Registrants1
Companies and organizations registered with the FDA for this drug approval, including their contact information and regulatory details.
Manufacturing Establishments1
FDA-registered manufacturing facilities and establishments involved in the production, packaging, or distribution of this drug product.
Carlsbad Technology, Inc.
Yung Shin Pharmaceutical Industrual Co., Ltd.
658843289
Products2
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Meloxicam
Product Details
Meloxicam
Product Details
Drug Labeling Information
Complete FDA-approved labeling information including indications, dosage, warnings, contraindications, and other essential prescribing details.
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL
NDC 61422-127-10
Meloxicam
Tablets, USP
15 mg
Rx Only
ATTENTION DISPENSER:
Accompanying Medication Guide must be dispensed with this product.
Carlsbad Technology, Inc.
1000 Tablets

DESCRIPTION SECTION
11 DESCRIPTION
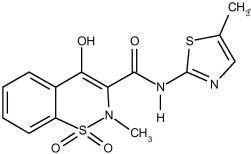
Meloxicam, an oxicam derivative, is a member of the enolic acid group of nonsteroidal anti-inflammatory drugs (NSAIDs). Each pastel yellow Meloxicam Tablets, USP contains 7.5 mg or 15 mg meloxicam for oral administration. Meloxicam is chemically designated as 4-hydroxy-2-methyl- N-(5-methyl-2-thiazolyl)-2 H-1,2-benzothiazine-3-carboxamide-1,1-dioxide. The molecular weight is 351.4. Its empirical formula is C 14H 13N 3O 4S 2 and it has the following structural formula:
Meloxicam is a pastel yellow solid, practically insoluble in water, with higher solubility observed in strong acids and bases. It is very slightly soluble in methanol. Meloxicam has an apparent partition coefficient (log P) app = 0.1 in n-octanol/buffer pH 7.4. Meloxicam has pKa values of 1.1 and 4.2.
Meloxicam is available as a tablet for oral administration containing 7.5 mg or 15 mg meloxicam.
The inactive ingredients in Meloxicam Tablets, USP include Colloidal Silicon Dioxide, Sodium Starch Glycolate, Lactose, Magnesium Stearate, Microcrystalline Cellulose, Povidone K-30, and Sodium Citrate.
INDICATIONS & USAGE SECTION
Highlight: Meloxicam is a non-steroidal anti-inflammatory drug indicated for:
- Osteoarthritis (OA) ( 1.1)
- Rheumatoid Arthritis (RA) ( 1.2)
- Juvenile Rheumatoid Arthritis (JRA) in patients 2 years of age or older ( 1.3)
1 INDICATIONS AND USAGE
1.1 Osteoarthritis (OA)
Meloxicam tablets, USP is indicated for relief of the signs and symptoms of osteoarthritis [ see Clinical Studies ( 14.1) ].
1.2 Rheumatoid Arthritis (RA)
Meloxicam tablets, USP is indicated for relief of the signs and symptoms of rheumatoid arthritis [ see Clinical Studies ( 14.1) ].
1.3 Juvenile Rheumatoid Arthritis (JRA) Pauciarticular and Polyarticular
Course
Meloxicam tablets, USP is indicated for relief of the signs and symptoms of pauciarticular or polyarticular course Juvenile Rheumatoid Arthritis in patients 2 years of age and older [ see Clinical Studies ( 14.2) ].
DOSAGE & ADMINISTRATION SECTION
Highlight: Use the lowest effective dose for the shortest duration consistent with individual treatment goals for the individual patient.
- OA ( 2.2) and RA ( 2.3):
- Starting dose: 7.5 mg once daily
- Dose may be increased to 15 mg once daily
2 DOSAGE AND ADMINISTRATION
2.1 General Instructions
Carefully consider the potential benefits and risks of meloxicam and other treatment options before deciding to use meloxicam. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals [ see Warnings and Precautions ( 5.4) ].
After observing the response to initial therapy with meloxicam, adjust the dose to suit an individual patient's needs.
