Gabapentin
These highlights do not include all the information needed to use GABAPENTIN safely and effectively. See full prescribing information for GABAPENTIN. GABAPENTIN capsules, for oral use GABAPENTIN tablets, for oral use Initial U.S. Approval: 1993
d35cd5ac-5ed6-2405-e053-2995a90a6946
HUMAN PRESCRIPTION DRUG LABEL
Oct 5, 2023
NuCare Pharmaceuticals,Inc.
DUNS: 010632300
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Gabapentin
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (6)
Drug Labeling Information
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Package/Label Display Panel

HOW SUPPLIED SECTION
16 HOW SUPPLIED/STORAGE AND HANDLING
Gabapentin capsules and tablets, USP are supplied as follows:
100 mg capsules:
White to off-white powder filled in size “3” hard gelatin capsules with opaque white colored cap and opaque white colored body imprintedSG on cap and 179 on body with black ink, available in:
BOTTLES OF 30 NDC 68071-2600-3
BOTTLES OF 60 NDC 68071-2600-6
Store gabapentin capsules and tablets at 25°C (77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
DOSAGE & ADMINISTRATION SECTION
2 DOSAGE AND ADMINISTRATION
2.1 Dosage for Postherpetic Neuralgia
In adults with postherpetic neuralgia, gabapentin may be initiated on Day 1 as a single 300 mg dose, on Day 2 as 600 mg/day (300 mg two times a day), and on Day 3 as 900 mg/day (300 mg three times a day). The dose can subsequently be titrated up as needed for pain relief to a dose of 1800 mg/day (600 mg three times a day). In clinical studies, efficacy was demonstrated over a range of doses from 1800 mg/day to 3600 mg/day with comparable effects across the dose range; however, in these clinical studies, the additional benefit of using doses greater than 1800 mg/day was not demonstrated.
2.2 Dosage for Epilepsy with Partial Onset Seizures
Patients 12 years of age and above
The starting dose is 300 mg three times a day. The recommended maintenance dose of gabapentin is 300 mg to 600 mg three times a day. Dosages up to 2,400 mg/day have been well tolerated in long-term clinical studies. Doses of 3,600 mg/day have also been administered to a small number of patients for a relatively short duration, and have been well tolerated. Administer gabapentin three times a day using 300 mg or 400 mg capsules, or 600 mg or 800 mg tablets. The maximum time between doses should not exceed 12 hours.
Pediatric Patients Age 3 to 11 years
The starting dose range is 10 mg/kg/day to 15 mg/kg/day, given in three divided doses, and the recommended maintenance dose reached by upward titration over a period of approximately 3 days. The recommended maintenance dose of gabapentin in patients 3 to 4 years of age is 40 mg/kg/day, given in three divided doses. The recommended maintenance dose of gabapentin in patients 5 to 11 years of age is 25 mg/kg/day to 35 mg/kg/day, given in three divided doses. Gabapentin may be administered as capsule, or tablet, or using combinations of these formulations. Dosages up to 50 mg/kg/day have been well tolerated in a long-term clinical study. The maximum time interval between doses should not exceed 12 hours.
2.3 Dosage Adjustment in Patients with Renal Impairment
Dosage adjustment in patients 12 years of age and older with renal impairment or undergoing hemodialysis is recommended, as follows (see dosing recommendations above for effective doses in each indication):
TABLE 1. Gabapentin Dosage Based on Renal Function
|
Renal Function Creatinine Clearance (mL/min) |
Total Daily Dose Range (mg/day) |
Dose Regimen (mg) | ||||
|
≥ 60 |
900 to 3,600 |
300 TID |
400 TID |
600 TID |
800 TID |
1,200 TID |
|
400 to 1,400 |
200 BID |
300 BID |
400 BID |
500 BID |
700 BID |
|
200 to 700 |
200 QD |
300 QD |
400 QD |
500 QD |
700 QD |
|
15 a |
100 to 300 |
100 QD |
125 QD |
150 QD |
200 QD |
300 QD |
|
Post-Hemodialysis Supplemental Dose (mg) b | ||||||
|
Hemodialysis |
125 b |
150 b |
200 b |
250 b |
350 b |
TID = Three times a day; BID = Two times a day; QD = Single daily dose
a For patients with creatinine clearance <15 mL/min, reduce daily dose in proportion to creatinine clearance (e.g., patients with a creatinine clearance of 7.5 mL/min should receive one-half the daily dose that patients with a creatinine clearance of 15 mL/min receive).
