Azithromycin
These highlights do not include all the information needed to use AZITHROMYCIN TABLETS safely and effectively. See full prescribing information for AZITHROMYCIN TABLETS. AZITHROMYCIN tablets, for oral use Initial U.S. Approval: 1991
d5c95c30-564f-4b67-ac6d-9179cdfaa5a8
HUMAN PRESCRIPTION DRUG LABEL
Jan 13, 2023
Alembic Pharmaceuticals Limited
DUNS: 650574663
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Azithromycin
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (11)
Drug Labeling Information
INDICATIONS & USAGE SECTION
1 INDICATIONS AND USAGE
Azithromycin tablets are a macrolide antibacterial drug indicated for the treatment of patients with mild to moderate infections caused by susceptible strains of the designated microorganisms in the specific conditions listed below. Recommended dosages and durations of therapy in adult and pediatric patient populations vary in these indications [see Dosage and Administration (2)].
1.1 Adult Patients
· Acute bacterial exacerbations of chronic bronchitis due to Haemophilus influenzae, Moraxella catarrhalis, or Streptococcus pneumoniae.
· Acute bacterial sinusitis due to Haemophilus influenzae, Moraxella catarrhalis. or Streptococcus pneumoniae.
· Community-acquired pneumonia due to Chlamydophila pneumoniae, Haemophilus influenzae, Mycoplasma pneumoniae, or Streptococcus pneumoniae in patients appropriate for oral therapy.
· Pharyngitis/tonsillitis caused by Streptococcus pyogenes as an alternative to first-line therapy in individuals who cannot use first-line therapy.
· Uncomplicated skin and skin structure infections due to Staphylococcus aureus, Streptococcus pyogenes, or Streptococcus agalactiae.
· Urethritis and cervicitis due to Chlamydia trachomatis or Neisseria gonorrhoeae.
· Genital ulcer disease in men due to Haemophilus ducreyi (chancroid). Due to the small number of women included in clinical trials, the efficacy of azithromycin in the treatment of chancroid in women has not been established.
1.2 Pediatric Patients
[see Use in Specific Populations (8.4) and Clinical Studies (14.2)]
- Acute otitis media**(****>6 months of age)** caused by Haemophilus influenzae, Moraxella catarrhalis, or Streptococcus pneumoniae.
- Community-acquired pneumonia**(>6 months of age)** due to Chlamydophila pneumoniae, Haemophilus influenzae, Mycoplasma pneumoniae, or Streptococcus pneumoniae in patients appropriate for oral therapy.
- Pharyngitis/tonsillitis**(>2 years of age****)** caused by Streptococcus pyogenes as an alternative to first-line therapy in individuals who cannot use first-line therapy.
1.3 Limitations of Use
Azithromycin should not be used in patients with pneumonia who are judged to be inappropriate for oral therapy because of moderate to severe illness or risk factors such as any of the following:
· patients with cystic fibrosis,
· patients with nosocomial infections,
· patients with known or suspected bacteremia,
· patients requiring hospitalization,
· elderly or debilitated patients, or
· patients with significant underlying health problems that may compromise their ability to respond to their illness (including immunodeficiency or functional asplenia).
1.4 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of azithromycin tablets and other antibacterial drugs, azithromycin tablets should be used only to treat infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
CONTRAINDICATIONS SECTION
4 CONTRAINDICATIONS
4.1 Hypersensitivity
Azithromycin tablets are contraindicated in patients with known hypersensitivity to azithromycin, erythromycin, any macrolide or ketolide drug.
4.2 Hepatic Dysfunction
Azithromycin tablets are contraindicated in patients with a history of cholestatic jaundice/hepatic dysfunction associated with prior use of azithromycin.
· Patients with known hypersensitivity to azithromycin, erythromycin, any macrolide or ketolide drug. (4.1) (4)
· Patients with a history of cholestatic jaundice/hepatic dysfunction associated with prior use of azithromycin. (4.2) (4)
ADVERSE REACTIONS SECTION
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In clinical trials, most of the reported side effects were mild to moderate in severity and were reversible upon discontinuation of the drug. Potentially serious adverse reactions of angioedema and cholestatic jaundice were reported. Approximately 0.7% of the patients (adults and pediatric patients) from the 5-day multiple-dose clinical trials discontinued azithromycin therapy because of treatment-related adverse reactions. In adults given 500 mg/day for 3 days, the discontinuation rate due to treatment-related adverse reactions was 0.6%. In clinical trials in pediatric patients given 30 mg/kg, either as a single dose or over 3 days, discontinuation from the trials due to treatment- related adverse reactions was approximately 1%. Most of the adverse reactions leading to discontinuation were related to the gastrointestinal tract, e.g., nausea, vomiting, diarrhea, or abdominal pain [see Clinical Studies (14.2)].
Adults
Multiple-dose regimens: Overall, the most common treatment-related adverse reactions in adult patients receiving multiple-dose regimens of azithromycin were related to the gastrointestinal system with diarrhea/loose stools (4 to 5%), nausea (3%), and abdominal pain (2 to 3%) being the most frequently reported.
No other adverse reactions occurred in patients on the multiple-dose regimens of azithromycin with a frequency greater than 1%. Adverse reactions that occurred with a frequency of 1% or less included the following:
Cardiovascular:Palpitations, chest pain.
Gastrointestinal:Dyspepsia, flatulence, vomiting, melena, and cholestatic jaundice.
Genitourinary:Monilia, vaginitis, and nephritis.
Nervous System:Dizziness, headache, vertigo, and somnolence.
