Pregabalin
These highlights do not include all the information needed to use PREGABALIN CAPSULES safely and effectively. See full prescribing information for PREGABALIN CAPSULES. PREGABALIN capsules, for oral use, CVInitial U.S. Approval: 2004
17a8087c-579f-42de-ae92-006ea64def2a
HUMAN PRESCRIPTION DRUG LABEL
Jan 18, 2023
Cipla USA, Inc.
DUNS: 078719707
Products 8
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
PREGABALIN
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (14)
PREGABALIN
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (14)
PREGABALIN
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (14)
PREGABALIN
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (17)
PREGABALIN
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (17)
PREGABALIN
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (17)
PREGABALIN
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (17)
PREGABALIN
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (17)
Drug Labeling Information
RECENT MAJOR CHANGES SECTION
RECENT MAJOR CHANGES
|
Indications and Usage (1) |
5/2019 |
|
Warnings and Precautions, Respiratory Depression (5.4) |
4/2020 |
|
Warning and Precautions, Dizziness and Somnolence (5.5) |
5/2019 |
USE IN SPECIFIC POPULATIONS SECTION
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in
women exposed to pregabalin capsules during pregnancy. To provide information
regarding the effects of in utero exposure to pregabalin capsules, physicians
are advised to recommend that pregnant patients taking pregabalin capsules
enroll in the North American Antiepileptic Drug (NAAED) Pregnancy Registry.
This can be done by calling the toll free number 1-888-233-2334, and must be
done by patients themselves.
Information on the registry can also be found at the website
http://www.aedpregnancyregistry.org/.
Risk Summary
There are no adequate and well-controlled studies with pregabalin capsules in
pregnant women.
However, in animal reproduction studies, increased incidences of fetal structural abnormalities and other manifestations of developmental toxicity, including skeletal malformations, retarded ossification, and decreased fetal body weight were observed in the offspring of rats and rabbits given pregabalin orally during organogenesis, at doses that produced plasma pregabalin exposures (AUC) greater than or equal to 16 times human exposure at the maximum recommended dose (MRD) of 600 mg/day [see Data]. In an animal development study, lethality, growth retardation, and nervous and reproductive system functional impairment were observed in the offspring of rats given pregabalin during gestation and lactation. The no-effect dose for developmental toxicity was approximately twice the human exposure at MRD. The background risk of major birth defects and miscarriage for the indicated populations are unknown. However, the background risk in the U.S. general population of major birth defects is 2-4% and of miscarriage is 15-20% of clinically recognized pregnancies. Advise pregnant women of the potential risk to a fetus.
Data
Animal Data
When pregnant rats were given pregabalin (500, 1,250, or 2,500 mg/kg) orally throughout the period of organogenesis, incidences of specific skull alterations attributed to abnormally advanced ossification (premature fusion of the jugal and nasal sutures) were increased at greater than or equal to 1,250 mg/kg, and incidences of skeletal variations and retarded ossification were increased at all doses. Fetal body weights were decreased at the highest dose. The low dose in this study was associated with a plasma exposure (AUC) approximately 17 times human exposure at the MRD of 600 mg/day. A no-effect dose for rat embryo-fetal developmental toxicity was not established.
When pregnant rabbits were given pregabalin capsules (250, 500, or 1,250 mg/kg) orally throughout the period of organogenesis, decreased fetal body weight and increased incidences of skeletal malformations, visceral variations, and retarded ossification were observed at the highest dose. The no-effect dose for developmental toxicity in rabbits (500 mg/kg) was associated with a plasma exposure approximately 16 times human exposure at the MRD.
In a study in which female rats were dosed with pregabalin capsules (50, 100, 250, 1,250, or 2,500 mg/kg) throughout gestation and lactation, offspring growth was reduced at greater than or equal to 100 mg/kg and offspring survival was decreased at greater than or equal to 250 mg/kg. The effect on offspring survival was pronounced at doses greater than or equal to 1,250 mg/kg, with 100% mortality in high-dose litters. When offspring were tested as adults, neurobehavioral abnormalities (decreased auditory startle responding) were observed at greater than or equal to 250 mg/kg and reproductive impairment (decreased fertility and litter size) was seen at 1,250 mg/kg. The no-effect dose for pre- and postnatal developmental toxicity in rats (50 mg/kg) produced a plasma exposure approximately 2 times human exposure at the MRD.
In the prenatal-postnatal study in rats, pregabalin prolonged gestation and induced dystocia at exposures greater than or equal to 50 times the mean human exposure (AUC (0–24) of 123 μg∙hr/mL) at theMRD.
8.2 Lactation
Risk Summary
Small amounts of pregabalin have been detected in the milk of lactating women.
A pharmacokinetic study in lactating women detected pregabalin in breast milk
at average steady state concentrations approximately 76% of those in maternal
plasma. The estimated average daily infant dose of pregabalin from breast milk
(assuming mean milk consumption of 150 mL/kg/day) was 0.31 mg/kg/day, which on
a mg/kg basis would be approximately 7% of the maternal dose [see Data]. The
study did not evaluate the effects of pregabalin capsules on milk production
or the effects of pregabalin capsules on the breastfed infant.
