Regulatory Information
HSA regulatory responsibility and product classification details
Regulatory Responsibility
Product Classification
Formulation Information
INJECTION
**DOSAGE AND ADMINISTRATION** NAROPIN should only be used by or under the supervision of clinicians experienced in regional anaesthesia. The presentations of NAROPIN injection solutions are intended for single use only. Any solution remaining from an opened container should be discarded. The lowest dosage that results in effective anaesthesia should be used and should be based on the status of the patient and the type of regional anaesthesia intended. In general, surgical anaesthesia requires the use of higher concentrations and doses than those required for analgesia. The following table is a guide to dosage. The clinician’s experience and knowledge of the patient’s physical status are of importance when deciding the dose. **Adults and children above 12 years of age** RECOMMENDED DOSAGES FOR NAROPIN SOLUTION FOR VARIOUS ANAESTHETIC PROCEDURES IN THE AVERAGE, HEALTHY, 70KG ADULT PATIENT. 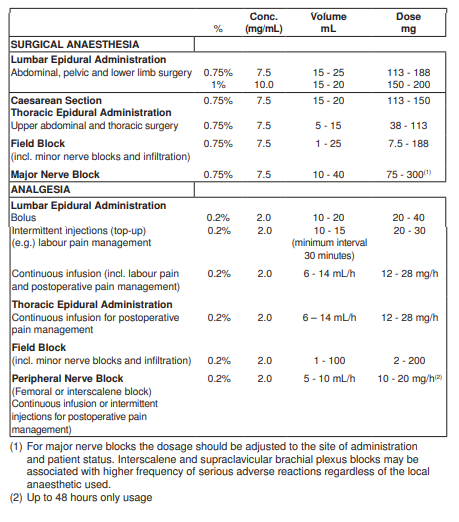 The appropriate concentration and volume for each procedure should be selected. The 1% (10 mg/mL) formulation is recommended for epidural anaesthesia in which a profound motor block is essential for surgery. There is no information available regarding the use of concentrations above 0.75% (7.5 mg/mL) for caesarean section. For further details of procedures please see current standard textbooks. **NOTE** **Careful aspiration before and during injection is recommended to avoid intravascular injection.** 1. **Test Dose** For epidural anaesthesia, or when a large dose is to be injected, a 3 – 5 mL test dose of a local anaesthetic solution, preferably containing 5 mcg/mL of adrenaline (e.g. 3 mL of Xylocaine® 2.0% with adrenaline 1:200,000) should be administered. Verbal contact and repeated monitoring of heart rate and blood pressure should be maintained for 5 minutes following the test dose after which, in the absence of signs of subarachnoid, intravascular or intrathecal injection, the main dose may be administered. An inadvertent intravascular injection may be recognised by a temporary increase in heart rate and an accidental intrathecal injection by signs of a spinal block. Prior to and during administration of the total dose, aspiration should be repeated. The main dose should be injected **slowly** at a rate of 25 – 50 mg/min, while closely observing the patient’s vital functions and maintaining verbal contact. If toxic symptoms or signs occur, the injection should be stopped immediately. 2. **Analgesia** When calculating the dosage for postoperative analgesia, the use of intraoperative local anaesthetic/s should be taken into account. For treatment of postoperative pain, the following technique can be recommended: Epidural analgesia is maintained with NAROPIN 0.2% (2 mg/mL) infusion. Infusion rates of 6 – 14 mL (12 – 28 mg) per hour provide adequate analgesia with only slight and non-progressive motor block in most cases of moderate to severe postoperative pain. With this technique a significant reduction in the need for opioids has been observed. Clinical experience supports the use of NAROPIN 0.2% (2 mg/mL) epidural infusions for up to 72 hours. Data for peripheral nerve block administered as a continuous peripheral infusion or intermittent injections support the use for up to 48 hours only at dosages of 10 – 20 mg/hr (5 – 10mL/hr). When prolonged epidural blocks are used, either by continuous infusion or repeated bolus administration, the risks of reaching a toxic plasma concentration or inducing local neural injury must be considered. Cumulative doses of up to 800 mg ropivacaine for surgery and postoperative analgesia administered over 24 hours were well tolerated in adults, as were postoperative continuous epidural infusions at rates up to 28 mg/hour for 72 hours. When prolonged peripheral nerve blocks are applied, either through continuous infusion or through repeated injections, the risk of reaching a toxic plasma concentration or inducing local neural injury must be considered. In clinical studies, femoral nerve block was established with 300 mg NAROPIN 0.75% (7.5 mg/mL) and interscalene block with 225 mg NAROPIN 0.75% (7.5 mg/mL), respectively, before surgery. Analgesia was then maintained with NAROPIN 0.2% (2 mg/mL). Infusion rates or intermittent injections of 10 – 20 mg per hour for 48 hours provided