General:Fatigue.
Allergic:Rash, pruritus, photosensitivity, and angioedema.
Single 1-gram dose regimen:
Overall, the most common adverse reactions in patients receiving a single-dose regimen of 1 gram of azithromycin were related to the gastrointestinal system and were more frequently reported than in patients receiving the multiple-dose regimen.
Adverse reactions that occurred in patients on the single 1-gram dosing regimen of azithromycin with a frequency of 1% or greater included diarrhea/loose stools (7%), nausea (5%), abdominal pain (5%), vomiting (2%), dyspepsia (1%), and vaginitis (1%).
Single 2-gram dose regimen:
Overall, the most common adverse reactions in patients receiving a single 2-gram dose of azithromycin were related to the gastrointestinal system. Adverse reactions that occurred in patients in this study with a frequency of 1% or greater included nausea (18%), diarrhea/loose stools (14%), vomiting (7%), abdominal pain (7%), vaginitis (2%), dyspepsia (1%), and dizziness (1%). The majority of these complaints were mild in nature.
Pediatric Patients
Single and Multiple-dose regimens: The types of adverse reactions in pediatric patients were comparable to those seen in adults, with different incidence rates for the dosage regimens recommended in pediatric patients.
Acute Otitis Media:For the recommended total dosage regimen of 30 mg/kg, the most frequent adverse reactions (≥1%) attributed to treatment were diarrhea, abdominal pain, vomiting, nausea, and rash [see Dosage and Administration (2) and Clinical Studies (14.2)].
The incidence, based on dosing regimen, is described in the table below:
|
Dosage****Regimen |
Diarrhea**%** |
Abdominal****Pain % |
Vomiting**%** |
Nausea**%** |
Rash**%** |
|
1-day |
4.3% |
1.4% |
4.9% |
1% |
1% |
|
3-day |
2.6% |
1.7% |
2.3% |
0.4% |
0.6% |
|
5-day |
1.8% |
1.2% |
1.1% |
0.5% |
0.4% |
Community-Acquired Pneumonia:For the recommended dosage regimen of 10 mg/kg on Day 1 followed by 5 mg/kg on Days 2 to 5, the most frequent adverse reactions attributed to treatment were diarrhea/loose stools, abdominal pain, vomiting, nausea, and rash.
The incidence is described in the table below:
|
Dosage****Regimen |
Diarrhea/Loose****stools % |
Abdominal****Pain % |
Vomiting**%** |
Nausea******%** |
Rash******%** |
|
5-day |
5.8% |
1.9% |
1.9% |
1.9% |
1.6% |
Pharyngitis/Tonsillitis:For the recommended dosage regimen of 12 mg/kg on Days 1 to 5, the most frequent adverse reactions attributed to treatment were diarrhea, vomiting, abdominal pain, nausea, and headache.
The incidence is described in the table below:
|
Dosage****Regimen |
Diarrhea**%** |
Abdominal****Pain % |
Vomiting**%** |
Nausea******%** |
Rash**%** |
Headache**%** |
|
5-day |
5.4% |
3.4% |
5.6% |
1.8% |
0.7% |
1.1% |
With any of the treatment regimens, no other adverse reactions occurred in pediatric patients treated with azithromycin with a frequency greater than 1%. Adverse reactions that occurred with a frequency of 1% or less included the following:
Cardiovascular:Chest pain.
Gastrointestinal:Dyspepsia, constipation, anorexia, enteritis, flatulence, gastritis, jaundice, loose stools, and oral moniliasis.
Hematologic and Lymphatic:Anemia and leukopenia.
Nervous System:Headache (otitis media dosage), hyperkinesia, dizziness, agitation, nervousness, and insomnia.
General:Fever, face edema, fatigue, fungal infection, malaise, and pain.
Allergic:Rash and allergic reaction.
Respiratory:Cough, pharyngitis, pleural effusion, and rhinitis.
Skin and Appendages:Eczema, fungal dermatitis, pruritus, sweating, urticaria, and vesiculobullous rash.
Special Senses:Conjunctivitis.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of azithromycin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions reported with azithromycin during the postmarketing period in adult and/or pediatric patients for which a causal relationship may not be established include:
Allergic:Arthralgia, edema, urticaria, and angioedema.
Cardiovascular:Arrhythmias including ventricular tachycardia and hypotension. There have been reports of QT prolongation and torsades de pointes.
Gastrointestinal:Anorexia, constipation, dyspepsia, flatulence, vomiting/diarrhea, pseudomembranous colitis, pancreatitis, oral candidiasis, pyloric stenosis, and reports of tongue discoloration.
General:Asthenia, paresthesia, fatigue, malaise, and anaphylaxis.
Genitourinary:Interstitial nephritis and acute renal failure and vaginitis.
Hematopoietic:Thrombocytopenia.
Liver/Biliary:Abnormal liver function, hepatitis, cholestatic jaundice, hepatic necrosis, and hepatic failure [see Warnings and Precautions (5.2)].
Nervous System:Convulsions, dizziness/vertigo, headache, somnolence, hyperactivity, nervousness, agitation, and syncope.
Psychiatric:Aggressive reaction and anxiety.
Skin/Appendages:Pruritus serious skin reactions including erythema multiforme, AGEP, Stevens-Johnson Syndrome, and toxic epidermal necrolysis, and DRESS.
Special Senses:Hearing disturbances including hearing loss, deafness and/or tinnitus, and reports of taste/smell perversion and/or loss.