Based on animal studies, there is a potential risk of tumorigenicity with pregabalin exposure via breast milk to the breastfed infant [see Nonclinical Toxicology (13.1)]. Available clinical study data in patients greater than 12 years of age do not provide a clear conclusion about the potential risk of tumorigenicity with pregabalin [see Warnings and Precautions (5.9)]. Because of the potential risk of tumorigenicity, breastfeeding is not recommended during treatment with pregabalin capsules.
Data
A pharmacokinetic study in ten lactating women, who were at least 12 weeks
postpartum, evaluated the concentrations of pregabalin in plasma and breast
milk. pregabalin capsules 150 mg oral capsule was given every 12 hours (300 mg
daily dose) for a total of four doses. pregabalin was detected in breast milk
at average steady-state concentrations approximately 76% of those in maternal
plasma. The estimated average daily infant dose of pregabalin from breast milk
(assuming mean milk consumption of 150 mL/kg/day) was 0.31 mg/kg/day, which on
a mg/kg basis would be approximately 7% of the maternal dose. The study did
not evaluate the effects of pregabalin capsules on milk production. Infants
did not receive breast milk obtained during the dosing period, therefore, the
effects of pregabalin capsules on the breast fed infant were not evaluated.
8.3 Females and Males of Reproductive Potential
Infertility
Male
Effects on Spermatogenesis
In a randomized, double-blind, placebo-controlled non-inferiority study to assess the effect of pregabalin on sperm characteristics, healthy male subjects received pregabalin at a daily dose up to 600 mg (n=111) or placebo (n=109) for 13 weeks (one complete sperm cycle) followed by a 13-week washout period (off-drug). A total of 65 subjects in the pregabalin group (59%) and 62 subjects in the placebo group (57%) were included in the per protocol (PP) population. These subjects took study drug for at least 8 weeks, had appropriate timing of semen collections and did not have any significant protocol violations. Among these subjects, approximately 9% of the pregabalin group (6/65) vs. 3% in the placebo group (2/62) had greater than or equal to 50% reduction in mean sperm concentrations from baseline at Week 26 (the primary endpoint). The difference between pregabalin and placebo was within the pre-specified non-inferiority margin of 20%. There were no adverse effects of pregabalin on sperm morphology, sperm motility, serum FSH or serum testosterone levels as compared to placebo. In subjects in the PP population with greater than or equal to 50% reduction in sperm concentration from baseline, sperm concentrations were no longer reduced by greater than or equal to 50% in any affected subject after an additional 3 months off-drug. In one subject, however, subsequent semen analyses demonstrated reductions from baseline of greater than or equal to 50% at 9 and 12 months off- drug. The clinical relevance of these data is unknown.
In the animal fertility study with pregabalin in male rats, adverse reproductive and developmental effects were observed [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
Neuropathic Pain Associated with Diabetic Peripheral Neuropathy, Postherpetic Neuralgia, and Neuropathic Pain Associated with Spinal Cord Injury
Safety and effectiveness in pediatric patients have not been established.
Fibromyalgia
Safety and effectiveness in pediatric patients have not been established.
Adjunctive Therapy for Partial-Onset Seizures
Safety and effectiveness in pediatric patients below the age of 1 month have not been established.
Juvenile Animal Data
In studies in which pregabalin (50 to 500 mg/kg) was orally administered to young rats from early in the postnatal period (Postnatal Day 7) through sexual maturity, neurobehavioral abnormalities (deficits in learning and memory, altered locomotor activity, decreased auditory startle responding and habituation) and reproductive impairment (delayed sexual maturation and decreased fertility in males and females) were observed at doses greater than or equal to 50 mg/kg. The neurobehavioral changes of acoustic startle persisted at greater than or equal to 250 mg/kg and locomotor activity and water maze performance at greater than or equal to 500 mg/kg in animals tested after cessation of dosing and, thus, were considered to represent long-term effects. The low effect dose for developmental neurotoxicity and reproductive impairment in juvenile rats (50 mg/kg) was associated with a plasma pregabalin exposure (AUC) approximately equal to human exposure at the maximum recommended dose of 600 mg/day. A no-effect dose was not established.
Information describing a clinical study in which efficacy was not demonstrated in patients is approved for Pfizer Inc.’s Lyrica® (pregabalin) products. Additional pediatric use information is approved for Pfizer’s LYRICA (pregabalin) Capsules and Oral Solution products. However, due to Pfizer’s marketing exclusivity rights, this drug product is not labeled with that pediatric information.
8.5 Geriatric Use
In controlled clinical studies of pregabalin capsules in neuropathic pain associated with diabetic peripheral neuropathy, 246 patients were 65 to 74 years of age, and 73 patients were 75 years of age or older.
In controlled clinical studies of pregabalin capsules in neuropathic pain associated with postherpetic neuralgia, 282 patients were 65 to 74 years of age, and 379 patients were 75 years of age or older.
In controlled clinical studies of pregabalin capsules in epilepsy, there were only 10 patients 65 to 74 years of age, and 2 patients who were 75 years of age or older.
No overall differences in safety and efficacy were observed between these patients and younger patients.
