP antagonist who developed new-onset hypertension had risk factors for hypertension. There were cases requiring initiation of pharmacological treatment for hypertension and, in some cases, hospitalization. Hypertension may occur at any time during treatment, but was most frequently reported within 7 days of therapy initiation. The CGRP antagonist was discontinued in many of the reported cases.
Monitor patients treated with ZAVZPRET for new-onset hypertension or worsening of pre-existing hypertension, and consider whether discontinuation of ZAVZPRET is warranted if evaluation fails to establish an alternative etiology or blood pressure is inadequately controlled.
5.3 Raynaud’s Phenomenon
Development of Raynaud’s phenomenon and recurrence or worsening of pre- existing Raynaud’s phenomenon have been reported in the postmarketing setting following the use of CGRP antagonists.
In reported cases with small molecule CGRP antagonists, symptom onset occurred a median of 1.5 days following dosing. Many of the cases reported serious outcomes, including hospitalizations and disability, generally related to debilitating pain. In most reported cases, discontinuation of the CGRP antagonist resulted in resolution of symptoms.
ZAVZPRET should be discontinued if signs or symptoms of Raynaud’s phenomenon develop, and patients should be evaluated by a healthcare provider if symptoms do not resolve. Patients with a history of Raynaud’s phenomenon should be monitored for, and informed about the possibility of, worsening or recurrence of signs and symptoms.
•
Hypersensitivity Reactions: If a serious hypersensitivity reaction occurs, discontinue ZAVZPRET and initiate appropriate therapy. Hypersensitivity reactions including anaphylaxis, facial swelling, and urticaria, have occurred with ZAVZPRET. (5.1)
•
Hypertension: New-onset or worsening of pre-existing hypertension may occur. (5.2)
•
Raynaud’s Phenomenon: New-onset or worsening of pre-existing Raynaud’s phenomenon may occur. (5.3)
ADVERSE REACTIONS SECTION
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
•
Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
•
Hypertension [see Warnings and Precautions (5.2)]
•
Raynaud’s Phenomenon [see Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of ZAVZPRET for the acute treatment of migraine in adults has been evaluated in two randomized, double-blind, placebo-controlled trials (Study 1 and Study 2) in patients with migraine who received one 10 mg dose of ZAVZPRET nasal spray (N=1023) or placebo (N=1056) [see Clinical Studies (14)]. Approximately 83% were female, 81% were White, 20% were Hispanic or Latino, and 15% were Black. The mean age at study entry was 41 years (range 18-79 years of age).
Adverse reactions in Study 1 and 2 are shown in Table 1.
Table 1: Adverse Reactions Occurring in At Least 2% of Patients Treated with ZAVZPRET and at a Frequency Greater than Placebo in Study 1 and 2
| ||
|
Adverse Reaction |
ZAVZPRET N=1023 % |
Placebo N=1056 % |
|
Taste Disorders* |
18 |
4 |
|
Nausea |
4 |
1 |
|
Nasal Discomfort |
3 |
1 |
|
Vomiting |
2 |
<1 |
Hypersensitivity, including facial swelling and urticaria, occurred in less than 1% of patients treated with ZAVZPRET [see Contraindications (4) and Warnings and Precautions (5.1)].