b Patients on hemodialysis should receive maintenance doses based on estimates of creatinine clearance as indicated in the upper portion of the table and a supplemental post-hemodialysis dose administered after each 4 hours of hemodialysis as indicated in the lower portion of the table.
Creatinine clearance (CLCr) is difficult to measure in outpatients. In patients with stable renal function, creatinine clearance can be reasonably well estimated using the equation of Cockcroft and Gault:
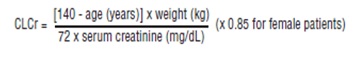
The use of gabapentin in patients less than 12 years of age with compromised renal function has not been studied.
2.4 Dosage in Elderly
Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and dose should be adjusted based on creatinine clearance values in these patients.
2.5 Administration Information
Administer gabapentin orally with or without food. Gabapentin capsules should be swallowed whole with water. Inform patients that, should they divide the scored 600 mg or 800 mg gabapentin tablet in order to administer a half- tablet, they should take the unused half-tablet as the next dose. Half-tablets not used within 28 days of dividing the scored tablet should be discarded. If the gabapentin dose is reduced, discontinued, or substituted with an alternative medication, this should be done gradually over a minimum of 1 week (a longer period may be needed at the discretion of the prescriber).
- Postherpetic Neuralgia ( 2.1)
- Dose can be titrated up as needed to a dose of 1800 mg/day
- Day 1: Single 300 mg dose
- Day 2: 600 mg/day (i.e., 300 mg two times a day)
- Day 3: 900 mg/day (i.e., 300 mg three times a day)
- Epilepsy with Partial Onset Seizures ( 2.2)
- Patients 12 years of age and older: starting dose is 300 mg three times daily; may be titrated up to 600 mg three times daily
- Patients 3 to 11 years of age: starting dose range is 10 to 15 mg/kg/day, given in three divided doses; recommended dose in patients 3 to 4 years of age is 40 mg/kg/day, given in three divided doses; the recommended dose in patients 5 to 11 years of age is 25 to 35 mg/kg/day, given in three divided doses. The recommended dose is reached by upward titration over a period of approximately 3 days
- Dose should be adjusted in patients with reduced renal function ( 2.3, 2.4)
DESCRIPTION SECTION
11 DESCRIPTION
The active ingredient in gabapentin capsules and tablets, USP is gabapentin, which has the chemical name 1-(aminomethyl)cyclohexaneacetic acid.
The molecular formula of gabapentin is C 9H 17NO 2 and the molecular weight is 171.24. The structural formula of gabapentin is:
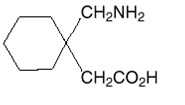
Gabapentin, USP is a white to off-white crystalline solid with a pK a1 of 4.72±0.10 and a pK a2 of 10.27±0.29. It is freely soluble in water and both basic and acidic aqueous solutions. The log of the partition coefficient is -1.083±0.235 at 25°C temperature.
Each gabapentin capsule contains 100 mg, 300 mg or 400 mg of gabapentin, USP and the following inactive ingredients: pregelatinized starch (maize), and talc. The 100 mg capsule shell contains gelatin, sodium lauryl sulfate (SLS) and titanium dioxide. The 300 mg capsule shell contains gelatin, titanium dioxide, FD&C Red 40, D&C Yellow 10, and sodium lauryl sulfate (SLS). The 400 mg capsule shell contains gelatin, titanium dioxide, sodium lauryl sulfate (SLS), D&C Yellow 10, and FD&C Red 40. The imprinting ink contains shellac, dehydrated alcohol, isopropyl alcohol, butyl alcohol, propylene glycol, strong ammonia solution, black iron oxide, and potassium hydroxide.