In adults, the maximum recommended daily oral dose of meloxicam is 15 mg regardless of formulation. In patients with hemodialysis, a maximum daily dosage of 7.5 mg is recommended [ see Warnings and Precautions ( 5.6), Use in Specific Populations ( 8.7), and Clinical Pharmacology ( 12.3) ].
Meloxicam may be taken without regard to timing of meals.
2.2 Osteoarthritis
For the relief of the signs and symptoms of osteoarthritis the recommended starting and maintenance oral dose of meloxicam is 7.5 mg once daily. Some patients may receive additional benefit by increasing the dose to 15 mg once daily.
2.3 Rheumatoid Arthritis
For the relief of the signs and symptoms of rheumatoid arthritis, the recommended starting and maintenance oral dose of meloxicam is 7.5 mg once daily. Some patients may receive additional benefit by increasing the dose to 15 mg once daily.
CONTRAINDICATIONS SECTION
Highlight: * Known hypersensitivity (e.g., anaphylactoid reactions and serious skin reactions) to meloxicam ( 4.1)
- History of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs ( 4.1)
- Use during the peri-operative period in the setting of coronary artery bypass graft (CABG) surgery ( 4.2)
4 CONTRAINDICATIONS
4.1 Allergic Reactions
Meloxicam is contraindicated in patients with known hypersensitivity (e.g., anaphylactoid reactions and serious skin reactions) to meloxicam.
Meloxicam should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients [ see Warnings and Precautions ( 5.7, 5.13) ].
4.2 Coronary Surgery
Meloxicam is contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery [ see Warnings and Precautions ( 5.1) ].
WARNINGS AND PRECAUTIONS SECTION
Highlight: * Serious and potentially fatal cardiovascular (CV) thrombotic events, myocardial infarction, and stroke. Patients with known CV disease/risk factors may be at greater risk. ( 5.1)
- Serious gastrointestinal (GI) adverse events which can be fatal. The risk is greater in patients with a prior history of ulcer disease or GI bleeding, and in patients at higher risk for GI events, especially the elderly. ( 5.2)
- Elevated liver enzymes, and rarely, severe hepatic reactions. Discontinue use immediately if abnormal liver enzymes persist or worsen. ( 5.3)
- New onset or worsening of hypertension. Blood pressure should be monitored closely during treatment. ( 5.4)
- Fluid retention and edema. Should be used with caution in patients with fluid retention or heart failure. ( 5.5)
- Renal papillary necrosis and other renal injury with long-term use. Use with caution in the elderly, those with impaired renal function, heart failure, liver dysfunction, and those taking diuretics, ACE-inhibitors, or angiotensin II antagonists. The use of meloxicam in patients with severe renal impairment is not recommended. ( 5.6)
- Serious skin adverse events such as exfoliative dermatitis, Stevens-Johnson syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal and can occur without warning. Discontinue meloxicam at first appearance of rash or skin reactions. ( 5.8)
5 WARNINGS AND PRECAUTIONS
5.1 Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years' duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, myocardial infarction, and stroke, which can be fatal. All NSAIDs, both COX-2 selective and nonselective, may have a similar risk. Patients with known CV disease or risk factors for CV disease may be at greater risk. To minimize the potential risk for an adverse CV event in patients treated with an NSAID, the lowest effective dose should be used for the shortest duration possible. Physicians and patients should remain alert for the development of such events, even in the absence of previous CV symptoms. Patients should be informed about the signs and/or symptoms of serious CV events and the steps to take if they occur.
Two large, controlled, clinical trials of a COX-2 selective NSAID for the treatment of pain in the first 10 to 14 days following CABG surgery found an increased incidence of myocardial infarction and stroke [ see Contraindications ( 4.2) ].
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID does increase the risk of serious GI events [ see Warnings and Precautions ( 5.2) ].
5.2 Gastrointestinal (GI) Effects - Risk of GI Ulceration, Bleeding, and
Perforation
NSAIDs, including meloxicam, can cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration, and perforation of the stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs. Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs, occur in approximately 1% of patients treated for 3 to 6 months, and in about 2 to 4% of patients treated for one year. These trends continue with longer duration of use, increasing the likelihood of developing a serious GI event at some time during the course of therapy. However, even short-term therapy is not without risk.
