Palonosetron
These highlights do not include all the information needed to use PALONOSETRON HYDROCHLORIDE INJECTION safely and effectively. See full prescribing information for PALONOSETRON HYDROCHLORIDE INJECTION. PALONOSETRON HYDROCHLORIDE Injection, for intravenous use Initial U.S. Approval: 2003
b93fae8e-2b76-421e-95bc-ba08c211a7ea
HUMAN PRESCRIPTION DRUG LABEL
Jan 11, 2021
Fresenius Kabi USA, LLC
DUNS: 608775388
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Palonosetron
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (5)
Drug Labeling Information
WARNINGS AND PRECAUTIONS SECTION
5. WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Hypersensitivity reactions, including anaphylaxis and anaphylactic shock, have been reported with administration of Palonosetron Hydrochloride Injection [see Adverse Reactions (6.2)]. These reactions occurred in patients with or without known hypersensitivity to other 5-HT3 receptor antagonists. If hypersensitivity reactions occur, discontinue Palonosetron Hydrochloride Injection and initiate appropriate medical treatment. Do not reinitiate Palonosetron Hydrochloride Injection in patients who have previously experienced symptoms of hypersensitivity [see Contraindications (4)].
5.2 Serotonin Syndrome
The development of serotonin syndrome has been reported with 5-HT3 receptor antagonists. Most reports have been associated with concomitant use of serotonergic drugs (e.g., selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors, mirtazapine, fentanyl, lithium, tramadol, and intravenous methylene blue). Some of the reported cases were fatal. Serotonin syndrome occurring with overdose of another 5-HT3 receptor antagonist alone has also been reported. The majority of reports of serotonin syndrome related to 5-HT3 receptor antagonist use occurred in a post-anesthesia care unit or an infusion center.
Symptoms associated with serotonin syndrome may include the following combination of signs and symptoms: mental status changes (e.g. agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, with or without gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea). Patients should be monitored for the emergence of serotonin syndrome, especially with concomitant use of Palonosetron Hydrochloride Injection and other serotonergic drugs. If symptoms of serotonin syndrome occur, discontinue Palonosetron Hydrochloride Injection and initiate supportive treatment. Patients should be informed of the increased risk of serotonin syndrome, especially if Palonosetron Hydrochloride Injection is used concomitantly with other serotonergic drugs [see Drug Interactions (7.1)].
- Hypersensitivity reactions, including anaphylaxis and anaphylactic shock: reported in patients with or without known hypersensitivity to other selective 5-HT3 receptor antagonists. If symptoms occur, discontinue Palonosetron Hydrochloride Injection and initiate appropriate medical treatment. (5.1)
- Serotonin syndrome: reported with 5-HT3 receptor antagonists alone, but particularly with concomitant use of serotonergic drugs. (5.2, 7.1)
ADVERSE REACTIONS SECTION
6. ADVERSE REACTIONS
Serious or otherwise clinically significant adverse reactions reported in other sections of labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Serotonin Syndrome [see Warnings and Precautions (5.2)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Chemotherapy-Induced Nausea and Vomiting
Adults
In double-blind randomized clinical trials for the prevention of nausea and vomiting induced by MEC or HEC, 1,374 adult patients received a single dose of Palonosetron Hydrochloride Injection, ondansetron (Studies 1 and 3) or dolasetron (Study 2) administered 30 minutes prior to chemotherapy [see Clinical Studies (14.1)].
Adverse reactions were similar in frequency and severity in all 3 treatment groups. Common adverse reactions reported in at least 2% of patients in these trials are shown in Table 2.
Table 2: Common Adverse Reactions* in Adults with Receiving MEC (Studies 1 and 2) or HEC (Study 3)
| |||
|
Adverse Reaction |
Palonosetron |
Ondansetron |
Dolasetron |
|
Headache |
9% |
8% |
16% |
|
Constipation |
5% |
2% |
6% |
|
Diarrhea |
1% |
2% |
2% |
|
Dizziness |
1% |
2% |
2% |
|
Fatigue |
< 1% |
1% |
2% |
|
Abdominal Pain |
< 1% |
< 1% |
2% |
|
Insomnia |
< 1% |
1% |
2% |
Less common adverse reactions, reported in 1% or less of patients, in Studies 1, 2 and 3 were:
- Cardiovascular: non-sustained tachycardia, bradycardia, hypotension, hypertension, myocardial ischemia, extrasystoles, sinus tachycardia, sinus arrhythmia, supraventricular extrasystoles and QT prolongation.