Each gabapentin tablet contains 600 mg or 800 mg of gabapentin, USP and the following inactive ingredients: poloxamer 407, mannitol, magnesium stearate, hydroxypropyl cellulose, talc, copovidone, crospovidone, colloidal silicon dioxide and coating agent contains hypromellose, titanium dioxide, polyethylene glycol and talc.
CLINICAL STUDIES SECTION
14 CLINICAL STUDIES
14.1 Postherpetic Neuralgia
Gabapentin was evaluated for the management of postherpetic neuralgia (PHN) in two randomized, double-blind, placebo-controlled, multicenter studies. The intent-to-treat (ITT) population consisted of a total of 563 patients with pain for more than 3 months after healing of the herpes zoster skin rash (Table 6).
TABLE 6. Controlled PHN Studies: Duration, Dosages, and Number of Patients|
Study |
Study Duration |
Gabapentin |
Patients Receiving Gabapentin |
Patients Receiving Placebo |
|---|---|---|---|---|
|
1 |
8 weeks |
3600 |
113 |
116 |
|
2 |
7 weeks |
1800, 2400 |
223 |
111 |
|
Total |
336 |
227 |
aGiven in 3 divided doses (TID)
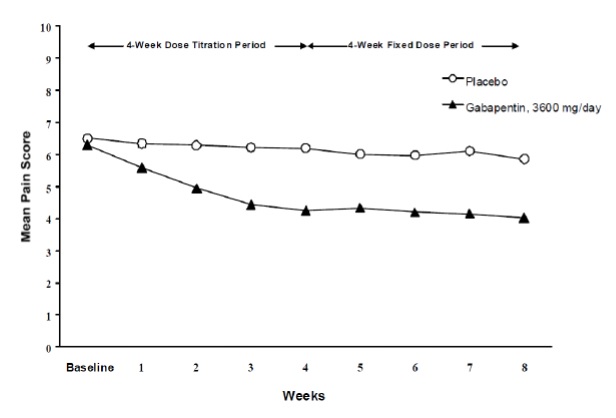
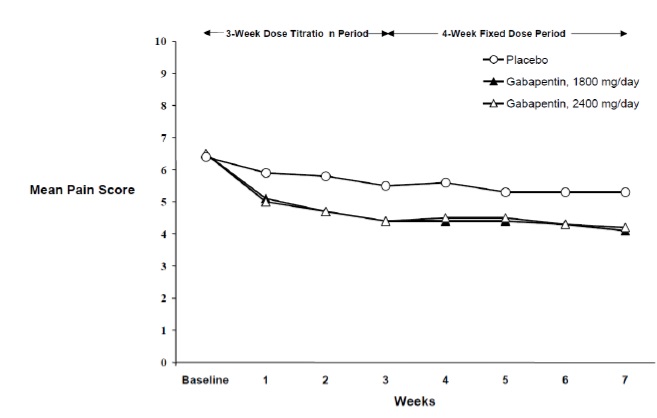
Each study included a 7- or 8-week double-blind phase (3 or 4 weeks of titration and 4 weeks of fixed dose). Patients initiated treatment with titration to a maximum of 900 mg/day gabapentin over 3 days. Dosages were then to be titrated in 600 to 1200 mg/day increments at 3- to 7-day intervals to the target dose over 3 to 4 weeks. Patients recorded their pain in a daily diary using an 11-point numeric pain rating scale ranging from 0 (no pain) to 10 (worst possible pain). A mean pain score during baseline of at least 4 was required for randomization. Analyses were conducted using the ITT population (all randomized patients who received at least one dose of study medication).
Both studies demonstrated efficacy compared to placebo at all doses tested.