Prescribe NSAIDs, including meloxicam, with extreme caution in those with a prior history of ulcer disease or gastrointestinal bleeding. Patients with a prior history of peptic ulcer disease and/or gastrointestinal bleeding who use NSAIDs have a greater than 10-fold increased risk for developing a GI bleed compared to patients with neither of these risk factors. Other factors that increase the risk for GI bleeding in patients treated with NSAIDs include concomitant use of oral corticosteroids or anticoagulants, longer duration of NSAID therapy, smoking, use of alcohol, older age, and poor general health status. Most spontaneous reports of fatal GI events are in elderly or debilitated patients and therefore, special care should be taken in treating this population.
To minimize the potential risk for an adverse GI event in patients treated with an NSAID, use the lowest effective dose for the shortest possible duration. Patients and physicians should remain alert for signs and symptoms of GI ulceration and bleeding during meloxicam therapy and promptly initiate additional evaluation and treatment if a serious GI adverse event is suspected. This should include discontinuation of meloxicam until a serious GI adverse event is ruled out. For high-risk patients, consider alternate therapies that do not involve NSAIDs.
5.3 Hepatic Effects
Borderline elevations of one or more liver tests may occur in up to 15% of patients taking NSAIDs including meloxicam. These laboratory abnormalities may progress, may remain unchanged, or may be transient with continuing therapy. Notable elevations of ALT or AST (approximately three or more times the upper limit of normal) have been reported in approximately 1% of patients in clinical trials with NSAIDs. In addition, rare cases of severe hepatic reactions, including jaundice and fatal fulminant hepatitis, liver necrosis and hepatic failure, some of them with fatal outcomes have been reported [ see Adverse Reactions ( 6.1) ].
A patient with symptoms and/or signs suggesting liver dysfunction, or in whom an abnormal liver test has occurred, should be evaluated for evidence of the development of a more severe hepatic reaction while on therapy with meloxicam. If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), discontinue meloxicam [ see Use in Specific Populations ( 8.6) and Clinical Pharmacology ( 12.3) ].
5.4 Hypertension
NSAIDs, including meloxicam, can lead to onset of new hypertension or worsening of pre-existing hypertension, either of which may contribute to the increased incidence of CV events. NSAIDs, including meloxicam, should be used with caution in patients with hypertension. Blood pressure (BP) should be monitored closely during the initiation of NSAID treatment and throughout the course of therapy.
Patients taking ACE inhibitors, thiazides, or loop diuretics may have impaired response to these therapies when taking NSAIDs.
5.5 Congestive Heart Failure and Edema
Fluid retention and edema have been observed in some patients taking NSAIDs. Use meloxicam with caution in patients with fluid retention, hypertension, or heart failure.
5.6 Renal Effects
Long-term administration of NSAIDs, including meloxicam, can result in renal papillary necrosis, renal insufficiency, acute renal failure, and other renal injury. Renal toxicity has also been seen in patients in whom renal prostaglandins have a compensatory role in the maintenance of renal perfusion. In these patients, administration of a nonsteroidal anti-inflammatory drug may cause a dose-dependent reduction in prostaglandin formation and, secondarily, in renal blood flow, which may precipitate overt renal decompensation. Patients at greatest risk of this reaction are those with impaired renal function, heart failure, liver dysfunction, those taking diuretics, ACE- inhibitors, and angiotensin II receptor antagonists, and the elderly. Discontinuation of NSAID therapy is usually followed by recovery to the pretreatment state.
A pharmacokinetic study in patients with mild and moderate renal impairment revealed that no dosage adjustments in these patient populations are required. Patients with severe renal impairment have not been studied. The use of meloxicam in patients with severe renal impairment with CrCl less than 20 mL/min is not recommended. A study performed in patients on hemodialysis revealed that although overall C max was diminished in this population, the proportion of free drug not bound to plasma was increased. Therefore it is recommended that meloxicam dosage in this population not exceed 7.5 mg per day. Closely monitor the renal function of patients with impaired renal function who are taking meloxicam [ see Dosage and Administration ( 2.1), Use in Specific Populations ( 8.7), and Clinical Pharmacology ( 12.3) ].