Long-term safety was assessed in an open-label extension study. That study evaluated 603 patients, dosing intermittently for up to one year, including 360 patients who were exposed to ZAVZPRET 10 mg for at least 6 months, and 298 who were exposed for at least one year, all of whom treated an average of at least two migraine attacks per month.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ZAVZPRET. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Hypersensitivity (e.g., anaphylaxis) [see Contraindications (4) and Warnings and Precautions (5.1)]
Vascular Disorders: Hypertension [see Warnings and Precautions (5.2)], Raynaud’s phenomenon [see Warnings and Precautions (5.3)]
Most common adverse reactions (at least 2% of patients treated with ZAVZPRET and greater than placebo) were taste disorders, nausea, nasal discomfort, and vomiting. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Pfizer Inc. at 1-800-438-1985 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
RECENT MAJOR CHANGES SECTION
RECENT MAJOR CHANGES
|
Contraindications (4) |
8/2025 |
|
Warnings and Precautions (5.1) |
8/2025 |
|
Warnings and Precautions (5.2, 5.3) |
3/2025 |
CLINICAL STUDIES SECTION
14 CLINICAL STUDIES
The efficacy of ZAVZPRET for the acute treatment of migraine with or without aura in adults was demonstrated in two randomized, double-blind, placebo- controlled trials (Study 1 and Study 2). In both studies, patients were instructed to treat a migraine of moderate to severe headache pain intensity. Rescue medication (i.e., NSAIDs, acetaminophen, and/or an antiemetic) was allowed 2 hours after the initial treatment. Other forms of rescue medication such as triptans were not allowed within 48 hours of initial treatment. In Study 1 and Study 2, 13.4% and 13.6% of patients were taking preventive medications for migraine at baseline, respectively. None of the patients were on concomitant preventive medication that act on the CGRP pathway.
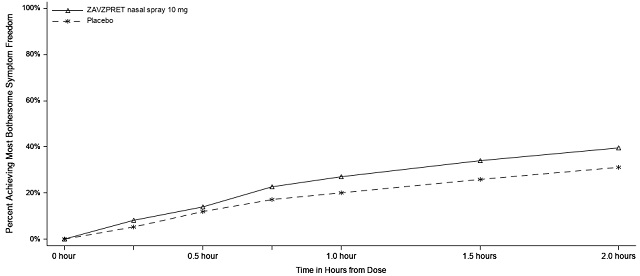
In Study 1 (NCT04571060), patients were randomized to receive a single dose of ZAVZPRET 10 mg (N=623) or placebo (N=646). Efficacy was demonstrated with ZAVZPRET 10 mg by an effect on the coprimary endpoints of pain freedom and most bothersome symptom (MBS) freedom at 2 hours after a single dose, compared to placebo. Pain freedom was defined as a reduction of moderate or severe headache pain to no headache pain, and MBS freedom was defined as the absence of the self-identified MBS (i.e., photophobia, phonophobia, or nausea). The most common MBS reported before dosing was photophobia (55%), followed by nausea (28%), and phonophobia (16%).
In Study 1, the percentage of patients achieving headache pain freedom and MBS freedom 2 hours after a single dose was statistically significantly greater in patients who received ZAVZPRET compared to those who received placebo (Table 2).
Table 2: Efficacy Endpoints in Study 1
| ||
|
ZAVZPRET 10 mg |
Placebo | |
|
Pain Free at 2 hours | ||
|
n/N* |
147/623 |
96/646 |
|
% Responders |
23.6 |
14.9 |
|
Difference from placebo (%) |
8.8 | |
|
p-value |
<0.001 | |
|
MBS†** Free at 2 hours** | ||
|
n/N* |
247/623 |
201/646 |
|
% Responders |
39.6 |
31.1 |
|
Difference from placebo (%) |
8.7 | |
|
p-value |
0.001 |
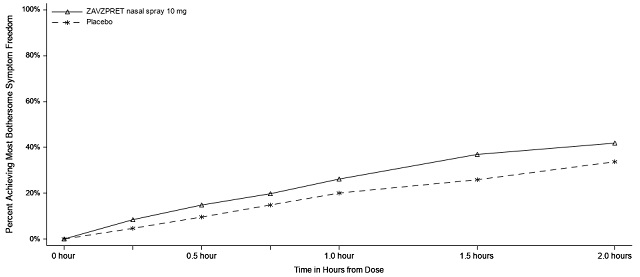
Figures 1 and 2 present the percentage of patients achieving migraine pain freedom and MBS freedom within 2 hours following treatment in Study 1.