6.3 Laboratory Abnormalities
Adults:
Clinically significant abnormalities (irrespective of drug relationship) occurring during the clinical trials were reported as follows: with an incidence of greater than 1%: decreased hemoglobin, hematocrit, lymphocytes, neutrophils, and blood glucose; elevated serum creatine phosphokinase, potassium, ALT, GGT, AST, BUN, creatinine, blood glucose, platelet count, lymphocytes, neutrophils, and eosinophils; with an incidence of less than 1%: leukopenia, neutropenia, decreased sodium, potassium, platelet count, elevated monocytes, basophils, bicarbonate, serum alkaline phosphatase, bilirubin, LDH, and phosphate. The majority of subjects with elevated serum creatinine also had abnormal values at baseline. When follow-up was provided, changes in laboratory tests appeared to be reversible.
In multiple-dose clinical trials involving more than 5000 patients, four patients discontinued therapy because of treatment-related liver enzyme abnormalities and one because of a renal function abnormality.
Pediatric Patients:
One, Three, and Five Day Regimens
Laboratory data collected from comparative clinical trials employing two 3-day regimens (30 mg/kg or 60 mg/kg in divided doses over 3 days), or two 5-day regimens (30 mg/kg or 60 mg/kg in divided doses over 5 days) were similar for regimens of azithromycin and all comparators combined, with most clinically significant laboratory abnormalities occurring at incidences of 1 to 5%. Laboratory data for patients receiving 30 mg/kg as a single dose were collected in one single center trial. In that trial, an absolute neutrophil count between 500 to 1500 cells/mm3 was observed in 10/64 patients receiving 30 mg/kg as a single dose, 9/62 patients receiving 30 mg/kg given over 3 days, and 8/63 comparator patients. No patient had an absolute neutrophil count <500 cells/mm3.
In multiple-dose clinical trials involving approximately 4700 pediatric patients, no patients discontinued therapy because of treatment-related laboratory abnormalities.
Most common adverse reactions are diarrhea (5 to 14%), nausea (3 to 18%), abdominal pain (3 to 7%), or vomiting (2 to 7%). (6.1) (6)
To report SUSPECTED ADVERSE REACTIONS, contact** FDA at 1-800-FDA-1088 or www.fda.gov/medwatch** (6)
DRUG INTERACTIONS SECTION
7 DRUG INTERACTIONS
7.1 Nelfinavir
Co-administration of nelfinavir at steady-state with a single oral dose of azithromycin resulted in increased azithromycin serum concentrations. Although a dose adjustment of azithromycin is not recommended when administered in combination with nelfinavir, close monitoring for known adverse reactions of azithromycin, such as liver enzyme abnormalities and hearing impairment, is warranted [see Adverse Reactions (6)].
7.2 Warfarin
Spontaneous postmarketing reports suggest that concomitant administration of azithromycin may potentiate the effects of oral anticoagulants such as warfarin, although the prothrombin time was not affected in the dedicated drug interaction study with azithromycin and warfarin. Prothrombin times should be carefully monitored while patients are receiving azithromycin and oral anticoagulants concomitantly.
7. 3 Potential Drug-Drug Interaction with Macrolides
Interactions with digoxin, colchicine or phenytoin have not been reported in clinical trials with azithromycin. No specific drug interaction studies have been performed to evaluate potential drug-drug interaction. However, drug interactions have been observed with other macrolide products. Until further data are developed regarding drug interactions when digoxin, colchicine or phenytoin are used with azithromycin careful monitoring of patients is advised.
· Nelfinavir: Close monitoring for known adverse reactions of azithromycin, such as liver enzyme abnormalities and hearing impairment, is warranted. (7.1). (7)
· Warfarin: Use with azithromycin may increase coagulation times; monitor prothrombin time. (7.2). (7)
DOSAGE & ADMINISTRATION SECTION
2 DOSAGE AND ADMINISTRATION
2.1 Adult Patients
[see Indications and Usage (1.1) and Clinical Pharmacology (12.3)]
|
Infection* |
Recommended Dose/Duration of Therapy |
|
Community-acquired pneumonia Pharyngitis/tonsillitis (second-line therapy) Skin/skin structure (uncomplicated) |
500 mg as a single dose on Day 1, followed by 250 mg once daily on Days 2 through 5 |
|
Acute bacterial exacerbations of chronic obstructive pulmonary disease |
500 mg once daily for 3 days |
|
Acute bacterial sinusitis |
500 mg once daily for 3 days |
|
Genital ulcer disease (chancroid) |
One single 1 gram dose |
|
Non-gonococcal urethritis and cervicitis |
One single 1 gram dose |
|
Gonococcal urethritis and cervicitis |
One single 2 gram dose |
|
*DUE TO THE INDICATED ORGANISMS [see Indications and Usage (1.1)] |
Azithromycin tablets can be taken with or without food.
