- Approval Id
- 3110720b39be3edb
- Drug Name
- ZEPOSIA CAPSULES TREATMENT INITIATION PACK
- Product Name
- ZEPOSIA CAPSULES TREATMENT INITIATION PACK
- Approval Number
- SIN16785P
- Approval Date
- 2023-05-17
- Registrant
- BRISTOL-MYERS SQUIBB (SINGAPORE) PTE. LTD.
- Licence Holder
- BRISTOL-MYERS SQUIBB (SINGAPORE) PTE. LTD.
- Drug Type
- Therapeutic
- Forensic Classification
- Prescription Only
- Dosage Form
- CAPSULE
- Dosage
- **4.2 Posology and method of administration**
Treatment should be initiated under the supervision of a physician experienced in the management of multiple sclerosis (MS).
Posology
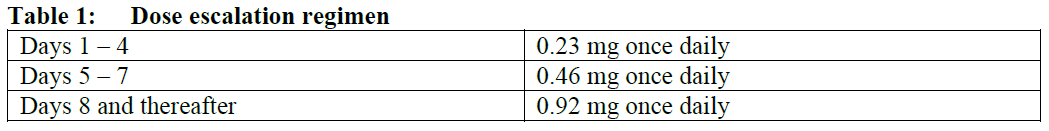
The recommended dose is 0.92 mg ozanimod once daily.
The initial dose escalation regimen of ozanimod from Day 1 to Day 7 is required and shown below in Table 1. Following the 7-day dose escalation, the once daily dose is 0.92 mg, starting on Day 8.
_Re-initiation of therapy following treatment interruption_
The same dose escalation regimen described in Table 1 is recommended when treatment is interrupted for:
- 1 day or more during the first 14 days of treatment.
- more than 7 consecutive days between Day 15 and Day 28 of treatment.
- more than 14 consecutive days after Day 28 of treatment.
If the treatment interruption is of shorter duration than the above, the treatment should be continued with the next dose as planned.
_Special populations_
_Adults over 55 years old and elderly population_
There are limited data available on RRMS patients > 55 years of. No dose adjustment is needed in patients over 55 years of age. Caution should be used in MS patients over 55, given the limited data available and potential for an increased risk of adverse reactions in this population, especially with long-term treatment (see section 5.1 and 5.2 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
_Renal impairment_
No dose adjustment is necessary for patients with renal impairment.
_Hepatic impairment_
Patients with mild or moderate chronic hepatic impairment (Child-Pugh class A or B) are recommended to complete the 7-day dose escalation regimen, and then take 0.92 mg once every other day (see section 5.2 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Ozanimod was not evaluated in patients with severe hepatic impairment. Therefore, patients with severe hepatic impairment (Child-Pugh class C) must not be treated with ozanimod (see sections 4.3 and 5.2 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
_Paediatric population_
The safety and efficacy of Zeposia in children and adolescents aged below 18 years have not yet been established. No data are available.
Method of administration
Oral use.
The capsules can be taken with or without food.
- Route Of Administration
- ORAL
- Indication Info
- **4.1 Therapeutic indications**
Multiple sclerosis
Zeposia is indicated for the treatment of adult patients with relapsing remitting multiple sclerosis (RRMS) with active disease as defined by clinical or imaging features, to decrease the frequency of clinical exacerbations. (see section 5.1 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
- Contraindications
- **4.3 Contraindications**
- Hypersensitivity to the active substance or to any of the excipients listed in section 6.1 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_.
- Immunodeficient state (see section 4.4 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
- Patients who in the last 6 months experienced myocardial infarction (MI), unstable angina, stroke, transient ischaemic attack (TIA), decompensated heart failure requiring hospitalisation or New York Heart Association (NYHA) Class III/IV heart failure.
- Patients with history or presence of second-degree atrioventricular (AV) block Type II or third-degree AV block or sick sinus syndrome unless the patient has a functioning pacemaker.
- Severe active infections, active chronic infections such as hepatitis and tuberculosis (see section 4.4 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
- Active malignancies.
- Severe hepatic impairment (Child-Pugh class C).
- During pregnancy and in women of childbearing potential not using effective contraception (see sections 4.4 and 4.6 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
- Patients who are taking a monoamine oxidase (MAO) inhibitor.
- Atc Code
- L04AA38
- Atc Item Name
- xl 04 aa 38
- Pharma Manufacturer Name
- BRISTOL-MYERS SQUIBB (SINGAPORE) PTE. LTD.
- Company Detail Path
- /organization/148e4d25c144e526/bristol-myers-squibb-singapore-pte-ltd