Figure 1: Percentage of Patients Achieving Pain Freedom within 2 Hours in Study 1
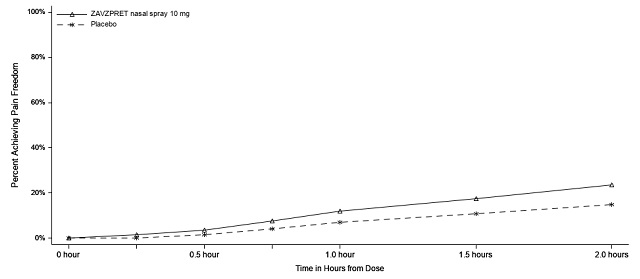
Figure 2: Percentage of Patients Achieving MBS Freedom within 2 Hours in Study 1
In Study 1, statistically significant effects of ZAVZPRET compared to placebo were demonstrated for the additional efficacy endpoints of pain relief at 2 hours post-dose, return to normal function at 2 hours post-dose, sustained pain freedom from 2 to 48 hours post-dose (Table 3), and phonophobia and photophobia freedom at 2 hours post-dose. Pain relief was defined as a reduction in migraine pain from moderate or severe severity to mild or none. The measurement of the percentage of patients reporting normal function at two hours after dosing was derived from a single item questionnaire, asking patients to select one response on a 4-point scale: normal function, mild impairment, severe impairment, or required bedrest.
Table 3: Additional Efficacy Endpoints in Study 1
| ||
|
ZAVZPRET 10 mg |
Placebo | |
|
Pain Relief at 2 hours | ||
|
n/N* |
366/623 |
321/646 |
|
% Responders |
58.7 |
49.7 |
|
Difference from placebo (%) |
9.0 | |
|
p-value |
0.001 | |
|
Percentage of Patients Reporting Normal Function at 2 hours† | ||
|
n/N* |
204/570 |
152/593 |
|
% Responders |
35.8 |
25.6 |
|
Difference from placebo (%) |
10.2 | |
|
p-value |
<0.001 | |
|
Sustained Pain Freedom from 2 to 48 hours | ||
|
n/N* |
77/623 |
56/646 |
|
% Responders |
12.4 |
8.7 |
|
Difference from placebo (%) |
3.7 | |
|
p-value |
0.031 |
The incidence of photophobia and phonophobia was reduced following administration of ZAVZPRET 10 mg as compared to placebo.
In Study 2 (NCT03872453), patients were randomized to receive a single dose of ZAVZPRET 10 mg (n=391) or placebo (n=401).
In Study 2, statistically significant efficacy was demonstrated with ZAVZPRET 10 mg by an effect on the coprimary endpoints of pain freedom and most bothersome symptom (MBS) freedom at 2 hours after a single dose, compared to placebo. Pain freedom was observed in 22.5% of patients receiving ZAVZPRET and 15.5% of patients receiving placebo (p-value = 0.011). MBS freedom was observed in 41.9% of patients receiving ZAVZPRET and 33.7% of patients receiving placebo (p-value = 0.016). The most common MBS reported before dosing was photophobia (53%), followed by nausea (31%), and phonophobia (15%).
Table 4: Efficacy Endpoints in Study 2
| ||
|
ZAVZPRET 10 mg |
Placebo | |
|
Pain Free at 2 hours | ||
|
n/N* |
88/391 |
62/401 |
|
% Responders |
22.5 |
15.5 |
|
Difference from placebo (%) |
7.0 | |
|
p-value |
0.011 | |
|
MBS†** Free at 2 hours** | ||
|
n/N* |
164/391 |
135/401 |
|
% Responders |
41.9 |
33.7 |
|
Difference from placebo (%) |
8.3 | |
|
p-value |
0.016 |
Figure 3: Percentage of Patients Achieving Pain Freedom within 2 Hours in Study 2
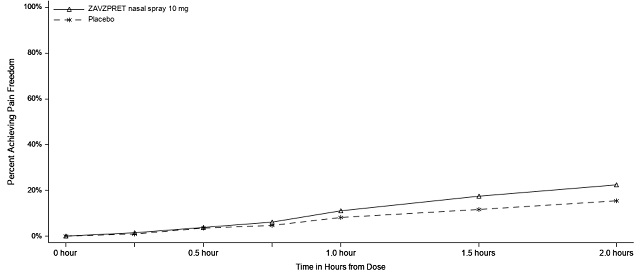
Figure 4: Percentage of Patients Achieving MBS Freedom within 2 Hours in Study 2
USE IN SPECIFIC POPULATIONS SECTION
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate data on the developmental risk associated with the use of ZAVZPRET in pregnant women. No adverse developmental effects were observed following subcutaneous administration of zavegepant to pregnant animals at doses associated with plasma exposures higher than those used clinically (see Data).