In controlled clinical studies of pregabalin capsules in fibromyalgia, 106 patients were 65 years of age or older. Although the adverse reaction profile was similar between the two age groups, the following neurological adverse reactions were more frequent in patients 65 years of age or older: dizziness, vision blurred, balance disorder, tremor, confusional state, coordination abnormal, and lethargy.
Pregabalin capsules are known to be substantially excreted by the kidney, and the risk of toxic reactions to pregabalin capsules may be greater in patients with impaired renal function. Because pregabalin capsules are eliminated primarily by renal excretion, adjust the dose for elderly patients with renal impairment [see Dosage and Administration (2.7)].
8.6 Renal Impairment
Pregabalin capsules is eliminated primarily by renal excretion and dose adjustment is recommended for adult patients with renal impairment [see Dosage and Administration (2.7) and Clinical Pharmacology (12.3)]. The use of pregabalin capsules in pediatric patients with compromised renal function has not been studied.
- Pregnancy: May cause fetal harm. Advise of potential risk to the fetus. (8.1)
- Lactation: Breastfeeding is not recommended. (8.2)
CLINICAL STUDIES SECTION
14 CLINICAL STUDIES
14.1 Neuropathic Pain Associated with Diabetic Peripheral Neuropathy
The efficacy of the maximum recommended dose of pregabalin capsules for the management of neuropathic pain associated with diabetic peripheral neuropathy was established in three double- blind, placebo-controlled, multicenter studies with three times a day dosing, two of which studied the maximum recommended dose. Patients were enrolled with either Type 1 or Type 2 diabetes mellitus and a diagnosis of painful distal symmetrical sensorimotor polyneuropathy for 1 to 5 years. A total of 89% of patients completed Studies DPN 1 and DPN 2. The patients had a minimum mean baseline pain score of greater than or equal to 4 on an 11-point numerical pain rating scale ranging from 0 (no pain) to 10 (worst possible pain). The baseline mean pain scores across the two studies ranged from 6.1 to 6.7. Patients were permitted up to 4 grams of acetaminophen per day as needed for pain, in addition to pregabalin. Patients recorded their pain daily in a diary.
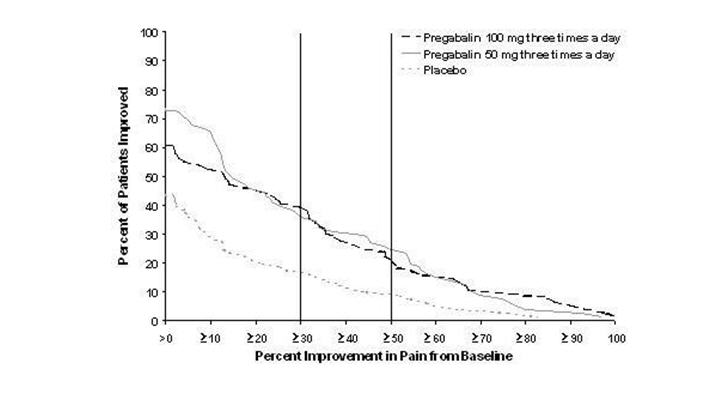
Study DPN 1: This 5-week study compared pregabalin capsules 25, 100, or 200 mg three times a day with placebo. Treatment with pregabalin capsules 100 and 200 mg three times a day statistically significantly improved the endpoint mean pain score and increased the proportion of patients with at least a 50% reduction in pain score from baseline. There was no evidence of a greater effect on pain scores of the 200 mg three times a day dose than the 100 mg three times a day dose, but there was evidence of dose dependent adverse reactions [see Adverse Reactions (6.1)]. For a range of levels of improvement in pain intensity from baseline to study endpoint, Figure 1 shows the fraction of patients achieving that level of improvement. The figure is cumulative, so that patients whose change from baseline is, for example, 50%, are also included at every level of improvement below 50%. Patients who did not complete the study were assigned 0% improvement. Some patients experienced a decrease in pain as early as Week 1, which persisted throughout the study.
Figure 1: Patients Achieving Various Levels of Improvement in Pain Intensity – Study DPN 1

Study DPN 2: This 8-week study compared pregabalin capsules 100 mg three times a day with placebo. Treatment with pregabalin capsules 100 mg three times a day statistically significantly improved the endpoint mean pain score and increased the proportion of patients with at least a 50% reduction in pain score from baseline. For various levels of improvement in pain intensity from baseline to study endpoint, Figure 2 shows the fraction of patients achieving that level of improvement. The figure is cumulative, so that patients whose change from baseline is, for example, 50%, are also included at every level of improvement below 50%. Patients who did not complete the study were assigned 0% improvement. Some patients experienced a decrease in pain as early as Week 1, which persisted throughout the study.
Figure 2: Patients Achieving Various Levels of Improvement in Pain Intensity– Study DPN 2

14.2 Postherpetic Neuralgia
The efficacy of pregabalin capsules for the management of postherpetic neuralgia was established in three double-blind, placebo-controlled, multicenter studies. These studies enrolled patients with neuralgia persisting for at least 3 months following healing of herpes zoster rash and a minimum baseline score of greater than or equal to 4 on an 11-point numerical pain rating scale ranging from 0 (no pain) to 10 (worst possible pain). Seventy- three percent of patients completed the studies. The baseline mean pain scores across the 3 studies ranged from 6 to 7. Patients were permitted up to 4 grams of acetaminophen per day as needed for pain, in addition to pregabalin. Patients recorded their pain daily in a diary.
