Metronidazole
METRONIDAZOLE TOPICAL CREAM 0.75%
43ed872b-63ac-4a22-9c9e-cc0e05bcc41e
HUMAN PRESCRIPTION DRUG LABEL
Jun 27, 2022
Fougera Pharmaceuticals Inc.
DUNS: 043838424
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Metronidazole
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (8)
Drug Labeling Information
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – 45 GRAM CARTON
NDC 0168-0323-46
Rx only
Fougera®
METRONIDAZOLE
TOPICAL CREAM 0.75%
FOR TOPICAL USE ONLY
NOT FOR OPHTHALMIC USE
NET WT 45 grams

INDICATIONS & USAGE SECTION
INDICATIONS AND USAGE
Metronidazole Topical Cream is indicated for topical application in the treatment of inflammatory papules and pustules of rosacea.
CONTRAINDICATIONS SECTION
CONTRAINDICATIONS
Metronidazole topical cream is contraindicated in individuals with a history of hypersensitivity to metronidazole, or other ingredients of the formulation.
ADVERSE REACTIONS SECTION
ADVERSE REACTIONS
In controlled clinical trials, the total incidence of adverse reactions associated with the use of metronidazole topical cream was approximately 10%. Skin discomfort (burning and stinging) was the most frequently reported event followed by erythema, skin irritation, pruritus and worsening of rosacea. All individual events occurred in less than 3% of patients.
The following additional adverse experiences have been reported with the topical use of metronidazole: dryness, transient redness, metallic taste, tingling or numbness of extremities and nausea.
SPL UNCLASSIFIED SECTION
FOR TOPICAL USE ONLY
NOT FOR OPHTHALMIC USE
Rx only
DESCRIPTION SECTION
DESCRIPTION
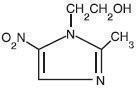
Metronidazole Topical Cream contains metronidazole, USP, at a concentration of 7.5 mg per gram (0.75%) in an emollient cream consisting of emulsifying wax, sorbitol solution, glycerin, isopropyl palmitate, benzyl alcohol, lactic acid, and/or sodium hydroxide to adjust pH, and purified water. Metronidazole is a member of the imidazole class of anti-bacterial agents and is classified therapeutically as an antiprotozoal and antibacterial agent. Chemically, metronidazole is 2-Methyl-5-nitroimidazole-1-ethanol. The molecular formula is C6H9N3O3 and molecular weight is 171.16. Metronidazole is represented by the following structural formula:
CLINICAL PHARMACOLOGY SECTION
CLINICAL PHARMACOLOGY
The mechanisms by which metronidazole acts in the treatment of rosacea are unknown, but appear to include an anti-inflammatory effect.
PRECAUTIONS SECTION
PRECAUTIONS
General: Topical metronidazole has been reported to cause tearing of the eyes. Therefore, contact with the eyes should be avoided. If a reaction suggesting local irritation occurs, patients should be directed to use the medication less frequently or discontinue use. Metronidazole is a nitroimidazole and should be used with care in patients with evidence of, or history of blood dyscrasia.
Information for patients: This medication is to be used as directed by the physician. It is for external use only. Avoid contact with the eyes.
Drug interactions: Oral metronidazole has been reported to potentiate the anticoagulant effect of warfarin and coumarin anticoagulants, resulting in a prolongation of prothrombin time. The effect of topical metronidazole on prothrombin time is not known.
Carcinogenesis, mutagenesis, impairment of fertility: Metronidazole has shown evidence of carcinogenic activity in a number of studies involving chronic, oral administration in mice and rats but not in studies involving hamsters.
Metronidazole has shown evidence of mutagenic activity in several in vitro bacterial assay systems. In addition, a dose-response increase in the frequency of micronuclei was observed in mice after intraperitoneal injections and an increase in chromosome aberrations have been reported in patients with Crohn's disease who were treated with 200-1200 mg/day of metronidazole for 1 to 24 months. However, no excess chromosomal aberrations in circulating human lymphocytes have been observed in patients treated for 8 months.
**Pregnancy:**Teratogenic effects: Pregnancy category B
There are no adequate and well-controlled studies with the use of metronidazole topical cream in pregnant women. Metronidazole crosses the placental barrier and enters the fetal circulation rapidly. No fetotoxicity was observed after oral metronidazole in rats or mice. However, because animal reproduction studies are not always predictive of human response and since oral metronidazole has been shown to be a carcinogen in some rodents, this drug should be used during pregnancy only if clearly needed.
Nursing mothers: After oral administration, metronidazole is secreted in breast milk in concentrations similar to those found in the plasma. Even though blood levels are significantly lower with topically applied metronidazole than those achieved after oral administration of metronidazole, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric use: Safety and effectiveness in pediatric patients have not been established.
DOSAGE & ADMINISTRATION SECTION
DOSAGE AND ADMINISTRATION
Apply and rub in a thin layer of metronidazole topical cream twice daily, morning and evening, to entire affected areas after washing.
Areas to be treated should be washed with a mild cleanser before application. Patients may use cosmetics after application of metronidazole topical cream.
HOW SUPPLIED SECTION
HOW SUPPLIED
Metronidazole Topical Cream 0.75% is supplied in a 45 gram tube,
NDC 0168-0323-46
Storage conditions: Store at 20°C to 25°C (68°F to 77°F), excursions permitted 15°C to 30°C
(59°F to 86°F). [See USP Controlled Room Temperature].
E. FOUGERA & CO.
A division of Fougera Pharmaceuticals Inc.
MELVILLE, NEW YORK 11747
46273863A
R07/2020
#29