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively. The estimated rate of major birth defects (2.2 to 2.9%) and miscarriage (17%) among deliveries to women with migraine are similar to rates reported in women without migraine.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
Published data have suggested that women with migraine may be at increased risk of preeclampsia and gestational hypertension during pregnancy.
Data
Animal Data
Subcutaneous administration of zavegepant to pregnant rats (0, 10, 20, or 40 mg/kg/day) or rabbits (0, 20, 40, or 60 mg/kg/day) during the period of organogenesis resulted in no adverse effects on embryofetal development. Plasma exposures (AUC) at the highest doses tested were approximately 4000 times that in humans at the maximum recommended human dose (MRHD) of 10 mg/day.
Subcutaneous administration of zavegepant (0, 5, 10, or 20 mg/kg/day) to rats throughout pregnancy and lactation resulted in no adverse effects on pre- and postnatal development. Plasma exposure (AUC) at the highest dose tested was approximately 2500 times that in humans at the MRHD.
8.2 Lactation
There are no data on the presence of zavegepant or its metabolites in human milk, the effects of zavegepant on the breastfed infant, or the effects of zavegepant on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for ZAVZPRET and any potential adverse effects on the breastfed infant from ZAVZPRET or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ZAVZPRET did not include sufficient numbers of patients 65 years of age and older to determine whether they respond differently from younger adult patients.
In a limited number of patients 65 years of age and older, no clinically significant pharmacokinetic differences were observed between elderly and younger subjects.
8.6 Hepatic Impairment
No dosage adjustment of ZAVZPRET is necessary in patients with mild hepatic impairment (Child-Pugh Class A) or moderate hepatic impairment (Child-Pugh Class B). ZAVZPRET has not been studied in patients with severe hepatic impairment (Child-Pugh Class C). Avoid use of ZAVZPRET in patients with severe hepatic impairment [see Clinical Pharmacology (12.3)].
8.7 Renal Impairment
No dosage adjustment of ZAVZPRET is necessary in patients with estimated creatine clearance (CLcr) 30 mL/min or greater. Avoid use of ZAVZPRET in patients with CLcr less than 30 mL/min [see Clinical Pharmacology (12.3)].
•
Avoid use in patients with severe hepatic impairment. (8.6)
•
Avoid use in patients with CLcr < 30 mL/min. (8.7)
SPL PATIENT PACKAGE INSERT SECTION
Patient Package Insert
|
PATIENT INFORMATION (zavegepant) nasal spray |
What is ZAVZPRET?
ZAVZPRET is a prescription medicine used in adults for the acute treatment of migraine attacks with or without aura.
ZAVZPRET is not used to prevent migraine attacks.
It is not known if ZAVZPRET is safe and effective in children.
Do not use ZAVZPRET if you are:
•
allergic to zavegepant, or any of the ingredients in ZAVZPRET.
See the end of this leaflet for a complete list of ingredients in ZAVZPRET.
Before you use ZAVZPRET, tell your healthcare provider about all of your medical conditions, including if you:
•
have high blood pressure.
•
have circulation problems in your fingers and toes.
•
have kidney problems.
•
have liver problems.
•
are pregnant or plan to become pregnant. It is not known if ZAVZPRET will harm your unborn baby.