Study PHN 1: This 13-week study compared pregabalin capsules 75, 150, and 300 mg twice daily with placebo. Patients with creatinine clearance (CLcr) between 30 to 60 mL/min were randomized to 75 mg, 150 mg, or placebo twice daily. Patients with creatinine clearance greater than 60 mL/min were randomized to 75 mg, 150 mg, 300 mg or placebo twice daily. In patients with creatinine clearance greater than 60 mL/min treatment with all doses of pregabalin capsules statistically significantly improved the endpoint mean pain score and increased the proportion of patients with at least a 50% reduction in pain score from baseline. Despite differences in dosing based on renal function, patients with creatinine clearance between 30 to 60 mL/min tolerated pregabalin capsules less well than patients with creatinine clearance greater than 60 mL/min as evidenced by higher rates of discontinuation due to adverse reactions. For various levels of improvement in pain intensity from baseline to study endpoint, Figure 3 shows the fraction of patients achieving that level of improvement. The figure is cumulative, so that patients whose change from baseline is, for example, 50%, are also included at every level of improvement below 50%. Patients who did not complete the study were assigned 0% improvement. Some patients experienced a decrease in pain as early as Week 1, which persisted throughout the study.
Figure 3: Patients Achieving Various Levels of Improvement in Pain Intensity– Study PHN 1

Study PHN 2: This 8-week study compared pregabalin capsules 100 or 200 mg three times a day with placebo, with doses assigned based on creatinine clearance. Patients with creatinine clearance between 30 to 60 mL/min were treated with 100 mg three times a day, and patients with creatinine clearance greater than 60 mL/min were treated with 200 mg three times daily.
Treatment with pregabalin capsules statistically significantly improved the endpoint mean pain score and increased the proportion of patients with at least a 50% reduction in pain score from baseline. For various levels of improvement in pain intensity from baseline to study endpoint, Figure 4 shows the fraction of patients achieving those levels of improvement. The figure is cumulative, so that patients whose change from baseline is, for example, 50%, are also included at every level of improvement below 50%. Patients who did not complete the study were assigned 0% improvement. Some patients experienced a decrease in pain as early as Week 1, which persisted throughout the study.
Figure 4: Patients Achieving Various Levels of Improvement in Pain Intensity – Study PHN 2

Study PHN 3: This 8-week study compared pregabalin capsules 50 or 100 mg three times a day with placebo with doses assigned regardless of creatinine clearance. Treatment with pregabalin capsules 50 and 100 mg three times a day statistically significantly improved the endpoint mean pain score and increased the proportion of patients with at least a 50% reduction in pain score from baseline.
Patients with creatinine clearance between 30 to 60 mL/min tolerated pregabalin capsules less well than patients with creatinine clearance greater than 60 mL/min as evidenced by markedly higher rates of discontinuation due to adverse reactions. For various levels of improvement in pain intensity from baseline to study endpoint, Figure 5 shows the fraction of patients achieving that level of improvement. The figure is cumulative, so that patients whose change from baseline is, for example, 50%, are also included at every level of improvement below 50%. Patients who did not complete the study were assigned 0% improvement. Some patients experienced a decrease in pain as early as Week 1, which persisted throughout the study.
Figure 5: Patients Achieving Various Levels of Improvement in Pain Intensity– Study PHN 3
14.3 Adjunctive Therapy for Partial-Onset Seizures in Patients 17 Years of
Age and Older
Adjunctive Therapy for Partial-Onset Seizures in Adult Patients
The efficacy of pregabalin capsules as adjunctive therapy for partial-onset seizures in adult patients was established in three 12-week, randomized, double-blind, placebo-controlled, multicenter studies.
Patients were enrolled who had partial-onset seizures with or without secondary generalization and were not adequately controlled with 1 to 3 concomitant antiepileptic drugs (AEDs). Patients taking gabapentin were required to discontinue gabapentin treatment 1 week prior to entering baseline. During an 8-week baseline period, patients had to experience at least 6 partial-onset seizures with no seizure-free period exceeding 4 weeks. The mean duration of epilepsy was 25 years in these 3 studies and the mean and median baseline seizure frequencies were 22.5 and 10 seizures per month, respectively. Approximately half of the patients were taking 2 concurrent AEDs at baseline. Among the pregabalin capsules-treated patients, 80% completed the double-blind phase of the studies.
Table 11 shows median baseline seizure rates and median percent reduction in seizure frequency by dose.