- Dermatological: allergic dermatitis, rash
- Hearing and Vision: motion sickness, tinnitus, eye irritation and amblyopia
- Gastrointestinal System: diarrhea, dyspepsia, abdominal pain, dry mouth, hiccups and flatulence
- General: weakness, fatigue, fever, hot flash, flu-like syndrome
- Liver: transient, asymptomatic increases in AST and/or ALT and bilirubin. These changes occurred predominantly in patients receiving highly emetogenic chemotherapy
- Metabolic: hyperkalemia, electrolyte fluctuations, hyperglycemia, metabolic acidosis, glycosuria, appetite decrease, anorexia
- Musculoskeletal: arthralgia
- Nervous System: dizziness, somnolence, insomnia, hypersomnia, paresthesia
- Psychiatric: anxiety, euphoric mood
- Urinary System: urinary retention
- Vascular: vein discoloration, vein distention
In other studies, 2 subjects experienced severe constipation following a single Palonosetron Hydrochloride Injection dose of approximately 0.75 mg (three times the recommended dose).
Pediatrics Aged 2 Months to 17 Years
In a pediatric clinical trial, 163 pediatric cancer patients with a mean age of 8 years received a single 20 mcg/kg (maximum 1.5 mg) intravenous infusion of Palonosetron Hydrochloride Injection 30 minutes before beginning the first cycle of emetogenic chemotherapy [see Clinical Studies (14.2)]. Adverse reactions were evaluated in pediatric patients receiving Palonosetron Hydrochloride Injection for up to 4 chemotherapy cycles. The following adverse reactions were reported in less than 1% of patients:
- Nervous System: headache, dizziness, dyskinesia.
- General: infusion site pain.
- Dermatological: allergic dermatitis, skin disorder.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of palonosetron HCl. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Hypersensitivity reactions: including dyspnea, bronchospasm, swelling/edema, erythema, pruritus, rash, urticaria, anaphylaxis and anaphylactic shock [see Warnings and Precautions (5.1)]
- Injection site reactions: including burning, induration, discomfort and pain
Most common adverse reactions in
- chemotherapy-induced nausea and vomiting in adults (≥5%) are: headache and constipation (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
CLINICAL STUDIES SECTION
14. CLINICAL STUDIES
14.1 Prevention of Nausea and Vomiting Associated with MEC and HEC in
Adults
Efficacy of a single intravenous dose of Palonosetron Hydrochloride Injection in preventing acute and delayed nausea and vomiting associated with MEC or HEC were studied in 4 trials. In these double-blind studies, complete response rates (no emetic episodes and no rescue medication) and other efficacy parameters were assessed through at least 120 hours after administration of chemotherapy.
The safety and efficacy of Palonosetron Hydrochloride Injection in repeated courses of chemotherapy was also assessed.
Moderately Emetogenic Chemotherapy
Two double-blind trials (Study 1 and Study 2) involving 1,132 patients compared a single dose of Palonosetron Hydrochloride Injection with either a single-dose of ondansetron (Study 1) or dolasetron (Study 2) given 30 minutes prior to MEC, including carboplatin, cisplatin ≤ 50 mg/m2, cyclophosphamide < 1,500 mg/m2, doxorubicin > 25 mg/m2, epirubicin, irinotecan, and methotrexate
250 mg/m2. Concomitant corticosteroids were not administered prophylactically in Study 1 and were only used by 4 to 6% of patients in Study
- The majority of patients in these studies were women (77%), White (65%) and naïve to previous chemotherapy (54%). The mean age was 55 years.
Highly Emetogenic Chemotherapy
A double-blind, dose-ranging trial evaluated the efficacy of a single intravenous dose of Palonosetron Hydrochloride Injection from 0.3 to 90 mcg/kg (equivalent to < 0.1 mg to 6 mg fixed dose) in 161 chemotherapy-naïve adult cancer patients receiving HEC, either cisplatin ≥ 70 mg/m2 or cyclophosphamide
1,100 mg/m2. Concomitant corticosteroids were not administered prophylactically. Analysis of data from this trial indicates that 0.25 mg is the lowest effective dose in preventing acute nausea and vomiting associated with HEC.