2.2 Pediatric Patients1
|
Infection* |
Recommended Dose/Duration of Therapy |
|
Acute otitis media |
30 mg/kg as a single dose or 10 mg/kg once daily for 3 days or 10 mg/kg as a single dose on Day 1 followed by 5 mg/kg/day on Days 2 through 5. |
|
Acute bacterial sinusitis |
10 mg/kg once daily for 3 days. |
|
Community-acquired pneumonia |
10 mg/kg as a single dose on Day 1 followed by 5 mg/kg once daily on Days 2 through 5. |
|
Pharyngitis/tonsillitis |
12 mg/kg once daily for 5 days. |
|
*DUE TO THE INDICATED ORGANISMS [see Indications and Usage (1.2)] |
PEDIATRIC DOSAGE GUIDELINES FOR OTITIS MEDIA, ACUTE BACTERIAL SINUSITIS, AND COMMUNITY-ACQUIRED PNEUMONIA
(Age 6 months and above,[see Use in Specific Populations (8.4)])
Based on Body Weight
|
OTITIS****MEDIA ANDCOMMUNITY-ACQUIREDPNEUMONIA: | ||||||
|
DosingCalculatedon****10 mg/kg/dayDay 1 and 5 mg/kg/dayDays 2 to 5. | ||||||
|
Weight |
100 mg/5mL |
200 mg/5mL |
TotalmLperTreatment Course |
TotalmgperTreatment Course | ||
|
Kg |
Day****1 |
Days**** |
Day****1 |
Days**** | ||
|
5 |
2.5 mL; (½ tsp) |
1.25mL; (¼tsp) |
7.5 mL |
150 mg | ||
|
10 |
5 mL; (1tsp) |
2.5 mL; (½ tsp) |
15 mL |
300 mg | ||
|
20 |
5 mL; |
2.5 mL; |
15 mL |
600 mg | ||
|
30 |
7.5 mL; |
3.75mL; |
22.5 mL |
900 mg | ||
|
40 |
10 mL; (2 tsp) |
5 mL; |
30 mL |
1200 mg | ||
|
50 andabove |
12.5 mL; |
6.25mL; |
37.5 mL |
1500 mg |
- Effectiveness of the 3-day or 1-day regimen in pediatric patients with community-acquired pneumonia has not been established.
|
OTITISMEDIA ANDACUTEBACTERIALSINUSITIS:(3-DayRegimen)* | ||||
|
DosingCalculatedon****10 mg/kg/day. | ||||
|
Weight |
100 mg/5mL |
200 mg/5mL |
TotalmLperTreatment Course |
Totalmgper Treatment****Course |
|
Kg |
Days** 1 to 3** |
Days** 1 to 3** | ||
|
5 |
2.5 mL; (1/2 tsp) |
7.5 mL |
150 mg | |
|
10 |
5 mL; (1 tsp) |
15 mL |
300 mg | |
|
20 |
5 mL (1 tsp) |
15 mL |
600 mg | |
|
30 |
7.5 mL (1½ tsp) |
22.5 mL |
900 mg | |
|
40 |
10 mL (2 tsp) |
30 mL |
1200 mg | |
|
50 andabove |
12.5 mL (2 ½ tsp) |
37.5 mL |
1500 mg |
- Effectiveness of the 5-day or 1-day regimeninpediatricpatientswithacutebacterialsinusitishasnot been established.
|
OTITISMEDIA:(1-DayRegimen) | |||
|
DosingCalculatedon 30 mg/kgas a singledose. | |||
|
Weight |
200 mg/5mL |
TotalmLperTreatmentCourse |
TotalmgperTreatmentCourse |
|
Kg |
1-DayRegimen | ||
|
5 |
3.75mL;(3/4 tsp) |
3.75mL |
150 mg |
|
10 |
7.5 mL;(1½ tsp) |
7.5 mL |
300 mg |
|
20 |
15 mL;(3 tsp) |
15 mL |
600 mg |
|
30 |
22.5 mL;(4½ tsp) |
22.5 mL |
900 mg |
|
40 |
30 mL;(6 tsp) |
30 mL |
1200 mg |
|
50 andabove |
37.5 mL;(7½ tsp) |
37.5 mL |
1500 mg |
The safety of re-dosing azithromycin in pediatric patients who vomit after receiving 30 mg/kg as a single dose has not been established. In clinical studies involving 487 patients with acute otitis media given a single 30 mg/kg dose of azithromycin, 8 patients who vomited within 30 minutes of dosing were re-dosed at the same total dose.
Pharyngitis/Tonsillitis
The recommended dose of azithromycin for children with pharyngitis/tonsillitis is 12 mg/kg once daily for 5 days. (See chart below.)
PEDIATRIC DOSAGE GUIDELINES FOR PHARYNGITIS/TONSILLITIS
(Age 2 years and above,[see Use in Specific Populations (8.4)])
Based on Body Weight
|
PHARYNGITIS/TONSILLITIS:(5-DayRegimen) | |||
|
DosingCalculatedon 12 mg/kg/dayfor 5 days. | |||
|
Weight |
200 mg/5mL |
TotalmLperTreatmentCourse |
Totalmgper TreatmentCourse |
|
Kg |
Day 1 to 5 | ||
|
8 |
2.5 mL; (½ tsp) |
12.5 mL |
500 mg |
|
17 |
5 mL; (1 tsp) |
25 mL |
1000 mg |
|
25 |
7.5 mL; (1½ tsp) |
37.5 mL |
1500 mg |
|
33 |
10 mL; (2 tsp) |
50 mL |
2000 mg |
|
40 |
12.5 mL; (2½ tsp) |
62.5 mL |
2500 mg |
DESCRIPTION SECTION
11 DESCRIPTION
Azithromycin tablets, USP contain the active ingredient azithromycin, USP a macrolide antibacterial drug, for oral administration. Azithromycin has the chemical name (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-13-[(2,6-dideoxy-3-C-methyl-3-O-methyl-α- L-ribo-hexopyranosyl) oxy]-2-ethyl-3,4,10-trihydroxy-3,5,6,8,10,12,14-heptamethyl-11-[[3,4,6-trideoxy-3-(dimethylamino)-β-D- xylo-hexopyranosyl]oxy]-1-oxa-6-azacyclopentadecan-15-one. Azithromycin is derived from erythromycin; however, it differs chemically from erythromycin in that a methyl-substituted nitrogen atom is incorporated into the lactone ring. Its molecular formula is C38H72N2O12, and its molecular weight is 749. Azithromycin has the following structural formula:
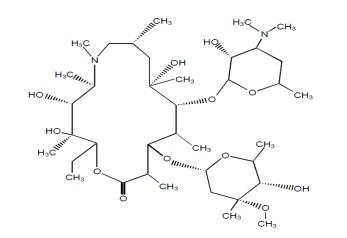
Azithromycin as the dihydrate, is a white or almost white powder with a molecular formula of C38H72N2O12•2H2O and a molecular weight of 785.02.