Table 11. Seizure Response in Controlled, Adjunctive Epilepsy Studies in Adults|
Daily Dose of Pregabalin |
Dosing Regimen |
N |
Baseline Seizure Frequency/mo |
Median % Change from Baseline |
p-value, vs. placebo |
|---|---|---|---|---|---|
|
Study E1 | |||||
|
Placebo |
BID |
100 |
9.5 |
0 | |
|
50 mg/day |
BID |
88 |
10.3 |
-9 |
0.4230 |
|
150 mg/day |
BID |
86 |
8.8 |
-35 |
0.0001 |
|
300 mg/day |
BID |
90 |
9.8 |
-37 |
0.0001 |
|
600 mg/day |
BID |
89 |
9.0 |
-51 |
0.0001 |
|
Study E2 | |||||
|
Placebo |
TID |
96 |
9.3 |
1 | |
|
150 mg/day |
TID |
99 |
11.5 |
-17 |
0.0007 |
|
600 mg/day |
TID |
92 |
12.3 |
-43 |
0.0001 |
|
Study E3 | |||||
|
Placebo |
BID/TID |
98 |
11 |
-1 | |
|
600 mg/day |
BID |
103 |
9.5 |
-36 |
0.0001 |
|
600 mg/day |
TID |
111 |
10 |
-48 |
0.0001 |
In the first study (E1), there was evidence of a dose-response relationship for total daily doses of pregabalin capsules between 150 and 600 mg/day; a dose of 50 mg/day was not effective. In the first study (E1), each daily dose was divided into two equal doses (twice a day dosing). In the second study (E2), each daily dose was divided into three equal doses (three times a day dosing). In the third study (E3), the same total daily dose was divided into two equal doses for one group (twice a day dosing) and three equal doses for another group (three times a day dosing). While the three times a day dosing group in Study E3 performed numerically better than the twice a day dosing group, this difference was small and not statistically significant.
A secondary outcome measure included the responder rate (proportion of patients with greater than or equal to 50% reduction from baseline in partial seizure frequency). The following figure displays responder rate by dose for two of the studies.
Figure 6: Responder Rate by Adjunctive Epilepsy Study

Figure 7: Seizure Reduction by Dose (All Partial-Onset Seizures) for Studies E1, E2, and E3

Subset evaluations of the antiseizure efficacy of pregabalin capsules showed no clinically important differences as a function of age, gender, or race.
Pediatric use information is approved for Pfizer’s LYRICA (pregabalin) Capsules and Oral Solution products. However, due to Pfizer’s marketing exclusivity rights, this drug product is not labeled with that pediatric information.
14.4 Management of Fibromyalgia
The efficacy of pregabalin capsules for management of fibromyalgia was established in one 14-week, double-blind, placebo-controlled, multicenter study (F1) and one six-month, randomized withdrawal study (F2). Studies F1 and F2 enrolled patients with a diagnosis of fibromyalgia using the American College of Rheumatology (ACR) criteria (history of widespread pain for 3 months, and pain present at 11 or more of the 18 specific tender point sites). The studies showed a reduction in pain by visual analog scale. In addition, improvement was demonstrated based on a patient global assessment (PGIC), and on the Fibromyalgia Impact Questionnaire (FIQ).
Study F1: This 14-week study compared pregabalin capsules total daily doses of 300 mg, 450 mg and 600 mg with placebo. Patients were enrolled with a minimum mean baseline pain score of greater than or equal to 4 on an 11-point numeric pain rating scale and a score of greater than or equal to 40 mm on the 100 mm pain visual analog scale (VAS). The baseline mean pain score in this trial was 6.7. Responders to placebo in an initial one-week run-in phase were not randomized into subsequent phases of the study. A total of 64% of patients randomized to pregabalin capsules completed the study. There was no evidence of a greater effect on pain scores of the 600 mg daily dose than the 450 mg daily dose, but there was evidence of dose-dependent adverse reactions [see Adverse Reactions (6.1)]. Some patients experienced a decrease in pain as early as Week 1, which persisted throughout the study. The results are summarized in Figure 9 and Table 14.
For various levels of improvement in pain intensity from baseline to study endpoint, Figure 9 shows the fraction of patients achieving that level of improvement. The figure is cumulative. Patients who did not complete the study were assigned 0% improvement. Some patients experienced a decrease in pain as early as Week 1, which persisted throughout the study.
Figure 9: Patients Achieving Various Levels of Improvement in Pain Intensity – Fibromyalgia Study F1

|
Patient Global Impression of Change | ||
|---|---|---|
|
Treatment Group |
% Any Improvement |
95% CI |
|
Placebo |
47.6 |
(40.0,55.2) |
|
PGB 300 |
68.1 |
(60.9, 75.3) |
|
PGB 450 |
77.8 |
(71.5, 84.0) |
|
PGB 600 |
66.1 |
(59.1, 73.1) |
|
PGB = Pregabalin |
Study F2: This randomized withdrawal study compared pregabalin capsules with placebo. Patients were titrated during a 6-week open-label dose optimization phase to a total daily dose of 300 mg, 450 mg, or 600 mg. Patients were considered to be responders if they had both: 1) at least a 50% reduction in pain (VAS) and, 2) rated their overall improvement on the PGIC as "much improved" or "very much improved.” Those who responded to treatment were then randomized in the double-blind treatment phase to either the dose achieved in the open-label phase or to placebo. Patients were treated for up to 6 months following randomization. Efficacy was assessed by time to loss of therapeutic response, defined as 1) less than 30% reduction in pain (VAS) from open-label baseline during two consecutive visits of the double-blind phase, or 2) worsening of FM symptoms necessitating an alternative treatment. Fifty-four percent of patients were able to titrate to an effective and tolerable dose of pregabalin capsules during the 6-week open-label phase. Of the patients entering the randomized treatment phase assigned to remain on pregabalin capsules, 38% of patients completed 26 weeks of treatment versus 19% of placebo-treated patients.