A double-blind trial involving 667 patients compared a single intravenous dose of Palonosetron Hydrochloride Injection with a single intravenous dose of ondansetron (Study 3) given 30 minutes prior to HEC, including cisplatin ≥ 60 mg/m2, cyclophosphamide > 1,500 mg/m2, and dacarbazine. Corticosteroids were co-administered prophylactically before chemotherapy in 67% of patients. Of the 667 patients, 51% were women, 60% White, and 59% naïve to previous chemotherapy. The mean age was 52 years.
Efficacy Results
Studies 1, 2 and 3 show that Palonosetron Hydrochloride Injection was effective in the prevention of nausea and vomiting associated with initial and repeat courses of MEC and HEC in the acute phase (0 to 24 hours) [Table 5]. Clinical superiority over other 5-HT3 receptor antagonists has not been adequately demonstrated in the acute phase. In Study 3, efficacy was greater when prophylactic corticosteroids were administered concomitantly.
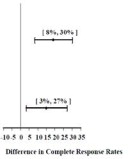
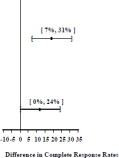
Studies 1 and 2 show that Palonosetron Hydrochloride Injection was effective in the prevention of nausea and vomiting associated with initial and repeat course of MEC in the delayed phase (24 to 120 hours) [Table 6] and overall phase (0 to 120 hours) [Table 7].
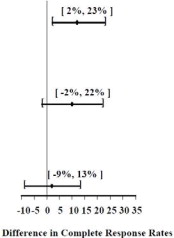
Table 5: Prevention of Acute Nausea and Vomiting (0 to 24 Hours) in Adults with Nausea and Vomiting Associated with MEC or HEC in Studies 1, 2 and 3: Complete Response Rates|
a Intent-to-treat cohort | ||||||
|
b 2-sided Fisher's exact test. Significance level at α=0.025. | ||||||
|
c These studies were designed to show non-inferiority. A lower bound greater than -15% demonstrates non-inferiority between Palonosetron Hydrochloride Injection and comparator. | ||||||
|
Chemotherapy |
Study |
Treatment Group |
N****a |
% with Complete Response |
p-value****b |
97.5% Confidence Interval Palonosetron Hydrochloride Injection minus Comparator****c
|
|
Moderately Emetogenic |
1 |
Palonosetron Hydrochloride Injection 0.25 mg intravenously |
189 |
81 |
0.009 | |
|
Ondansetron 32 mg intravenously |
185 |
69 | ||||
|
2 |
Palonosetron Hydrochloride Injection 0.25 mg intravenously |
189 |
63 |
NS | ||
|
Dolasetron 100 mg intravenously |
191 |
53 | ||||
|
Highly Emetogenic |
3 |
Palonosetron Hydrochloride Injection 0.25 mg intravenously |
223 |
59 |
NS | |
|
Ondansetron 32 mg intravenously |
221 |
57 |
|
a Intent-to-treat cohort | ||||||
|
b 2-sided Fisher's exact test. Significance level at α=0.025. | ||||||
|
c These studies were designed to show non-inferiority. A lower bound greater than -15% demonstrates non-inferiority between Palonosetron Hydrochloride Injection and comparator. | ||||||
|
d Ondansetron 32 mg intravenous was used in the clinical trial. Although this dose was used in the trial, it is no longer the currently recommended dose. Refer to the ondansetron prescribing information for the current recommended dose. | ||||||
|
Chemotherapy |
Study |
Treatment Group |
N****a |
% with Complete Response |
p-value****b |
97.5% Confidence Interval Palonosetron Hydrochloride Injection minus Comparator****c
|
|
Moderately Emetogenic |
1 |
Palonosetron Hydrochloride Injection 0.25 mg intravenously |
189 |
74 |
<0.001 | |
|
Ondansetron |
185 |
55 | ||||
|
2 |
Palonosetron Hydrochloride Injection 0.25 mg intravenously |
189 |
54 |
0.004 | ||
|
Dolasetron |
191 |
39 |
|
a Intent-to-treat cohort | ||||||
|
b 2-sided Fisher's exact test. Significance level at α=0.025. | ||||||
|
c These studies were designed to show non-inferiority. A lower bound greater than -15% demonstrates non- inferiority between Palonosetron Hydrochloride Injection and comparator. | ||||||
|
d Ondansetron 32 mg intravenously was used in the clinical trial. Although this dose was used in the trial, it is no longer the currently recommended dose. Refer to the ondansetron prescribing information for the current recommended dose. | ||||||
|
Chemotherapy |
Study |
Treatment Group |
N****a |
% with Complete Response |
p-value****b |