•
are breastfeeding or plan to breastfeed. It is not known whether ZAVZPRET passes into your breast milk. Talk with your healthcare provider about the best way to feed your baby if you use ZAVZPRET.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
How should I use ZAVZPRET?
•
Use ZAVZPRET exactly how your healthcare provider tells you to use it.
•
See the Instructions for Use for complete information on how to use ZAVZPRET nasal spray.
•
ZAVZPRET is given in the nose (nasal) only.
•
Each ZAVZPRET only sprays 1 time and cannot be reused.**Do not** test or prime the nasal spray before use.
•
Each dose of ZAVZPRET is provided in an individual pack. Use all of the medicine in 1 pack for a complete dose.
•
The recommended dose is 10 mg given as a single spray in one nostril.
•
Do not use more than 1 spray (10 mg) of ZAVZPRET nasal spray in a 24-hour period.
•
It is not known if it is safe to use more than 8 sprays (doses) of ZAVZPRET in 30 days.
•
Avoid using intranasal decongestants with ZAVZPRET. If you have to use an intranasal decongestant, use it at least 1 hour after using ZAVZPRET.
What are the possible side effects of ZAVZPRET?
ZAVZPRET may cause serious side effects including:
•
**Allergic reactions.** Allergic reactions, including trouble breathing, hives, and swelling of the face, can happen after you use ZAVZPRET. Call your healthcare provider or get emergency help right away if you have any of the following symptoms, which may be part of an allergic reaction:
o
swelling of the face, mouth, tongue, or throat
o
trouble breathing
o
rash
o
hives
•
**High blood pressure.** High blood pressure or worsening of high blood pressure can happen after you use ZAVZPRET. Contact your healthcare provider if you have an increase in blood pressure.
•
**Raynaud’s phenomenon.** A type of circulation problem can worsen or happen after you use ZAVZPRET. Raynaud’s phenomenon can lead to your fingers or toes feeling numb, cool, or painful, or changing color from pale, to blue, to red. Contact your healthcare provider if these symptoms occur.
The most common side effects of ZAVZPRET are:
•
unusual taste
•
nausea
•
nasal discomfort
•
vomiting
These are not the only possible side effects of ZAVZPRET.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store ZAVZPRET?
•
Store ZAVZPRET in the blister package that it comes in.
•
Store ZAVZPRET at room temperature between 68°F to 77°F (20°C to 25°C).
•
Do not freeze.
Keep ZAVZPRET and all medicines out of the reach of children.
General information about the safe and effective use of ZAVZPRET:
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ZAVZPRET for a condition for which it was not prescribed. Do not give ZAVZPRET to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ZAVZPRET that is written for health professionals.
What are the ingredients in ZAVZPRET?
Active ingredients in ZAVZPRET: zavegepant
Inactive ingredients in ZAVZPRET: dextrose, hydrochloric acid, sodium hydroxide, and succinic acid in water for injection.

For more information, go to www.zavzpret.com or call 1-800-438-1985.