Use caution when initiating treatment with meloxicam in patients with considerable dehydration. It is advisable to rehydrate patients first and then start therapy with meloxicam. Caution is also recommended in patients with pre-existing kidney disease.
The extent to which metabolites may accumulate in patients with renal impairment has not been studied with meloxicam. Because some meloxicam metabolites are excreted by the kidney, monitor patients with significant renal impairment closely.
5.7 Anaphylactoid Reactions
As with other NSAIDs, anaphylactoid reactions have occurred in patients without known prior exposure to meloxicam. Meloxicam should not be given to patients with the aspirin triad. This symptom complex typically occurs in asthmatic patients who experience rhinitis with or without nasal polyps, or who exhibit severe, potentially fatal bronchospasm after taking aspirin or other NSAIDs [ see Contraindications ( 4.1) and Warnings and Precautions ( 5.12) ]. Seek emergency help in cases where an anaphylactoid reaction occurs.
5.8 Adverse Skin Reactions
NSAIDs, including meloxicam, can cause serious skin adverse events such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. These serious events may occur without warning. Inform patients about the signs and symptoms of serious skin manifestations and discontinue use of the drug at the first appearance of skin rash or any other sign of hypersensitivity.
5.9 Pregnancy
Starting at 30 weeks gestation, avoid the use of meloxicam because it may cause premature closure of the ductus arteriosus [ see Use in Specific Populations ( 8.1) and Patient Counseling Information ( 17.8) ].
5.10 Corticosteroid Treatment
Meloxicam cannot be expected to substitute for corticosteroids or to treat corticosteroid insufficiency. Abrupt discontinuation of corticosteroids may lead to disease exacerbation. Slowly taper patients on prolonged corticosteroid therapy if a decision is made to discontinue corticosteroids.
5.11 Masking of Inflammation and Fever
The pharmacological activity of meloxicam in reducing fever and inflammation may diminish the utility of these diagnostic signs in detecting complications of presumed noninfectious, painful conditions.
5.12 Hematological Effects
Anemia may occur in patients receiving NSAIDs, including meloxicam. This may be due to fluid retention, occult or gross GI blood loss, or an incompletely described effect upon erythropoiesis. Patients on long-term treatment with NSAIDs, including meloxicam, should have their hemoglobin or hematocrit checked if they exhibit any signs or symptoms of anemia.
NSAIDs inhibit platelet aggregation and have been shown to prolong bleeding time in some patients. Unlike aspirin, their effect on platelet function is quantitatively less, of shorter duration, and reversible. Carefully monitor patients treated with meloxicam who may be adversely affected by alterations in platelet function, such as those with coagulation disorders or patients receiving anticoagulants.
5.13 Use in Patients with Pre-existing Asthma
Patients with asthma may have aspirin-sensitive asthma. The use of aspirin in patients with aspirin-sensitive asthma has been associated with severe bronchospasm, which can be fatal. Since cross reactivity, including bronchospasm, between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, meloxicam should not be administered to patients with this form of aspirin sensitivity and should be used with caution in patients with pre-existing asthma.
5.14 Monitoring
Because serious GI tract ulcerations and bleeding can occur without warning symptoms, physicians should monitor for signs or symptoms of GI bleeding. Patients on long-term treatment with NSAIDs should have their CBC and a chemistry profile checked periodically. If clinical signs and symptoms consistent with liver or renal disease develop, systemic manifestations occur (e.g., eosinophilia, rash, etc.) or if abnormal liver tests persist or worsen, meloxicam should be discontinued.
USE IN SPECIFIC POPULATIONS SECTION
Highlight: * Based on animal data, may cause fetal harm. Starting at 30 weeks gestation, meloxicam should be avoided as premature closure of the ductus arteriosus in the fetus may occur. ( 5.9, 8.1)
- Nursing Mothers: Use with caution, as meloxicam may be excreted in human milk ( 8.3)
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C; Category D starting 30 weeks gestation
There are no adequate and well-controlled studies in pregnant women. Meloxicam crosses the placental barrier. Prior to 30 weeks gestation, use meloxicam during pregnancy only if the potential benefit justifies the potential risk to the fetus. Starting at 30 weeks gestation, avoid meloxicam and other NSAIDs, in pregnant women as premature closure of the ductus arteriosus in the fetus may occur. If this drug is used during this time period in pregnancy, inform the patient of the potential hazard to a fetus [ see Warnings and Precautions ( 5.9) and Patient Counseling Information ( 17.8) ].