adequate analgesia and were well tolerated. **Use in Children** _**Dosage Recommendations for Paediatric Patients 0 up to and including 12 Years of Age**_ 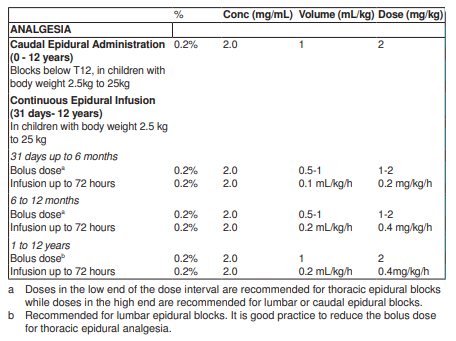 The doses in the table should be regarded as guidelines for use in paediatrics. Individual variations occur. In children with a high body weight a gradual reduction of the dosage is often necessary and should be based on the ideal body weight. The volume for single caudal epidural block and the volume for epidural bolus doses should not exceed 25 mL in any patient. Standard textbooks should be consulted for factors affecting specific block techniques and for individual patient requirements. Careful aspiration before and during injection is recommended to prevent intravascular injection. The patient’s vital functions should be observed closely during the injection. If toxic symptoms occur, the injection should be stopped immediately. A single caudal epidural injection of NAROPIN 0.2% (2 mg/mL) produces adequate postoperative analgesia below T12 in the majority of patients when a dose of 2 mg/kg is used in a volume of 1 mL/kg. In children above 4 years of age, doses up to 3 mg/kg have been used safely by the caudal route. The safety and efficacy of doses above 3 mg/kg have not been demonstrated and therefore cannot be recommended. The volume of the caudal epidural injection may be adjusted to achieve a different distribution to sensory block, as recommended in standard textbooks. Fractionation of the calculated local anaesthetic dose is recommended, whatever the route of administration. The use of NAROPIN in premature children has not been documented. **Use in Debilitated or Elderly Patients** Debilitated or elderly patients, including those with partial or complete heart conduction block, advanced liver disease or severe renal dysfunction should be given reduced dosage commensurate with their physical condition. Clinical studies with this group of patients have not been performed (see PRECAUTIONS – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
SUBCUTANEOUS, EPIDURAL
Medical Information
**INDICATIONS** **Surgical anaesthesia (Adults and children over 12 years of age)** - epidural block for surgery including caesarean section. - field block (minor nerve block and infiltration). - major nerve block. **Analgesia (Adults and children over 12 years of age)** - continuous epidural infusion or intermittent bolus epidural administration for analgesia in postoperative pain or labour pain. - field block (minor nerve block and infiltration). - continuous peripheral nerve block infusion or intermittent injections for post operative pain management. There are no safety or efficacy data to support the use of NAROPIN administered epidurally for analgesia for longer than 72 hours. Data for peripheral nerve block administered as a continuous peripheral infusion or intermittent injections support the use for up to 48 hours only. **Analgesia (Children aged 0 – 12 years)** For peri- and post-operative pain management - Caudal epidural block in neonates (> 37 weeks gestation and over 2500 g weight), infants and children up to and including 12 years - Continuous epidural infusion in infants (> 30 days and over 2500 g weight) and children up to and including 12 years There are no safety or efficacy data to support the use of NAROPIN administered epidurally for analgesia for longer than 72 hours.
**CONTRAINDICATIONS** 1. Allergy or hypersensitivity to amide type local anaesthetics. Detection of suspected hypersensitivity by skin testing is of limited value. 2. Intravenous administration. 3. Local anaesthetics are contraindicated for epidural and spinal anaesthesia in patients with uncorrected hypotension. 4. Local anaesthetic techniques must not be used when there is inflammation and/or sepsis in the region of the proposed injection and/or in the presence of septicaemia. 5. Intravenous regional anaesthesia (Bier’s block) as unintentional passage of local anaesthetic into the systemic circulation, despite the use of a tourniquet, may cause systemic toxic reactions. 6. The use of ropivacaine is not recommended for obstetric paracervical block. 7. General contraindications related to epidural anaesthesia, regardless of the local anaesthetic used, should be taken into account.
Pending
xpending
Manufacturer Information
DCH AURIGA SINGAPORE
ASTRAZENECA AB
Active Ingredients
Documents
Package Inserts
Naropin PI.pdf
Approved: February 18, 2019