Azithromycin tablets, USP are supplied for oral administration as pink modified capsule shaped, film-coated tablets containing azithromycin dihydrate equivalent to 500 mg azithromycin, USP and the following inactive ingredients: anhydrous dibasic calcium phosphate, croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, sodium lauryl sulfate, lactose monohydrate, hypromellose, titanium dioxide, triacetin and D&C Red #30 Aluminum Lake.
FDA approved dissolution specification differs from the USP dissolution specification.
NONCLINICAL TOXICOLOGY SECTION
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate carcinogenic potential. Azithromycin has shown no mutagenic potential in standard laboratory tests: mouse lymphoma assay, human lymphocyte clastogenic assay, and mouse bone marrow clastogenic assay. In fertility studies conducted in male and female rats, oral administration of azithromycin for 64 to 66 days (males) or 15 days (females) prior to and during cohabitation resulted in decreased pregnancy rate at 20 and 30 mg/kg/day when both males and females were treated with azithromycin. This minimal effect on pregnancy rate (approximately 12% reduction compared to concurrent controls) did not become more pronounced when the dose was increased from 20 to 30 mg/kg/day (approximately 0.4 to 0.6 times the adult daily dose of 500 mg based on body surface area) and it was not observed when only one animal in the mated pair was treated. There were no effects on any other reproductive parameters, and there were no effects on fertility at 10 mg/kg/day. The relevance of these findings to patients being treated with azithromycin at the doses and durations recommended in the prescribing information is uncertain.
13.2 Animal Toxicology and/or Pharmacology
Phospholipidosis (intracellular phospholipid accumulation) has been observed in some tissues of mice, rats, and dogs given multiple doses of azithromycin. It has been demonstrated in numerous organ systems (e.g., eye, dorsal root ganglia, liver, gallbladder, kidney, spleen, and/or pancreas) in dogs and rats treated with azithromycin at doses which, expressed on the basis of body surface area, are similar to or less than the highest recommended adult human dose. This effect has been shown to be reversible after cessation of azithromycin treatment. Based on the pharmacokinetic data, phospholipidosis has been seen in the rat (50 mg/kg/day dose) at the observed maximal plasma concentration of 1.3 mcg/mL (1.6 times the observed Cmax of 0.821 mcg/mL at the adult dose of 2 g). Similarly, it has been shown in the dog (10 mg/kg/day dose) at the observed maximal serum concentration of 1 mcg/mL (1.2 times the observed Cmax of 0.821 mcg/mL at the adult dose of 2 g). Phospholipidosis was also observed in neonatal rats dosed for 18 days at 30 mg/kg/day, which is less than the pediatric dose of 60 mg/kg based on the surface area. It was not observed in neonatal rats treated for 10 days at 40 mg/kg/day with mean maximal serum concentrations of 1.86 mcg/mL, approximately 1.5 times the Cmax of 1.27 mcg/mL at the pediatric dose. Phospholipidosis has been observed in neonatal dogs (10 mg/kg/day) at maximum mean whole blood concentrations of 3.54 mcg/mL, approximately 3 times the pediatric dose Cmax. The significance of these findings for animals and for humans is unknown.
CLINICAL STUDIES SECTION
14 CLINICAL STUDIES
14.1 Adult Patients
Acute Bacterial Exacerbations of Chronic Bronchitis
In a randomized, double-blind controlled clinical trial of acute exacerbation of chronic bronchitis (AECB), azithromycin (500 mg once daily for 3 days) was compared with clarithromycin (500 mg twice daily for 10 days). The primary endpoint of this trial was the clinical cure rate at Days 21 to 24. For the 304 patients analyzed in the modified intent-to-treat analysis at the Days 21 to 24 visit, the clinical cure rate for 3 days of azithromycin was 85% (125/147) compared to 82% (129/157) for 10 days of clarithromycin.
The following outcomes were the clinical cure rates at the Days 21 to 24 visit for the bacteriologically evaluable patients by pathogen:
|
Pathogen |
Azithromycin (3 Days)**** |
Clarithromycin (10 Days)**** |
|
S. pneumoniae**** |
29/32 (91%)**** |
21/27 (78%)**** |
|
H. influenzae**** |
12/14 (86%)**** |
14/16 (88%)**** |
|
M. catarrhalis**** |
11/12 (92%)**** |
12/15 (80%)**** |
Acute Bacterial Sinusitis
In a randomized, double-blind, double-dummy controlled clinical trial of acute bacterial sinusitis, azithromycin (500 mg once daily for 3 days) was compared with amoxicillin/clavulanate (500/125 mg three times a day for 10 days). Clinical response assessments were made at Day 10 and Day 28. The primary endpoint of this trial was prospectively defined as the clinical cure rate at Day 28. For the 594 patients analyzed in the modified intent to treat analysis at the Day 10 visit, the clinical cure rate for 3 days of azithromycin was 88% (268/303) compared to 85% (248/291) for 10 days of amoxicillin/clavulanate. For the 586 patients analyzed in the modified intent to treat analysis at the Day 28 visit, the clinical cure rate for 3 days of azithromycin was 71.5% (213/298) compared to 71.5% (206/288), with a 97.5% confidence interval of –8.4 to 8.3, for 10 days of amoxicillin/clavulanate.