Teratogenic Effects
Meloxicam was not teratogenic when administered to pregnant rats during fetal organogenesis at oral doses up to 4 mg/kg/day (2.6-fold greater than the maximum recommended human daily dose [MRHD] based on body surface area [BSA] comparison). Administration of meloxicam to pregnant rabbits throughout embryogenesis produced an increased incidence of septal defects of the heart at an oral dose of 60 mg/kg/day. The no effect level was 20 mg/kg/day (26-fold greater than the MRHD based on BSA conversion).
Nonteratogenic Effects
In rats and rabbits, embryolethality occurred at oral meloxicam doses of 1 mg/kg/day and 5 mg/kg/day, respectively (0.65-and 6.5-fold greater, respectively, than the MRHD based on BSA comparison) when administered throughout organogenesis.
8.2 Labor and Delivery
The effects of meloxicam on labor and delivery of pregnant women are unknown. Oral administration of meloxicam to pregnant rats during late gestation through lactation increased the incidence of dystocia, delayed parturition, and decreased offspring survival at meloxicam doses of 0.125 mg/kg/day or greater (at least 12.5 times lower than the maximum recommended human daily dose based on body surface area comparison).
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk; however, meloxicam was excreted in the milk of lactating rats at concentrations higher than those in plasma. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from meloxicam, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
The safety and effectiveness of meloxicam in pediatric JRA patients from 2 to 17 years of age has been evaluated in three clinical trials [ see Dosage and Administration ( 2.3), Adverse Reactions ( 6.1), and Clinical Studies ( 14.2) ]
8.5 Geriatric Use
As with any NSAID, caution should be exercised in treating the elderly (65 years and older).
Of the total number of subjects in clinical studies, 5157 were age 65 and over (4044 in OA studies and 1113 in RA studies). No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Hepatic Impairment
No dose adjustment is necessary in patients with mild to moderate hepatic impairment. Patients with severe hepatic impairment have not been adequately studied. Since meloxicam is significantly metabolized in the liver; the use of meloxicam in these patients should be done with caution [ see Warnings and Precautions ( 5.3) and Clinical Pharmacology ( 12.3) ].
8.7 Renal Impairment
No dose adjustment is necessary in patients with mild to moderate renal impairment. Patients with severe renal impairment have not been studied. The use of meloxicam in subjects with severe renal impairment is not recommended. Following a single dose of meloxicam, the free C max plasma concentrations were higher in patients with renal failure on chronic hemodialysis (1% free fraction) in comparison to healthy volunteers (0.3% free fraction). Therefore, it is recommended that meloxicam dosage in this population not exceed 7.5 mg per day Hemodialysis did not lower the total drug concentration in plasma; therefore, additional doses are not necessary after hemodialysis. Meloxicam is not dialyzable [ see Dosage and Administration ( 2.1), Warnings and Precautions ( 5.6), and Clinical Pharmacology ( 12.3) ].
8.8 Females of Reproductive Potential
Data from several small studies in humans and from studies in animals indicate that NSAIDs, including Meloxicam , may be associated with a reversible delay in ovulation. Therefore, in women who have difficulties conceiving, or who are undergoing investigation of infertility, use of meloxicam is not recommended.
CLINICAL STUDIES SECTION
14 CLINICAL STUDIES
14.1 Osteoarthritis and Rheumatoid Arthritis
The use of meloxicam for the treatment of the signs and symptoms of osteoarthritis of the knee and hip was evaluated in a 12-week, double-blind, controlled trial. Meloxicam (3.75 mg, 7.5 mg, and 15 mg daily) was compared to placebo. The four primary endpoints were investigator's global assessment, patient global assessment, patient pain assessment, and total WOMAC score (a self-administered questionnaire addressing pain, function, and stiffness). Patients on meloxicam 7.5 mg daily and meloxicam 15 mg daily showed significant improvement in each of these endpoints compared with placebo.