The reduction in weekly mean pain scores was seen by Week 1 in both studies, and were maintained to the end of treatment. Comparable treatment effects were observed in all active treatment arms. Pharmacokinetic/pharmacodynamic modeling provided confirmatory evidence of efficacy across all doses. Figures 1 and 2 show pain intensity scores over time for Studies 1 and 2.
|
Figure 1. Weekly Mean Pain Scores (Observed Cases in ITT Population): Study 1 |
|
|
|
Figure 2. Weekly Mean Pain Scores (Observed Cases in ITT Population): Study 2 |
|
|
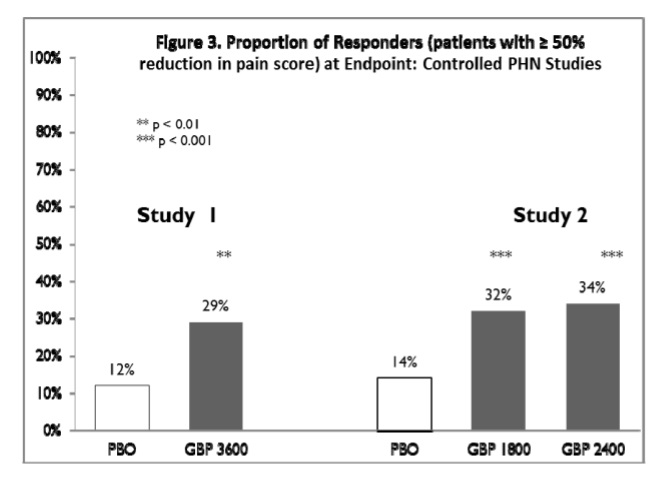
The proportion of responders (those patients reporting at least 50% improvement in endpoint pain score compared to baseline) was calculated for each study (Figure 3).
|
Figure 3. Proportion of Responders (patients with ≥ 50% reduction in pain score) at Endpoint: Controlled PHN Studies |
|
|
14.2 Epilepsy for Partial Onset Seizures (Adjunctive Therapy)
The effectiveness of gabapentin as adjunctive therapy (added to other antiepileptic drugs) was established in multicenter placebo-controlled, double-blind, parallel-group clinical trials in adult and pediatric patients (3 years and older) with refractory partial seizures.
Evidence of effectiveness was obtained in three trials conducted in 705 patients (age 12 years and above) and one trial conducted in 247 pediatric patients (3 to 12 years of age). The patients enrolled had a history of at least 4 partial seizures per month in spite of receiving one or more antiepileptic drugs at therapeutic levels and were observed on their established antiepileptic drug regimen during a 12-week baseline period (6 weeks in the study of pediatric patients). In patients continuing to have at least 2 (or 4 in some studies) seizures per month, gabapentin or placebo was then added on to the existing therapy during a 12-week treatment period. Effectiveness was assessed primarily on the basis of the percent of patients with a 50% or greater reduction in seizure frequency from baseline to treatment (the “responder rate”) and a derived measure called response ratio, a measure of change defined as (T -B)/(T + B), in which B is the patient’s baseline seizure frequency and T is the patient’s seizure frequency during treatment. Response ratio is distributed within the range -1 to +1. A zero value indicates no change while complete elimination of seizures would give a value of -1; increased seizure rates would give positive values. A response ratio of -0.33 corresponds to a 50% reduction in seizure frequency. The results given below are for all partial seizures in the intent-to-treat (all patients who received any doses of treatment) population in each study, unless otherwise indicated.
One study compared gabapentin 1,200 mg/day, in three divided doses with placebo. Responder rate was 23% (14/61) in the gabapentin group and 9% (6/66) in the placebo group; the difference between groups was statistically significant. Response ratio was also better in the gabapentin group (-0.199) than in the placebo group (-0.044), a difference that also achieved statistical significance.