When considering return of pain or withdrawal due to adverse events as loss of response (LTR), treatment with pregabalin capsules resulted in a longer time to loss of therapeutic response than treatment with placebo. Fifty-three percent of the pregabalin-treated subjects compared to 33% of placebo patients remained on study drug and maintained a therapeutic response to Week 26 of the study. Treatment with pregabalin capsules also resulted in a longer time to loss of response based on the FIQ1, and longer time to loss of overall assessment of patient status, as measured by the PGIC2.
1Time to worsening of the FIQ was defined as the time to a 1-point increase from double-blind baseline in each of the subscales, and a 5-point increase from double-blind baseline evaluation for the FIQ total score.
2Time to PGIC lack of improvement was defined as time to PGIC assessments indicating less improvement than “much improvement.”
Figure 10: Time to Loss of Therapeutic Response, Fibromyalgia Study F2 (Kaplan-Meier Analysis)

14.5 Management of Neuropathic Pain Associated with Spinal Cord Injury
The efficacy of pregabalin capsules for the management of neuropathic pain associated with spinal cord injury was established in two double-blind, placebo-controlled, multicenter studies. Patients were enrolled with neuropathic pain associated with spinal cord injury that persisted continuously for at least three months or with relapses and remissions for at least six months. A total of 63% of patients completed study 1 and 84% completed study 2. The patients had a minimum mean baseline pain score of greater than or equal to 4 on an 11-point numerical pain rating scale ranging from 0 (no pain) to 10 (worst possible pain). The baseline mean pain scores across the two studies ranged from 6.5 to 6.7.
Patients were allowed to take opioids, non-opioid analgesics, antiepileptic drugs, muscle relaxants, and antidepressant drugs if the dose was stable for 30 days prior to screening. Patients were allowed to take acetaminophen and nonsteroidal anti-inflammatory drugs during the studies.
Study SCI 1: This 12-week, randomized, double-blind, parallel-group, multicenter, flexible dose (150-600 mg/day) study compared pregabalin with placebo. The 12-week study consisted of a 3- week dose adjustment phase and a 9-week dose maintenance phase. Treatment with pregabalin capsules 150-600 mg/day statistically significantly improved the endpoint weekly mean pain score, and increased the proportion of patients with at least a 30% and 50% reduction in pain score from baseline. The fraction of patients achieving various levels of improvement in pain intensity from baseline to Week 12 is presented in Figure 11. Some patients experienced a decrease in pain as early as week 1, which persisted throughout the study.
Figure 11: Patients Achieving Various Levels of Improvement in Pain Intensity – Study SCI 1

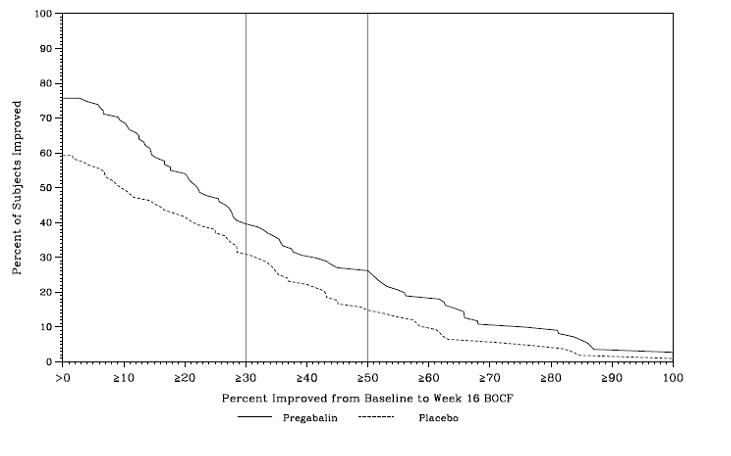
Study SCI 2: This 16-week, randomized, double-blind, placebo-controlled, parallel-group, multicenter, flexible dose (150-600 mg/day, in increments of 150 mg) study compared the efficacy, safety and tolerability of pregabalin with placebo. The 16-week study consisted of a 4-week dose adjustment phase and a 12-week dose maintenance phase. Treatment with pregabalin capsules statistically significantly improved the endpoint weekly mean pain score, and increased the proportion of patients with at least a 30% and 50% reduction in pain score from baseline. The fraction of patients achieving various levels of improvement in pain intensity from baseline to Week 16 is presented in Figure 12. Some patients experienced a decrease in pain as early as week 1, which persisted throughout the study.
Figure 12: Patients Achieving Various Levels of Improvement in Pain Intensity – Study SCI 2
SPL MEDGUIDE SECTION
** MEDICATION GUIDE**
** Pregabalin Capsules, CV**
** [pree gab’ a lin]**
Read this Medication Guide before you start taking pregabalin capsules and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment. If you have any questions about pregabalin capsules, ask your healthcare provider or pharmacist.