In an open label, non-comparative study requiring baseline transantral sinus punctures, the following outcomes were the clinical success rates at the Day 7 and Day 28 visits for the modified intent to treat patients administered 500 mg of azithromycin once daily for 3 days with the following pathogens:
Clinical Success Rates of Azithromycin (500 mg per day for 3 Days)
|
Pathogen**** |
Day 7**** |
Day 28**** |
|
S. pneumoniae**** |
23/26 (88%)**** |
21/25 (84%)**** |
|
H. influenzae**** |
28/32 (87%)**** |
24/32 (75%)**** |
|
M. catarrhalis**** |
14/15 (93%)**** |
13/15 (87%)**** |
14.2 Pediatric Patients
From the perspective of evaluating pediatric clinical trials, Days 11 to 14 were considered on-therapy evaluations because of the extended half-life of azithromycin. Days 11 to 14 data are provided for clinical guidance. Days 24 to 32 evaluations were considered the primary test of cure endpoint.
Pharyngitis/Tonsillitis
In three double-blind controlled studies, conducted in the United States, azithromycin (12 mg/kg once a day for 5 days) was compared to penicillin V (250 mg three times a day for 10 days) in the treatment of pharyngitis due to documented Group A β-hemolytic streptococci (GABHS or S. pyogenes). Azithromycin was clinically and microbiologically statistically superior to penicillin at Day 14 and Day 30 with the following clinical success (i.e., cure and improvement) and bacteriologic efficacy rates (for the combined evaluable patient with documented GABHS):
Three U.S. Streptococcal Pharyngitis Studies
Azithromycin vs. Penicillin V
EFFICACY RESULTS
|
Day 14 |
Day 30 | |
|
BacteriologicEradication: | ||
|
Azithromycin |
323/340 (95%) |
255/330 (77%) |
|
Penicillin V |
242/332 (73%) |
206/325 (63%) |
|
ClinicalSuccess(cure plus improvement): | ||
|
Azithromycin |
336/343 (98%) |
310/330 (94%) |
|
Penicillin V |
284/338 (84%) |
241/325 (74%) |
Approximately 1% of azithromycin-susceptible S. pyogenes isolates were resistant to azithromycin following therapy.
Acute Otitis Media
Efficacy using azithromycin given over 5 days(10 mg/kg on Day 1 followed by 5 mg/kg on Days 2 to 5):
Trial 1
In a double-blind, controlled clinical study of acute otitis media performed in the United States, azithromycin (10 mg/kg on Day 1 followed by 5 mg/kg on Days 2 to 5) was compared to amoxicillin/clavulanate potassium (4:1). For the 553 patients who were evaluated for clinical efficacy, the clinical success rate (i.e., cure plus improvement) at the Day 11 visit was 88% for azithromycin and 88% for the control agent. For the 521 patients who were evaluated at the Day 30 visit, the clinical success rate was 73% for azithromycin and 71% for the control agent.
Trial 2
In a non-comparative clinical and microbiologic trial performed in the United States, where significant rates of beta-lactamase producing organisms (35%) were found, 131 patients were evaluable for clinical efficacy. The combined clinical success rate (i.e., cure and improvement) at the Day 11 visit was 84% for azithromycin. For the 122 patients who were evaluated at the Day 30 visit, the clinical success rate was 70% for azithromycin.
Microbiologic determinations were made at the pre-treatment visit. Microbiology was not reassessed at later visits. The following clinical success rates were obtained from the evaluable group:
|
Day 11 |
Day 30 | |
|
Pathogen |
Azithromycin |
Azithromycin |
|
S. pneumoniae |
61/74 (82%) |
40/56 (71%) |
|
H. influenzae |
43/54 (80%) |
30/47 (64%) |
|
M.catarrhalis |
28/35 (80%) |
19/26 (73%) |
|
S. pyogenes |
11/11 (100%) |
7/7 (100%) |
|
Overall |
177/217 (82%) |
97/137 (73%) |
Trial 3
In another controlled comparative clinical and microbiologic study of otitis media performed in the United States, azithromycin (10 mg/kg on Day 1 followed by 5 mg/kg on Days 2 to 5) was compared to amoxicillin/clavulanate potassium (4:1). This study utilized two of the same investigators as Protocol 2 (above), and these two investigators enrolled 90% of the patients in Protocol 3. For this reason, Protocol 3 was not considered to be an independent study. Significant rates of beta-lactamase producing organisms (20%) were found. Ninety-two (92) patients were evaluable for clinical and microbiologic efficacy. The combined clinical success rate (i.e., cure and improvement) of those patients with a baseline pathogen at the Day 11 visit was 88% for azithromycin vs. 100% for control; at the Day 30 visit, the clinical success rate was 82% for azithromycin vs. 80% for control.