The use of meloxicam for the management of signs and symptoms of osteoarthritis was evaluated in six double-blind, active-controlled trials outside the U.S. ranging from 4 weeks' to 6 months' duration. In these trials, the efficacy of meloxicam, in doses of 7.5 mg/day and 15 mg/day, was comparable to piroxicam 20 mg/day and diclofenac SR 100 mg/day and consistent with the efficacy seen in the U.S. trial.
The use of meloxicam for the treatment of the signs and symptoms of rheumatoid arthritis was evaluated in a 12-week, double-blind, controlled multinational trial. Meloxicam (7.5 mg, 15 mg, and 22.5 mg daily) was compared to placebo. The primary endpoint in this study was the ACR20 response rate, a composite measure of clinical, laboratory, and functional measures of RA response. Patients receiving meloxicam 7.5 mg and 15 mg daily showed significant improvement in the primary endpoint compared with placebo. No incremental benefit was observed with the 22.5 mg dose compared to the 15 mg dose.
14.2 Juvenile Rheumatoid Arthritis (JRA) Pauciarticular and Polyarticular
Course
The use of meloxicam for the treatment of the signs and symptoms of pauciarticular or polyarticular course Juvenile Rheumatoid Arthritis in patients 2 years of age and older was evaluated in two 12-week, double-blind, parallel-arm, active-controlled trials.
Both studies included three arms: naproxen and two doses of meloxicam. In both studies, meloxicam dosing began at 0.125 mg/kg/day (7.5 mg maximum) or 0.25 mg/kg/day (15 mg maximum), and naproxen dosing began at 10 mg/kg/day. One study used these doses throughout the 12-week dosing period, while the other incorporated a titration after 4 weeks to doses of 0.25 mg/kg/day and 0.375 mg/kg/day (22.5 mg maximum) of meloxicam and 15 mg/kg/day of naproxen.
The efficacy analysis used the ACR Pediatric 30 responder definition, a composite of parent and investigator assessments, counts of active joints and joints with limited range of motion, and erythrocyte sedimentation rate. The proportion of responders were similar in all three groups in both studies, and no difference was observed between the meloxicam dose groups.
NONCLINICAL TOXICOLOGY SECTION
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis: There was no increase in tumor incidence in long-term carcinogenicity studies in rats (104 weeks) and mice (99 weeks) administered meloxicam at oral doses up to 0.8 mg/kg/day in rats and up to 8.0 mg/kg/day in mice (up to 0.5- and 2.6-fold, respectively, the maximum recommended human daily dose based on body surface area comparison).
Mutagenesis: Meloxicam was not mutagenic in an Ames assay, or clastogenic in a chromosome aberration assay with human lymphocytes and an in vivo micronucleus test in mouse bone marrow.
Impairment of Fertility: Meloxicam did not impair male and female fertility in rats at oral doses up to 9 mg/kg/day in males and 5 mg/kg/day in females (up to 5.8- and 3.2-fold greater, respectively, than the maximum recommended human daily dose based on body surface area comparison).
INFORMATION FOR PATIENTS SECTION
17 PATIENT COUNSELING INFORMATION
See FDA-approved Medication Guide
Patients should be informed of the following information before initiating therapy with an NSAID and periodically during the course of ongoing therapy.
17.1 Medication Guide
Inform patients of the availability of a Medication Guide for NSAIDs that accompanies each prescription dispensed, and instruct them to read the Medication Guide prior to using meloxicam.
17.2 Cardiovascular Effects
NSAIDs including meloxicam may cause serious CV side effects, such as MI or stroke, which may result in hospitalization and even death. Although serious CV events can occur without warning symptoms, patients should be alert for the signs and symptoms of chest pain, shortness of breath, weakness, slurring of speech, and should ask for medical advice when observing any indicative sign or symptoms. Patients should be apprised of the importance of this follow-up [ see Warnings and Precautions ( 5.1) ].