LAB-1545-3.0
|
This Patient Information has been approved by the US. Food and Drug Administration |
8/2025 |
INSTRUCTIONS FOR USE SECTION
Instructions for Use
|
INSTRUCTIONS FOR USE ZAVZPRET [zav-spret] (zavegepant) nasal spray For Nasal Use Only | |
|
This Instructions for Use contains information on how to give a single dose (10 mg) with ZAVZPRET nasal spray One (1) device delivers a single dose of ZAVZPRET. The device provides 1 spray that should be delivered into 1 nostril. | |
|
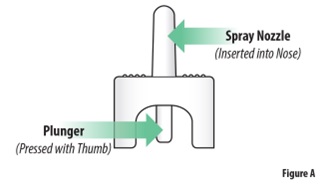
ZAVZPRET Device Parts
| |
|
Important Information You Need to Know Before Dosing with ZAVZPRET • • • • • • • • • Storage Information • • • • Preparing to Dose with ZAVZPRET Nasal Spray | |
|
1 |
Gently Blow Your Nose |
|
1.1 |
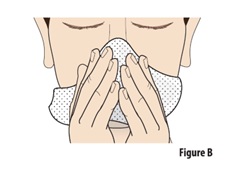
Before using ZAVZPRET, blow your nose to clear your nostrils (seeFigure B). This can be done while sitting or standing. |
|
| |
|
2 |
Remove 1 Blister from the Carton |
|
2.1 |
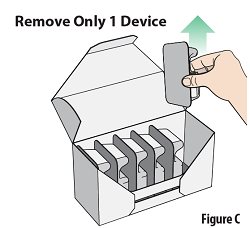
Remove 1 blister containing a device from the carton (seeFigure C). |
|
| |
|
3 |
Check Expiration Date on the Blister |
|
3.1 |
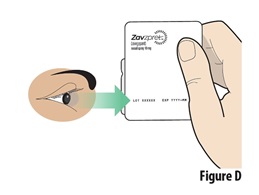
Check the expiration date on the blister containing a device (seeFigure D). |
|
Do not use if the expiration date has passed. Throw away the expired device in the trash if the expiration date has passed. | |
|
| |
|
4 |
Remove Device from Blister |
|
4.1 |
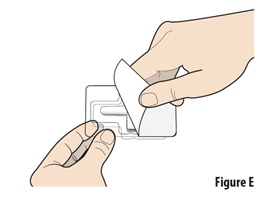
Peel open the blister seal from the corner and remove it completely (see Figure E). |
|
4.2 |
Carefully remove the device from the plastic tray. |
|
| |
|
Dosing with ZAVZPRET Nasal Spray | |
|
5 |
Check Expiration Date on the Device |
|
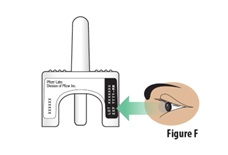
5.1 |
Check the expiration date on the device (seeFigure F). Do not use if the expiration date has passed. Throw away the expired device in the trash if the expiration date has passed. |
|
| |
|
6 |
Grip the Device |
|
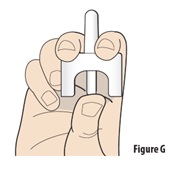
6.1 |
Hold the device upright with your thumb on the bottom of the plunger and two fingers on either side of the nozzle (seeFigure G). Do not press the plunger yet. If you press the plunger now, the medicine will spray and be wasted. |
|
| |
|
Dosing with ZAVZPRET Nasal Spray (Continued) | |
|
7 |
Close 1 Nostril |
|
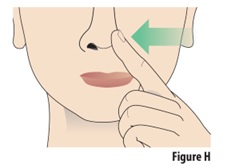
7.1 |
With your free hand, gently press 1 nostril to close it (seeFigure H). |
|
7.2 |
Continue breathing normally through your mouth. |
|
| |
|
8 |
Insert Spray Nozzle Into Open Nostril |
|
8.1 |
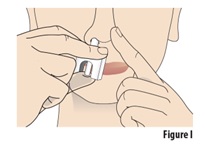
Insert the nozzle into the open nostril as much as you comfortably can (see Figure I). |
|
| |
|
9 |
Deliver the Dose by Spraying Into Nostril and Breathing In |
|
9.1 |
Keep your head level and upright and close your mouth. Do not tilt your head. Do not lay down while delivering the dose. |
|
9.2 |
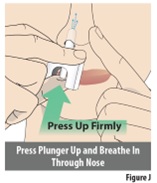
Slowly breathe in through your nose as you firmly press the plunger up with your thumb to release the spray (seeFigure J). Important: Hold the nozzle firmly in your nose as you deliver the dose. Do not let the nozzle come out when pressing the plunger. Spray only 1 time into 1 nostril. |
|
| |
|
10 |
Remove the Device and Keep Your Head Level for 20 Seconds |
|
10.1 |