A second study compared primarily gabapentin 1,200 mg/day, in three divided doses (N=101), with placebo (N=98). Additional smaller gabapentin dosage groups (600 mg/day, N=53; 1,800 mg/day, N=54) were also studied for information regarding dose response. Responder rate was higher in the gabapentin 1,200 mg/day group (16%) than in the placebo group (8%), but the difference was not statistically significant. The responder rate at 600 mg (17%) was also not significantly higher than in the placebo, but the responder rate in the 1,800 mg group (26%) was statistically significantly superior to the placebo rate. Response ratio was better in the gabapentin 1,200 mg/day group (-0.103) than in the placebo group (-0.022); but this difference was also not statistically significant (p = 0.224). A better response was seen in the gabapentin 600 mg/day group (-0.105) and 1,800 mg/day group (-0.222) than in the 1,200 mg/day group, with the 1,800 mg/day group achieving statistical significance compared to the placebo group.
A third study compared gabapentin 900 mg/day, in three divided doses (N=111), and placebo (N=109). An additional gabapentin 1,200 mg/day dosage group (N=52) provided dose-response data. A statistically significant difference in responder rate was seen in the gabapentin 900 mg/day group (22%) compared to that in the placebo group (10%). Response ratio was also statistically significantly superior in the gabapentin 900 mg/day group (-0.119) compared to that in the placebo group (-0.027), as was response ratio in 1,200 mg/day gabapentin (-0.184) compared to placebo.
Analyses were also performed in each study to examine the effect of gabapentin on preventing secondarily generalized tonic-clonic seizures. Patients who experienced a secondarily generalized tonic-clonic seizure in either the baseline or in the treatment period in all three placebo-controlled studies were included in these analyses. There were several response ratio comparisons that showed a statistically significant advantage for gabapentin compared to placebo and favorable trends for almost all comparisons.
Analysis of responder rate using combined data from all three studies and all doses (N=162, gabapentin; N=89, placebo) also showed a significant advantage for gabapentin over placebo in reducing the frequency of secondarily generalized tonic-clonic seizures.
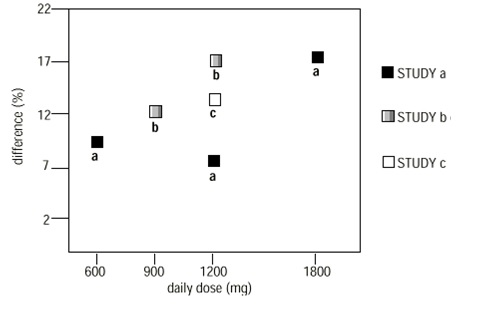
In two of the three controlled studies, more than one dose of gabapentin was used. Within each study, the results did not show a consistently increased response to dose. However, looking across studies, a trend toward increasing efficacy with increasing dose is evident (see Figure 4).
|
Figure 4. Responder Rate in Patients Receiving gabapentin Expressed as a Difference from Placebo by Dose and Study: Adjunctive Therapy Studies in Patients ≥12 Years of Age with Partial Seizures |
|
|
In the figure, treatment effect magnitude, measured on the Y axis in terms of the difference in the proportion of gabapentin and placebo-assigned patients attaining a 50% or greater reduction in seizure frequency from baseline, is plotted against the daily dose of gabapentin administered (X axis).
Although no formal analysis by gender has been performed, estimates of response (Response Ratio) derived from clinical trials (398 men, 307 women) indicate no important gender differences exist. There was no consistent pattern indicating that age had any effect on the response to gabapentin. There were insufficient numbers of patients of races other than Caucasian to permit a comparison of efficacy among racial groups.
A fourth study in pediatric patients age 3 to 12 years compared 25 to 35 mg/kg/day gabapentin (N=118) with placebo (N=127). For all partial seizures in the intent-to-treat population, the response ratio was statistically significantly better for the gabapentin group (-0.146) than for the placebo group (-0.079). For the same population, the responder rate for gabapentin (21%) was not significantly different from placebo (18%).
A study in pediatric patients age 1 month to 3 years compared 40 mg/kg/day gabapentin (N=38) with placebo (N=38) in patients who were receiving at least one marketed antiepileptic drug and had at least one partial seizure during the screening period (within 2 weeks prior to baseline). Patients had up to 48 hours of baseline and up to 72 hours of double-blind video EEG monitoring to record and count the occurrence of seizures. There were no statistically significant differences between treatments in either the response ratio or responder rate.