17.3 Gastrointestinal Effects
NSAIDs including meloxicam, can cause GI discomfort and, rarely, serious GI side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms including epigastric pain, dyspepsia, melena, and hematemesis. Patients should be apprised of the importance of this follow- up [ see Warnings and Precautions ( 5.2) ].
17.4 Hepatotoxicity
Inform patients of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If these occur, instruct patients to stop therapy and seek immediate medical therapy [ see Warnings and Precautions ( 5.3) ].
17.5 Adverse Skin Reactions
NSAIDs, including meloxicam, can cause serious skin side effects such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which may result in hospitalization and even death. Although serious skin reactions may occur without warning, patients should be alert for the signs and symptoms of skin rash and blisters, fever, or other signs of hypersensitivity such as itching, and should ask for medical advice when observing any indicative signs or symptoms. Advise patients to stop the drug immediately if they develop any type of rash and contact their physicians as soon as possible [ see Warnings and Precautions ( 5.8) ].
17.6 Weight Gain and Edema
Advise patients to promptly report signs or symptoms of unexplained weight gain or edema to their physicians [ see Warnings and Precautions ( 5.5) ].
17.7 Anaphylactoid Reactions
Inform patients of the signs of an anaphylactoid reaction (e.g., difficulty breathing, swelling of the face or throat). Instruct patients seek immediate emergency help [ see Warnings and Precautions ( 5.7) ].
17.8 Effects During Pregnancy
Starting at 30 weeks gestation, meloxicam should be avoided as premature closure of the ductus arteriosus in the fetus may occur [see Warnings and Precautions ( 5.9) and Use in Specific Populations ( 8.1)].
17.9 Effects On Female Fertility
Advise females of reproductive potential who desire pregnancy that NSAIDs, including Meloxicam, may be associated with a reversible delay in ovulation For women who have difficulties conceiving, or who are undergoing investigation of infertility, use of meloxicam is not recommended [ see Use in Specific Populations 8.8) ].
Please address medical inquiries to 1-760-431-8284
Manufactured by:
Yung Shin Pharmaceutical Ind. Co., Ltd.
Tachia, Taichung 43769
TAIWAN
Distributed by:
Carlsbad Technology, Inc.
5923 Balfour Ct.
Carlsbad, CA 92008 USA
Revised: 08/13
SPL MEDGUIDE SECTION
MEDICATION GUIDE
MELOXICAM Tablets, USP
Medication Guide for Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
(See the end of this Medication Guide for a list of prescription NSAID
medicines.)
What is the most important information I should know about medicines called Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)?
**NSAID medicines may increase the chance of a heart attack or stroke that can lead to death.**This chance increases:
- with longer use of NSAID medicines
- in people who have heart disease
NSAID medicines should never be used right before or after a heart surgery called a "coronary artery bypass graft (CABG)."
NSAID medicines can cause ulcers and bleeding in the stomach and intestines at any time during treatment. Ulcers and bleeding:
- can happen without warning symptoms
- may cause death
The chance of a person getting an ulcer or bleeding increases with:
- taking medicines called "corticosteroids" and "anticoagulants"
- longer use
- smoking
- drinking alcohol
- older age
- having poor health
NSAID medicines should only be used:
- exactly as prescribed
- at the lowest dose possible for your treatment
- for the shortest time needed
What are Non-Steroidal Anti- Inflammatory Drugs (NSAIDs)?
NSAID medicines are used to treat pain and redness, swelling, and heat (inflammation) from medical conditions such as:
- different types of arthritis
- menstrual cramps and other types of short-term pain
Who should not take a Non-Steroidal Anti-Inflammatory Drug (NSAID)?
Do not take an NSAID medicine:
- if you had an asthma attack, hives, or other allergic reaction with aspirin or any other NSAID medicine
- for pain right before or after heart bypass surgery
Tell your healthcare provider:
- about all of your medical conditions.
- about all of the medicines you take. NSAIDs and some other medicines can interact with each other and cause seri- ous side effects.Keep a list of your medicines to show to your healthcare provider and pharmacist.
