PROCTOFOAM
{'content': {'@styleCode': 'bold'}}
3831e931-ec0a-11e3-ac10-0800200c9a66
HUMAN PRESCRIPTION DRUG LABEL
Feb 13, 2018
Meda Pharmaceuticals Inc.
DUNS: 051229602
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
pramoxine hydrochloride hydrocortisone acetate
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (12)
Drug Labeling Information
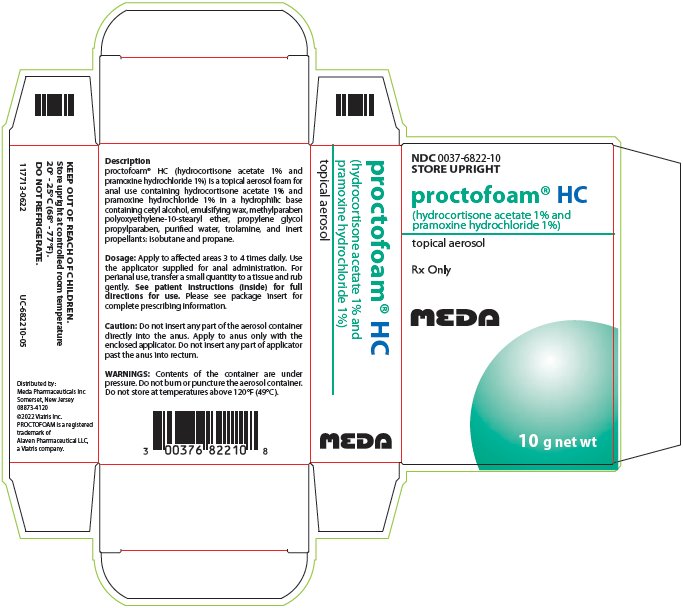
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 10 g
NDC0037-6822-10
STORE UPRIGHT
proctofoam® HC
(hydrocortisone acetate 1% and
pramoxine hydrochloride 1%)
topical aerosol
Rx Only
10 g net wt
Description
****proctofoam® HC (hydrocortisone acetate 1% and
pramoxine hydrochloride 1%) is a topical aerosol foam for
anal use containing hydrocortisone acetate 1% and
pramoxine hydrochloride 1% in a hydrophilic base
containing cetyl alcohol, emulsifying wax, methylparaben
polyoxyethylene-10-stearyl ether, propylene glycol
propylparaben, purified water, trolamine, and inert
propellants: isobutane and propane.
**Dosage:**Apply to affected areas 3 to 4 times daily. Use
the applicator supplied for anal administration. For
perianal use, transfer a small quantity to a tissue and rub
gently.See patient instructions (inside) for full
**directions for use.**Please see package insert for
complete prescribing information.
**Caution:**Do not insert any part of the aerosol container
directly into the anus. Apply to anus only with the
enclosed applicator. Do not insert any part of applicator
past the anus into rectum.
**WARNINGS:**Contents of the container are under
pressure. Do not burn or puncture the aerosol container.
Do not store at temperatures above 120°F (49°C).
KEEP OUT OF REACH OF CHILDREN.
Store upright at controlled room temperature
20° - 25°C (68° - 77°F).
DO NOT REFRIGERATE.
117713-0622 UC-682210-05
Distributed by:
Meda Pharmaceuticals Inc
Somerset, New Jersey
08873-4120
©2022 Viatris Inc.
PROCTOFOAM is a registered
trademark of
Alaven Pharmaceutical LLC,
a Viatris company.