Microbiologic determinations were made at the pre-treatment visit. Microbiology was not reassessed at later visits. At the Day 11 and Day 30 visits, the following clinical success rates were obtained from the evaluable group:
|
Day 11 |
Day 30 | |||
|---|---|---|---|---|
|
Pathogen |
Azithromycin |
Control |
Azithromycin |
Control |
|
S. pneumoniae |
25/29 (86%) |
26/26 (100%) |
22/28 (79%) |
18/22 (82%) |
|
H. influenzae |
9/11 (82%) |
9/9 (100%) |
8/10 (80%) |
6/8 (75%) |
|
M.catarrhalis |
7/7 (100%) |
5/5 (100%) |
5/5 (100%) |
2/3 (66%) |
|
S. pyogenes |
2/2 (100%) |
5/5 (100%) |
2/2 (100%) |
4/4 (100%) |
|
Overall |
43/49 (88%) |
45/45 (100%) |
37/45 (82%) |
30/37 (81%) |
Efficacy using azithromycin given over 3 days (10 mg/kg/day):
Trial 4
In a double-blind, controlled, randomized clinical study of acute otitis media in pediatric patients from 6 months to 12 years of age, azithromycin (10 mg/kg per day for 3 days) was compared to amoxicillin/clavulanate potassium (7:1) in divided doses q12h for 10 days. Each patient received active drug and placebo matched for the comparator.
For the 366 patients who were evaluated for clinical efficacy at the Day 12 visit, the clinical success rate (i.e., cure plus improvement) was 83% for azithromycin and 88% for the control agent. For the 362 patients who were evaluated at the Days 24 to 28 visit, the clinical success rate was 74% for azithromycin and 69% for the control agent.
Efficacy using azithromycin 30 mg/kg given as a single dose:
Trial 5
A double-blind, controlled, randomized trial was performed at nine clinical centers. Pediatric patients from 6 months to 12 years of age were randomized 1:1 to treatment with either azithromycin (given at 30 mg/kg as a single dose on Day 1) or amoxicillin/clavulanate potassium (7:1), divided q12h for 10 days. Each child received active drug, and placebo matched for the comparator.
Clinical response (Cure, Improvement, Failure) was evaluated at End of Therapy (Days 12 to 16) and Test of Cure (Days 28 to 32). Safety was evaluated throughout the trial for all treated subjects. For the 321 subjects who were evaluated at End of Treatment, the clinical success rate (cure plus improvement) was 87% for azithromycin, and 88% for the comparator. For the 305 subjects who were evaluated at Test of Cure, the clinical success rate was 75% for both azithromycin and the comparator.
Trial 6
In a non-comparative clinical and microbiological trial, 248 patients from 6 months to 12 years of age with documented acute otitis media were dosed with a single oral dose of azithromycin (30 mg/kg on Day 1).
For the 240 patients who were evaluable for clinical modified Intent-to-Treat (MITT) analysis, the clinical success rate (i.e., cure plus improvement) at Day 10 was 89% and for the 242 patients evaluable at Days 24 to 28, the clinical success rate (cure) was 85%.
|
Presumed****Bacteriologic Eradication | ||
|
Day 10 |
Days 24 to 28 | |
|
S. pneumoniae |
70/76 (92%) |
67/76 (88%) |
|
H. influenzae |
30/42 (71%) |
28/44 (64%) |
|
M.catarrhalis |
10/10 (100%) |
10/10 (100%) |
|
Overall |
110/128 (86%) |
105/130 (81%) |
SPL PATIENT PACKAGE INSERT SECTION
Patient Information
Azithromycin (ay-ZITH-roe-MYE-sin) Tablets, USP
Read this Patient Information leaflet before you start taking azithromycin tablets and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
What are azithromycin tablets?
Azithromycin tablets are a macrolide antibiotic prescription medicine used in
adults 18 years or older to treat certain infections caused by certain germs
called bacteria. These bacterial infections include:
• acute worsening of chronic bronchitis
• acute sinus infection
• community-acquired pneumonia
• infected throat or tonsils
• skin infections
• infections of the urethra or cervix
• genital ulcers in men
Azithromycin tablets are also used in children to treat:
• ear infections
• community-acquired pneumonia
• infected throat or tonsils
Azithromycin should not be taken by people who cannot tolerate oral
medications because they are very ill or have certain other risk factors
including:
• have cystic fibrosis
• have hospital acquired infections
• have known or suspected bacteria in the blood
• need to be in the hospital
• are elderly
• have any medical problems that can lower the ability of the immune system to
fight infections
Azithromycin tablets are not for viral infections such as the common cold.
It is not known if azithromycin tablets are safe and effective for genital ulcers in women.
It is not known if azithromycin tablets are safe and effective for children with ear infections, sinus infections, and community-acquired pneumonia under 6 months of age.
It is not known if azithromycin tablets are safe and effective for infected throat or tonsils in children under 2 years of age.
Who should not take azithromycin tablets?
** Do not take azithromycin tablets if you:**
• have had a severe allergic reaction to certain antibiotics known as
macrolides or ketolides including azithromycin and erythromycin.
• have a history of cholestatic jaundice or hepatic dysfunction that happened
with the use of azithromycin.
What should I tell my healthcare provider before taking azithromycin
tablets?
** Before you take azithromycin tablets, tell your healthcare provider if
you:**
• have pneumonia
• have cystic fibrosis
• have known or suspected bacteremia (bacterial infection in the blood)
• have liver or kidney problems
• have an irregular heartbeat, especially a problem called “QT prolongation”
• have a problem that causes muscle weakness (myasthenia gravis)
• have any other medical problems
• are pregnant or plan to become pregnant. It is not known if azithromycin
will harm your unborn baby.
• are breastfeeding or plan to breastfeed. Azithromycin has been reported to
pass into breast milk. Talk to your healthcare provider about the best way to
feed your baby while you take azithromycin tablets.