- if you are pregnant.NSAID medicines should not be used by pregnant women late in their pregnancy.
- if you are breastfeeding.Talk to your doctor.
|
What are the possible side effects of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)? Serious side effects include:
Other side effects include:
|
Get emergency help right away if you have any of the following symptoms:
- shortness of breath or trouble breathing
- chest pain
- weakness in one part or side of your body
- slurred speech
- swelling of the face or throat
Stop your NSAID medicine and call your healthcare provider right away if you have any of the following symptoms:
- nausea
- more tired or weaker than usual
- itching
- your skin or eyes look yellow
- stomach pain
- flu-like symptoms
- vomit blood
- there is blood in your bowel movement or it is black and sticky like tar
- unusual weight gain
- skin rash or blisters with fever
- swelling of the arms and legs, hands and feet
These are not all the side effects with NSAID medicines. Talk to your healthcare provider or pharmacist for more information about NSAID medicines.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Other information about Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
- Aspirin is an NSAID medicine but it does not increase the chance of a heart attack. Aspirin can cause bleeding in the brain, stomach, and intestines. Aspirin can also cause ulcers in the stomach and intestines.
- Some of these NSAID medicines are sold in lower doses without a prescription (over-the¬counter). Talk to your healthcare provider before using over-the-counter NSAIDs for more than 10 days.
NSAID medicines that need a prescription
|
Generic Name |
** Tradename** |
|
Celecoxib |
Celebrex |
|
Diclofenac |
Cataflam, Voltaren, Arthrotec (combined with misoprostol) |
|
Diflunisal |
Dolobid |
|
Etodolac |
Lodine, Lodine XL |
|
Fenoprofen |
Nalfon, Nalfon 200 |
|
Flurbiprofen |
Ansaid |
|
Ibuprofen |
Motrin, Tab-Profen, Vicoprofen* (combined with hydrocodone), Combunox (combined with oxycodone) |
|
Indomethacin |
Indocin, Indocin SR, Indo-Lemmon, Indomethagan |
|
Ketoprofen |
Oruvail |
|
Ketorolac |
Toradol |
|
Mefenamic Acid |
Ponstel |
|
Meloxicam |
Mobic |
|
Nabumetone |
Relafen |
|
Naproxen |
Naprosyn, Anaprox, Anaprox DS, EC-Naprosyn, Naprelan, Naprapac (co-packaged with lansoprazole) |
|
Oxaprozin |
Daypro |
|
Piroxicam |
Feldene |
|
Sulindac |
Clinoril |
|
Tolmetin |
Tolectin, Tolectin DS, Tolectin 600 |
*Vicoprofen contains the same dose of ibuprofen as over-the-counter (OTC) NSAIDs, and is usually used for less than 10 days to treat pain. The OTC NSAID label warns that long term continuous use may increase the risk of heart attack or stroke.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Manufactured by:
Yung Shin Pharmaceutical Ind. Co., Ltd.
Tachia, Taichung 43769
TAIWAN
Distributed by:
Carlsbad Technology, Inc.
5923 Balfour Ct.
Carlsbad, CA 92008 USA
Revised: 08/13
HOW SUPPLIED SECTION
16 HOW SUPPLIED/STORAGE AND HANDLING
Meloxicam is available as a pastel yellow, round, biconvex, uncoated tablet containing meloxicam 7.5 mg or 15 mg. The 7.5 mg tablet is impressed with “5” mark on one side, and the 15 mg tablet is impressed with “100” mark on one side.
Meloxicam Tablets, USP 7.5 mg is available as follows:
NDC 61442-126-30; Bottles of 30
NDC 61442-126-01; Bottles of 100
NDC 61442-126-10; Bottles of 1000
Meloxicam Tablets, USP 15 mg is available as follows:
NDC 61442-127-30; Bottles of 30
NDC 61442-127-01; Bottles of 100
NDC 61442-127-10; Bottles of 1000
Storage
Store at 20° to 25°C (68° to 77°F)[see USP Controlled Room Temperature].
Keep Meloxicam Tablets, USP in a dry place.
Dispense tablets in a tight container.
Keep this and all medications out of the reach of children.