Remove the device from your nostril and your finger from your other nostril. |
|
10.2 |
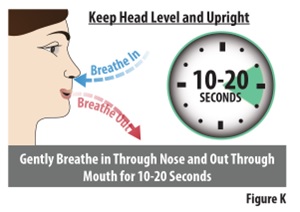
While keeping your head level and upright, gently breathe in through your nose and out through your mouth for 10 to 20 seconds (seeFigure K). |
|
If you feel a drip from your nose, gently sniff so you do not lose any of your dose. | |
|
| |
|
Throwing away (Disposing of) Device | |
|
11 |
Throw Away Used Device |
|
11.1 |
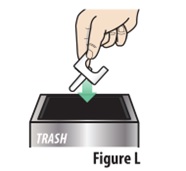
Throw away the used device into trash (seeFigure L) |
|
| |
|
You have received your full dose, if you have: ✓ ✓ Additional Information For more information on ZAVZPRET (zavegepant), visit www.zavzpret.com or call 1-800-438-1985. This Instructions for Use has been approved by the U.S. Food and Drug Administration Approved: 7/2024
LAB-1546-2.0 |
DESCRIPTION SECTION
11 DESCRIPTION
ZAVZPRET (zavegepant) nasal spray contains zavegepant hydrochloride, a calcitonin gene-related peptide receptor antagonist. Zavegepant hydrochloride is described chemically as (R)-N-(3-(7-methyl-1H-indazol-5-yl)-1-(4-(1-methylpiperidin-4-yl) piperazin-1-yl)-1-oxopropan-2-yl)-4-(2-oxo-1,2-dihydroquinolin-3-yl) piperidine-1-carboxamide hydrochloride and its structural formula is:
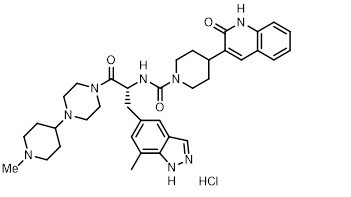
Its molecular formula is C36H46N8O3․HCl, representing a molecular weight of 675.28 g/mol. Zavegepant free base has a molecular weight of 638.82 g/mol. Zavegepant hydrochloride is a white to off-white powder, freely soluble in water, and has pKa values of 4.8 and 8.8.
Each unit-dose ZAVZPRET device for nasal administration delivers 10 mg of zavegepant (equivalent to 10.6 mg of zavegepant hydrochloride) in a buffered aqueous solution containing dextrose, hydrochloric acid, sodium hydroxide, and succinic acid in water for injection. The solution has a pH of 5.3 to 6.7.
INFORMATION FOR PATIENTS SECTION
17 PATIENT COUNSELING INFORMATION
Advise patients to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Hypersensitivity Reactions
Inform patients about the signs and symptoms of hypersensitivity reactions, and that these reactions can occur after administration of ZAVZPRET. Advise patients to contact their healthcare provider immediately if signs or symptoms of hypersensitivity reactions occur [see Warnings and Precautions (5.1)].
Hypertension
Inform patients that hypertension can develop or pre-existing hypertension can worsen with ZAVZPRET, and that they should contact their healthcare provider if they experience elevation in their blood pressure [see Warnings and Precautions (5.2)].
Raynaud’s Phenomenon
Inform patients that Raynaud’s phenomenon can develop or worsen with ZAVZPRET. Advise patients to discontinue ZAVZPRET and contact their healthcare provider if they experience signs or symptoms of Raynaud’s phenomenon [see Warnings and Precautions (5.3)].
Drug Interactions
Advise patients to speak with their healthcare provider about any prescription or over-the-counter medications or herbal supplements that they take or plan to take. Inform patients that if they need to use an intranasal decongestant it should be administered at least 1 hour after ZAVZPRET administration [see Drug Interactions (7.3)].
This product’s labeling may have been updated. For the most recent prescribing information, please visit www.pfizer.com.

LAB-1544-3.0