Goodsense Regular strength Stomach Relief 122
Good sense Regular strength stomach relief 122
f0588ab8-d1fc-419a-8d68-bee42332d50f
HUMAN OTC DRUG LABEL
Aug 1, 2025
Preferred Pharmaceuticals Inc.
DUNS: 791119022
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Bismuth Subsalicylate
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (6)
Drug Labeling Information
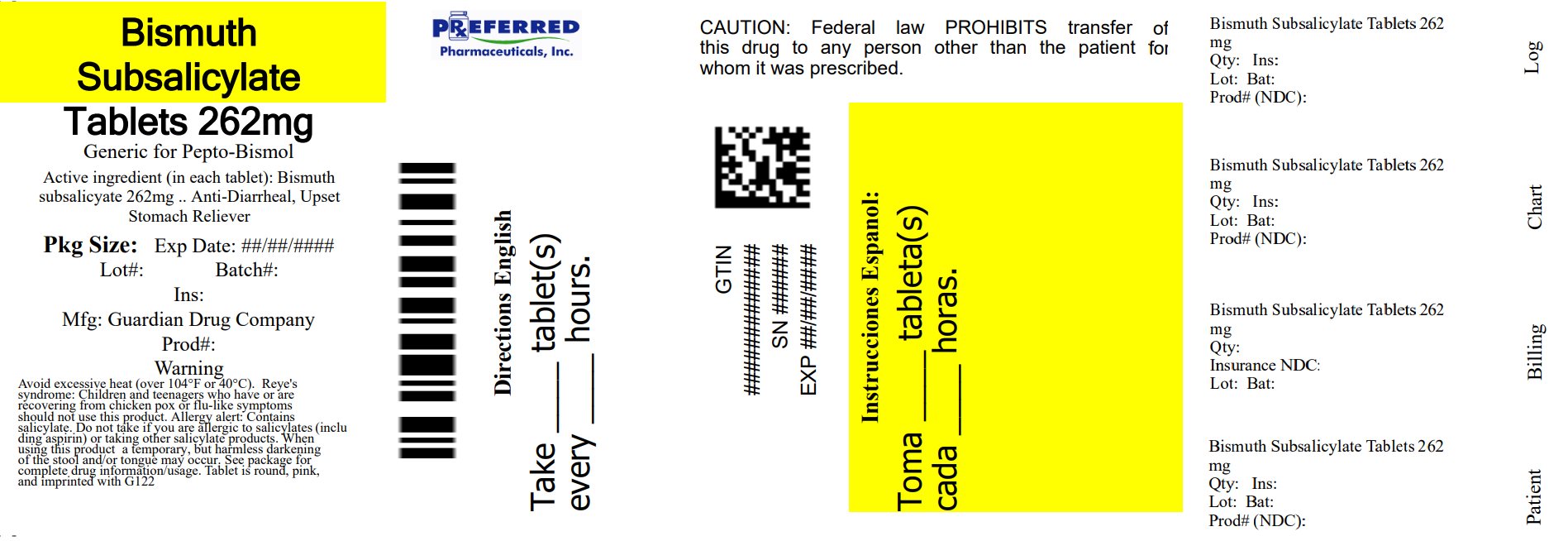
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL
OODSENSE** ®**
Regular Strength
NDC 50804-152-49
Stomach Relief Tablets
Bismuth Subsalicylate 262 mg
Antidiarrheal/Upset Stomach Reliever
Soothing Relief For
5 Symptoms
•
**Upset Stomach**
•
**Indigestion**
•
**Heartburn**
•
**Nausea**
•
**Diarrhea**
Actual Size
*** Compare to active ingredient of Pepto-Bismol****®**** Chewable Tablets**
30 Chewable Tablets
NDC 68788-4002-3
Relabeled By: Preferred Pharmaceuticals Inc.
INDICATIONS & USAGE SECTION
USE(S)
relieves:
•
diarrhea
•
heartburn
•
indigestion
•
nausea
•
upset stomach associated with these symptoms
OTC - ACTIVE INGREDIENT SECTION
ACTIVE INGREDIENT(S)
Bismuth subsalicylate 262 mg (total salicylate 102 mg per tablet)
OTC - PURPOSE SECTION
PURPOSE
Upset stomach reliever and anti-diarrheal
INACTIVE INGREDIENT SECTION
INACTIVE INGRADIENTS
calcium carbonate, D&C red 27 aluminum lake, flavor, Magnesium stearate, mannitol, pregelatinized starch, saccharin sodium.
WARNINGS SECTION
WARNINGS
**Reye's syndrome:**Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye’s syndrome, a rare but serious illness.
____________________________________________________________________________________________________________
Allergy alert: Contains salicylate. Do not take if you are
•
allergic to salicylates (including aspirin)
•
taking other salicylate products
OTC - DO NOT USE SECTION
DO NOT USE
if you have
•
bloody or black stool
•
an ulcer
•
a bleeding problem
OTC - ASK DOCTOR SECTION
ASK A DOCTOR BEFORE USE IF YOU HAVE
•
fever
•
mucus in the stool
OTC - ASK DOCTOR/PHARMACIST SECTION
ASK A DOCTOR OR PHARMACIST BEFORE USE IF YOU ARE
taking any drug for
•
anticoagulation (thinning of the blood)
•
diabetes
•
gout
•
arthritis
OTC - WHEN USING SECTION
WHEN USING THIS PRODUCT
a temporary, but harmless darkening of the stool and/or tongue may occur.
OTC - STOP USE SECTION
STOP USE AND ASK DOCTOR IF
•
symptoms get worse
•
ringing in the ears or loss of hearing occurs
•
diarrhea lasts more than 2 days
OTC - PREGNANCY OR BREAST FEEDING SECTION
IF PREGNANT OR BREAST-FEEDING,
ask a health professional before use.
OTC - KEEP OUT OF REACH OF CHILDREN SECTION
KEEP OUT OF REACH OF CHILDREN
In case of overdose, get medical help or contact a Poison Control Center immediately.
DOSAGE & ADMINISTRATION SECTION
DIRECTIONS
•
chew or dissolve in mouth
•
adults and children 12 years and over: 2 tablets every 1/2 to 1 hour as needed
•
do not take more than 8 doses (16 tablets) in 24 hours
•
children under 12 years: ask a doctor
•
drink plenty of clear fluids to help prevent dehydration caused by diarrhea
•
use until diarrhea stops but not more than 2 days
OTHER SAFETY INFORMATION
OTHER INFORMATION
•
each tablet contains:
•
sodium less than 1 mg
•
salicylate 102 mg
•
very low sodium
•
avoid excessive heat (over 104oF or 40oC).
•
**TAMPER EVIDENT: Do not use if individual compartments are torn or open.**