What is the most important information I should know about Pregabalin capsules?
Pregabalin capsules may cause serious side effects including:
*Serious, even life-threatening, allergic reactions *Suicidal thoughts or actions *Swelling of your hands, legs and feet *Dizziness and sleepiness *serious breathing problems
These serious side effects are described below:
*Serious, even life-threatening, allergic reactions.
Stop taking pregabalin capsules and call your healthcare provider right away
if you have any of these signs of a serious allergic reaction:
* swelling of your face, mouth, lips, gums, tongue, throat or neck
* trouble breathing
* rash, hives (raised bumps) or blisters
***Like other antiepileptic drugs, pregabalin capsules may cause suicidal thoughts or actions in a very small number of people, about 1 in 500.**Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you:
|
• thoughts about suicide or dying |
• trouble sleeping (insomnia) |
|
• attempts to commit suicide |
• new or worse irritability |
|
• new or worse depression |
• acting aggressive, being angry, or violent |
|
• new or worse anxiety |
• acting on dangerous impulses |
|
• feeling agitated or restless |
• an extreme increase in activity and talking (mania) |
|
• panic attacks |
• other unusual changes in behavior or mood |
If you have suicidal thoughts or actions, do not stop Pregabalin capsules without first talking to a healthcare provider.
- Stopping pregabalin capsules suddenly can cause serious problems.
- Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes.
How can I watch for early symptoms of suicidal thoughts and actions?
- Pay attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings.
- Keep all follow-up visits with your healthcare provider as scheduled.
- Call your healthcare provider between visits as needed, especially if you are worried about symptoms.
*Serious breathing problems can occur when Pregabalin Capsules is taken with other medicines that can cause severe sleepiness or decreased awareness, or when it is taken by someone who already has breathing problems. Watch for increased sleepiness or decreased breathing when starting Pregabalin Capsules or when the dose is increased. Get help right away if breathing problems occur. *Swelling of your hands, legs and feet. This swelling can be a serious problem for people with heart problems. *Dizziness and sleepiness. Do not drive a car, work with machines, or do other dangerous activities until you know how pregabalin capsules affects you. Ask your healthcare provider about when it will be okay to do these activities.
What are Pregabalin capsules?
Pregabalin capsules are a prescription medicine used in adults, 18 years of age and older, to treat:
|
• pain from damaged nerves (neuropathic pain) that happens with diabetes |
• fibromyalgia (pain all over your body) |
|
• pain from damaged nerves (neuropathic pain) that follows healing of shingles |
• pain from damaged nerves (neuropathic pain) that follows spinal cord injury |
It is not known if pregabalin capsules is safe and effective in people under 18 years of age for the treatment of fibromyalgia and neuropathic pain with diabetes, shingles, or spinal cord injury.
Pregabalin capsules is a prescription medicine used in people 17 years of age and older to treat:
- partial-onset seizures when taken together with other seizure medicines.
For the treatment of partial-onset seizures when taken together with other seizure medicines, it is not known if pregabalin capsules is safe and effective in children under 1 month of age
Who should not take Pregabalin capsules?
Do not take Pregabalin capsules if you are allergic to pregabalin or any of
the ingredients in Pregabalin capsules.
See**“What is the most important information I should know about pregabalin
capsules?”** for the signs of an allergic reaction.
See the end of this Medication Guide for a complete list of ingredients in pregabalin capsules.
What should I tell my healthcare provider before taking Pregabalin capsules?
Before taking pregabalin capsules, tell your healthcare provider about all your medical conditions, including if you:
- have or have had depression, mood problems or suicidal thoughts or behavior
- have breathing problems.
- have kidney problems or get kidney dialysis
- have heart problems including heart failure
- have a bleeding problem or a low blood platelet count
- have abused prescription medicines, street drugs, or alcohol in the past
- have ever had swelling of your face, mouth, tongue, lips, gums, neck, or throat (angioedema)
- plan to father a child. Animal studies have shown that pregabalin, the active ingredient in pregabalin capsules, made male animals less fertile and caused sperm to change. Also, in animal studies, birth defects were seen in the offspring (babies) of male animals treated with pregabalin. It is not known if these problems can happen in people who take pregabalin capsules.
***are pregnant or plan to become pregnant. Pregabalin capsules may harm your unborn baby.**You and your healthcare provider will decide if you should take pregabalin capsules while you are pregnant.
- If you become pregnant while taking pregabalin capsules, talk to your healthcare provider about registering with the North American Antiepileptic Drug Pregnancy Registry. You can enroll in this registry by calling 1-888-233-2334. The purpose of this registry is to collect information about the safety of antiepileptic drugs during pregnancy. Information about the registry can also be found at the website, http://www.aedpregnancyregistry.org/. *are breastfeeding or plan to breastfeed. Pregabalin capsules passes into your breast milk. It is not known if pregabalin capsule can harm your baby. Talk to your healthcare provider about the best way to feed your baby if you take pregabalin capsules.Breastfeeding is not recommended while taking pregabalin capsules.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins or herbal supplements. Pregabalin capsules and other medicines may affect each other causing side effects. Especially tell your healthcare provider if you take:
- angiotensin converting enzyme (ACE) inhibitors, which are used to treat many conditions, including high blood pressure. You may have a higher chance for swelling and hives if these medicines are taken with pregabalin capsules.
