Walgreen
Walgreens Handsoap-0363-0242
c992cf19-786c-d794-e053-2995a90af067
HUMAN OTC DRUG LABEL
Aug 20, 2025
Walgreen Co
DUNS: 008965063
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
BENZALKONIUM CHLORIDE
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (17)
Drug Labeling Information
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Principal Display Panel
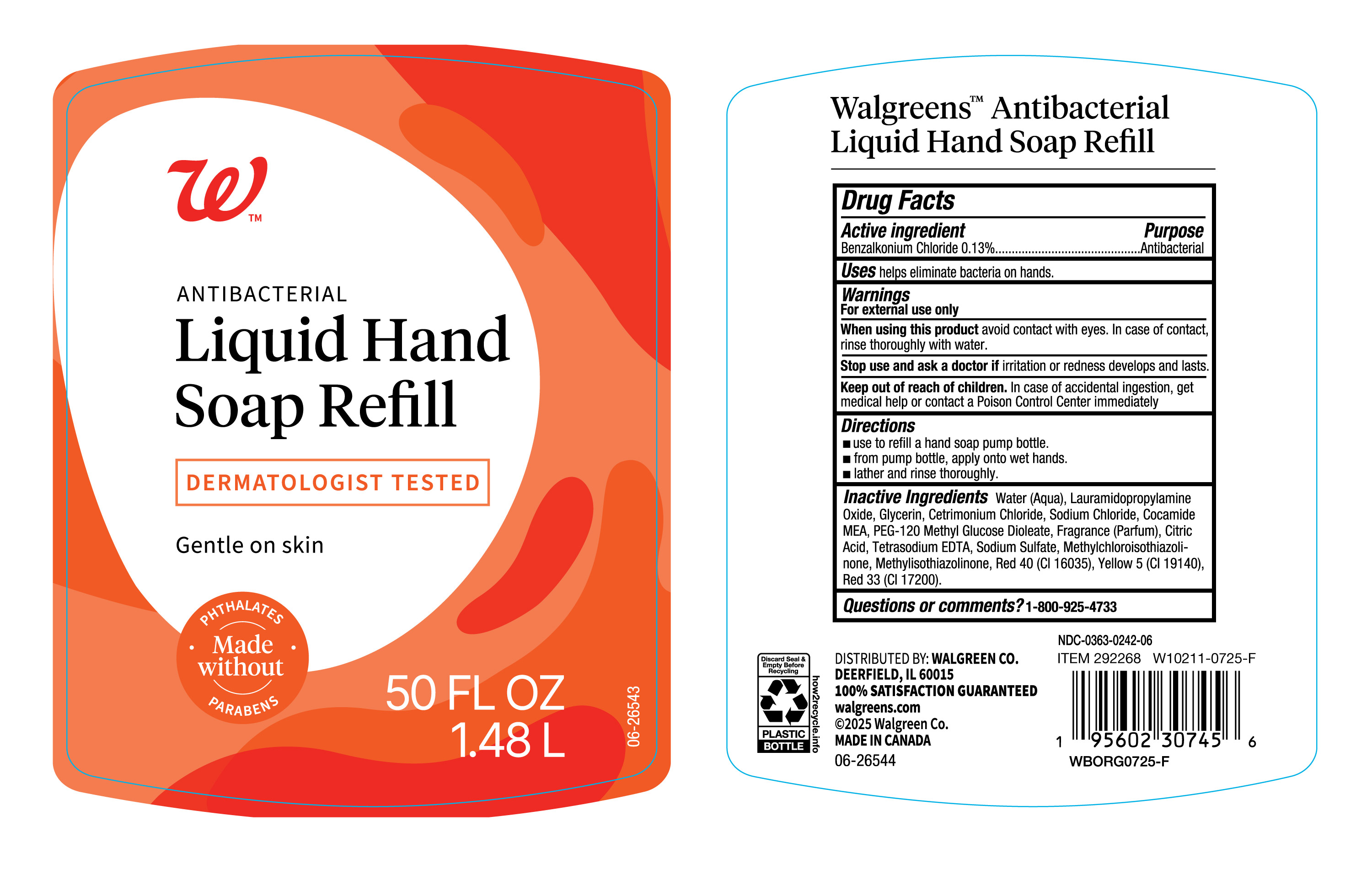
INDICATIONS & USAGE SECTION
Uses
helps eliminate bacteria on hands.
DOSAGE & ADMINISTRATION SECTION
Directions
- use to refill soap pump bottle
- from pump bottle, apply onto wet hands
- Lather and rinse thoroughly.
STORAGE AND HANDLING SECTION
Other Information
Store at room temperature
OTC - ACTIVE INGREDIENT SECTION
Active Ingredient.
Benzalkonium Chloride (0.13%)
OTC - PURPOSE SECTION
Purpose
Antibacterial
WARNINGS SECTION
Warnings
For external use only
When using this product
Avoid contact with eyes. In case of contact, rinse thoroughly with water.
Stop use and ask a doctor if
irritation or redness develops and lasts
Keep out of reach of children.
In case of accidental ingestion, get medical help or contact Poison Control Center immediately.
INACTIVE INGREDIENT SECTION
Inactive Ingredients
Water (Aqua), Lauramidopropylamine Oxide, Glycerin, Cetrimonium Chloride, Sodium Chloride, Cocamide MEA, PEG-120 Methyl Glucose Dioleate, Fragrance (Parfum), Citric Acid, Tetrasodium EDTA, Sodium Sulfate, Methylchloroisothiazolinone, Methylisothiazolinone, Red 40 (Cl 16035), Yellow 5 (Cl 19140), Red 33 (Cl 17200).
OTC - QUESTIONS SECTION
Questions or comments?
1-800-925-4733
