- Approval Id
- 45ad67a78d483a78
- Drug Name
- TABRECTA FILM-COATED TABLET 150mg
- Product Name
- TABRECTA FILM-COATED TABLET 150mg
- Approval Number
- SIN16350P
- Approval Date
- 2021-10-22
- Registrant
- NOVARTIS (SINGAPORE) PTE LTD
- Licence Holder
- NOVARTIS (SINGAPORE) PTE LTD
- Drug Type
- Therapeutic
- Forensic Classification
- Prescription Only
- Dosage Form
- TABLET, FILM COATED
- Dosage
- **4 Dosage regimen and administration**
**Patient selection**
Patients should be selected for treatment with Tabrecta based on the presence of a MET exon 14 skipping mutation in tumor or plasma specimens using a validated test. If a MET exon 14 skipping mutation is not detected in a plasma specimen, tumor tissue should be tested if feasible.
**Dosage regimen**
**General target population**
The recommended dose of Tabrecta is 400 mg orally twice daily with or without food (see section 11 Clinical pharmacology – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
**Treatment duration**
Treatment should be continued based on individual safety and tolerability and as long as the patient is deriving clinical benefit from therapy.
**Dose modifications for adverse drug reactions**
The recommended dose reduction schedule for the management of adverse drug reactions (ADRs) based on individual safety and tolerability is listed in Table 4-1.
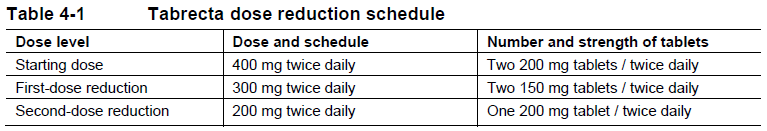
Tabrecta should be permanently discontinued in patients unable to tolerate 200 mg orally twice daily.
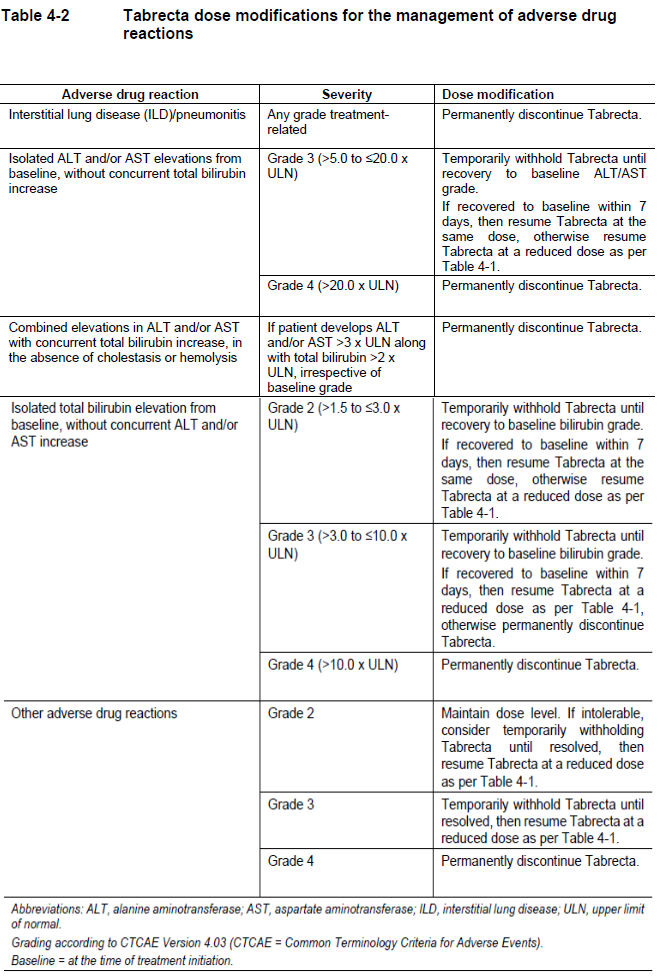
Recommendations for dose modifications of Tabrecta for ADRs are provided in Table 4-2.
**Special populations**
**Renal impairment**
No dose adjustment is necessary in patients with mild or moderate renal impairment based on population pharmacokinetic evaluations; Tabrecta has not been studied in patients with severe renal impairment (see section 11 Clinical pharmacology – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
**Hepatic impairment**
No dose adjustment is necessary in patients with mild, moderate, or severe hepatic impairment (see section 11 Clinical pharmacology – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
**Pediatric patients (below 18 years of age)**
The safety and efficacy of Tabrecta in pediatric patients have not been established.
**Geriatric patients (65 years of age or older)**
No dose adjustment is necessary in patients 65 years of age or older.
**Method of administration**
Tabrecta should be taken orally twice daily with or without food. The tablets should be swallowed whole and should not be broken, chewed, or crushed.
If a dose of Tabrecta is missed or vomiting occurs, the patient should not make up the dose, but take the next dose at the scheduled time.
- Route Of Administration
- ORAL
- Indication Info
- **3 Indications**
Tabrecta is indicated for the treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) with a MET exon 14 skipping mutation.
- Contraindications
- **5 Contraindications**
None.
- Atc Code
- pending
- Atc Item Name
- xpending
- Pharma Manufacturer Name
- NOVARTIS (SINGAPORE) PTE LTD
- Company Detail Path
- /organization/b4e5c80b52263481/novartis-singapore-pte-ltd