- Avandia (rosiglitazone) or Actos (pioglitazone) for diabetes. You may have a higher chance of weight gain or swelling of your hands or feet if these medicines are taken with pregabalin capsules.
- any opioid pain medicine (such as oxycodone), or medicines for anxiety (such as lorazepam) or insomnia (such as zolpidem). You may have a higher chance for dizziness, sleepiness or serious breathing problems if these medicines are taken with pregabalin capsules.
- any medicines that make you sleepy
Know the medicines you take. Keep a list of them with you to show your healthcare provider and pharmacist each time you get a new medicine. Do not start a new medicine without talking with your healthcare provider.
How should I take Pregabalin capsules?
- Take pregabalin capsules exactly as prescribed. Your healthcare provider will tell you how much pregabalin capsules to take and when to take it.
- Pregabalin capsules may be taken with or without food.
- Your healthcare provider may change your dose. Do not change your dose without talking to your healthcare provider.
- Do not stop taking pregabalin capsules without talking to your healthcare provider. If you stop taking pregabalin capsules suddenly you may have headaches, nausea, diarrhea, trouble sleeping, increased sweating, or you may feel anxious. If you have epilepsy and you stop taking pregabalin capsules suddenly, you may have seizures more often. Talk with your healthcare provider about how to stop pregabalin capsules slowly.
- If you miss a dose, take it as soon as you remember. If it is almost time for your next dose, just skip the missed dose. Take the next dose at your regular time. Do not take 2 doses at the same time.
- If you take too much pregabalin capsules, call your healthcare provider or poison control center, or go to the nearest emergency room right away.
What should I avoid while taking Pregabalin capsules?
*Do not drive a car, work with machines, or do other dangerous activities until you know how pregabalin capsules affects you. *Do not drink alcohol while taking pregabalin capsules. Pregabalin capsules and alcohol can affect each other and increase side effects such as sleepiness and dizziness.
What are the possible side effects of Pregabalin capsules?
Pregabalin capsules may cause serious side effects, including:
- See “What is the most important information I should know about pregabalin capsules?" *Muscle problems, muscle pain, soreness, or weakness. If you have these symptoms, especially if you feel sick and have a fever, tell your healthcare provider right away. ***Problems with your eyesight, including blurry vision.**Call your healthcare provider if you have any changes in your eyesight. ***Weight gain.**If you have diabetes, weight gain may affect the management of your diabetes. Weight gain can also be a serious problem for people with heart problems. *Feeling “high”
The most common side effects of pregabalin capsules in adults are:
|
• dizziness |
• trouble concentrating |
|
• blurry vision |
• swelling of hands and feet |
|
• weight gain |
• dry mouth |
|
• sleepiness |
Pregabalin capsules caused skin sores in animal studies. Skin sores did not happen in studies in people. If you have diabetes, you should pay attention to your skin while taking pregabalin capsules and tell your healthcare provider about any sores or skin problems.
Tell your healthcare provider about any side effect that bothers you or that does not go away.
These are not all the possible side effects of pregabalin capsules. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088
How should I store Pregabalin capsules?
- Store pregabalin capsules at room temperature between 20°C to 25°C (68°F to 77°F); [see USP Controlled Room Temperature].
- Safely throw away any pregabalin capsules that is out of date or no longer needed.
Keep Pregabalin capsules and all medicines out of the reach of children.
General information about the safe and effective use of Pregabalin capsules
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use pregabalin capsules for a condition for which it was not prescribed. Do not give pregabalin capsules to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about pregabalin capsules that is written for health professionals.
For more information about pregabalin capsules, call Cipla Ltd. at 1-866-604-3268
What are the ingredients in Pregabalin capsules?
Active ingredient: Pregabalin
Inactive ingredients: pregelatinized starch, corn starch, talc.
Capsule shells for 25 mg, 50 mg & 150 mg contains: gelatin and titanium dioxide
Capsule shells for 75 mg, 100 mg, 200 mg, 225 mg & 300 mg contains: gelatin, FD&C Blue No.1, FD&C Red No.40, FD&C Yellow No.6 and titanium dioxide.
Imprinting ink contains: shellac, dehydrated alcohol, isopropyl alcohol, butyl alcohol, propylene glycol, strong ammonia solution, black iron oxide, potassium hydroxide and purified water.
Pediatric use information is approved for Pfizer’s LYRICA (pregabalin) Capsules and Oral Solution products. However, due to Pfizer’s marketing exclusivity rights, this drug product is not labeled with that pediatric information.
All brand names mentioned are registered trademarks of their respective owners and are not of Cipla Limited.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Manufactured by:
InvaGen Pharmaceuticals, Inc.
(a subsidiary of Cipla Ltd.)
Hauppauge, NY 11788
Manufactured for:
Cipla USA, Inc.
10 Independence Boulevard, Suite 300
Warren, NJ 07059
Barcode# 962-04-2020
Revision: 04/2020