97.5% Confidence Interval Palonosetron Hydrochloride Injection minus Comparator****c
|
|
Moderately Emetogenic |
1 |
Palonosetron Hydrochloride Injection |
189 |
69 |
<0.001 | |
|
Ondansetron |
185 |
50 | ||||
|
2 |
Palonosetron Hydrochloride Injection |
189 |
46 |
0.021 | ||
|
Dolasetron |
191 |
34 |
14.2 Prevention of Nausea and Vomiting Associated with Emetogenic
Chemotherapy, Including HEC in Pediatric Patients
One double-blind, active-controlled clinical trial was conducted in pediatric cancer patients. The total population (N = 327) had a mean age of 8.3 years (range 2 months to 16.9 years) and were 53% male; and 96% white. Patients were randomized and received a 20 mcg/kg (maximum 1.5 mg) intravenous infusion of Palonosetron Hydrochloride Injection 30 minutes prior to the start of emetogenic chemotherapy (followed by placebo infusions 4 and 8 hours after the dose of Palonosetron Hydrochloride injection) or 0.15 mg/kg of intravenous ondansetron 30 minutes prior to the start of emetogenic chemotherapy (followed by ondansetron 0.15 mg/kg infusions 4 and 8 hours after the first dose of ondansetron, with a maximum total dose of 32 mg). Emetogenic chemotherapies administered included doxorubicin, cyclophosphamide (< 1,500 mg/m2), ifosfamide, cisplatin, dactinomycin, carboplatin, and daunorubicin. Adjuvant corticosteroids, including dexamethasone, were administered with chemotherapy in 55% of patients.
Complete Response in the acute phase of the first cycle of chemotherapy was defined as no vomiting, no retching, and no rescue medication in the first 24 hours after starting chemotherapy. Efficacy was based on demonstrating non- inferiority of intravenous Palonosetron Hydrochloride injection compared to intravenous ondansetron. Non-inferiority criteria were met if the lower bound of the 97.5% confidence interval for the difference in Complete Response rates of intravenous Palonosetron Hydrochloride injection minus intravenous ondansetron was larger than -15%. The non-inferiority margin was 15%.
Efficacy Results
As shown in Table 8, intravenous Palonosetron Hydrochloride Injection 20 mcg/kg (maximum 1.5 mg) demonstrated non-inferiority to the active comparator during the 0 to 24-hour time interval.
Table 8: Prevention of Acute Nausea and Vomiting (0 to 24 Hours) Associated with Emetogenic Chemotherapy in Pediatric Patients: Complete Response Rates|
a To adjust for multiplicity of treatment groups, a lower-bound of a 97.5% confidence interval was used to compare to -15%, the negative value of the non-inferiority margin. | ||
|
Palonosetron Hydrochloride Injection 20 mcg/kg intravenously |
Ondansetron |
Difference [97.5% Confidence Interval]a: Palonosetron Hydrochloride Injection
minus |
|
59.4% |
58.6% |
0.36% [-11.7%, 12.4%] |
In patients that received Palonosetron Hydrochloride Injection at a lower dose than the recommended dose of 20 mcg/kg, non-inferiority criteria were not met.
INFORMATION FOR PATIENTS SECTION
17. PATIENT COUNSELING INFORMATION
Advise the patient or caregiver to read the FDA-approved patient labeling (Patient Information).
Hypersensitivity Reactions
Advise patients that hypersensitivity reactions, including anaphylaxis and anaphylactic shock, have been reported in patients with or without known hypersensitivity to other 5-HT3 receptor antagonists. Advise patients to seek immediate medical attention if any signs or symptoms of a hypersensitivity reaction occur with administration of Palonosetron Hydrochloride Injection [see Warnings and Precautions (5.1)].
Serotonin Syndrome
Advise patients of the possibility of serotonin syndrome, especially with concomitant use of Palonosetron Hydrochloride injection and another serotonergic agent such as medications to treat depression and migraines. Advise patients to seek immediate medical attention if the following symptoms occur: changes in mental status, autonomic instability, neuromuscular symptoms with or without gastrointestinal symptoms [see Warnings and Precautions (5.2)].

Lake Zurich, IL 60047
Made in Austria
www.fresenius-kabi.com/us
451400B