Contact your healthcare provider immediately if you are giving azithromycin tablets to a young child (less than 6 weeks of age) and he or she vomits or becomes irritable when fed.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
Azithromycin tablets and other medicines may affect each other causing side effects. Azithromycin tablets may affect the way other medicines work, and other medicines may affect how azithromycin tablets works.
Especially tell your healthcare provider if you take:
• nelfinavir
• a blood thinner (warfarin)
• digoxin
• colchicine
• phenytoin
• an antacid that contains aluminum or magnesium
Know the medicines you take. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine.
How should I take azithromycin tablets?
• Take azithromycin tablets exactly as your healthcare provider tells you to
take it.
• Azithromycin tablets can be taken with or without food.
• Do not skip any doses of azithromycin tablets or stop taking it, even if you
begin to feel better, until you finish your prescribed treatment unless you
have a serious allergic reaction or your healthcare provider tells you to stop
taking azithromycin tablets.“See What are the possible side effects of
azithromycin tablets?” If you skip doses, or do not complete the total
course of azithromycin tablets your treatment may not work as well and your
infection may be harder to treat. Taking all of your azithromycin tablets
doses will help lower the chance that the bacteria will become resistant to
azithromycin tablets.
• If the bacteria becomes resistant to azithromycin, azithromycin tablets and
other antibiotic medicines may not work for you in the future.
• If you take too much azithromycin tablets, call your healthcare provider or
get medical help right away.
What are the possible side effects of azithromycin tablets?
Azithromycin tablets can cause serious side effects, including:
•Serious allergic reactions. Allergic reactions can happen in people
taking azithromcyin the active ingredient in azithromycin tablets, even after
only 1 dose. Stop taking azithromycin tablets and get emergency medical help
right away if you have any of the following symptoms of a severe allergic
reaction:
o trouble breathing or swallowing
o swelling of the lips, tongue, face
o throat tightness, hoarseness
o rapid heartbeat
o faintness
o skin rash (hives)
o new onset of fever and swollen lymph nodes
Stop taking azithromycin tablets at the first sign of a skin rash and call your healthcare provider. Skin rash may be a sign of a more serious reaction to azithromycin tablets.
•Liver damage (hepatotoxicity). Hepatotoxicity can happen in people who take azithromycin tablets. Call your healthcare provider right away if you have unexplained symptoms such as:
|
o nausea or vomiting |
o loss of appetite |
|
o stomach pain |
o change in the color of your bowel movements |
|
o fever | |
|
o weakness |
o dark colored urine |
|
o abdominal pain or tenderness |
o yellowing of your skin or of the whites of your eyes |
|
o itching | |
|
o unusual tiredness |
Stop taking azithromycin tablets and tell your healthcare provider right away if you have yellowing of your skin or white part of your eyes, or if you have dark urine. These can be signs of a serious reaction to azithromycin tablets (a liver problem).
•Serious heart rhythm changes (QT prolongation and torsades de pointes).
Tell your healthcare provider right away if you have a change in your
heartbeat (a fast or irregular heartbeat), or if you feel faint and dizzy.
Azithromycin tablets may cause a rare heart problem known as prolongation of
the QT interval. This condition can cause an abnormal heartbeat and can be
very dangerous. The chances of this happening are higher in people:
o who are elderly
o with a family history of prolonged QT interval
o with low blood potassium
o who take certain medicines to control heart rhythm (antiarrhythmics)
•Worsening of myasthenia gravis (a problem that causes muscle weakness).
Certain antibiotics like azithromycin tablets may cause worsening of
myasthenia gravis symptoms, including muscle weakness and breathing problems.
Call your healthcare provider right away if you have any worsening muscle
weakness or breathing problems.
•Diarrhea. Tell your healthcare provider right away if you have watery diarrhea, diarrhea that does not go away, or bloody stools. You may experience cramping and a fever. This could happen after you have finished your azithromycin tablets.
The most common side effects of azithromycin tablets include:
• nausea
• stomach pain
• vomiting
These are not all the possible side effects of azithromycin tablets. Tell your healthcare provider about any side effect that bothers you or that does not go away.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store azithromycin tablets?
• Store azithromycin tablets at 25°C (77°F); excursions permitted to 15° to
30°C (59° to 86°F) [see USP Controlled Room Temperature].
• Safely throw away any medicine that is out of date or no longer needed.
Keep azithromycin tablets and all medicines out of the reach of children.
General information about the safe and effective use of azithromycin
tablets.
Medicines are sometimes prescribed for purposes other than those listed in the
Patient Information leaflet. Do not use azithromycin tablets for a condition
for which it was not prescribed. Do not give azithromycin tablets to other
people, even if they have the same symptoms you have. It may harm them.
This Patient Information leaflet summarizes the most important information about azithromycin tablets. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about azithromycin tablets that is written for health professionals.
What are the ingredients in azithromycin tablets?
Active ingredient: azithromycin dihydrate
Inactive ingredients: anhydrous dibasic calcium phosphate, croscarmellose sodium, hydroxypropyl cellulose, magnesium stearate, sodium lauryl sulfate, lactose monohydrate, hypromellose, titanium dioxide, triacetin and D&C Red #30 Aluminum Lake.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Manufactured by:
Alembic Pharmaceuticals Limited
(Formulation Division),
Village Panelav, P. O. Tajpura,
Near Baska, Taluka-Halol,
Panchmahal 389350, Gujarat, India.
Revised: 07/2019
