- Approval Id
- 4b52a98ad04cef0f
- Drug Name
- ATSWIFT 40 TABLET 40MG
- Product Name
- ATSWIFT 40 TABLET 40MG
- Approval Number
- SIN16064P
- Approval Date
- 2020-12-22
- Registrant
- GOLDPLUS UNIVERSAL PTE LTD
- Licence Holder
- GOLDPLUS UNIVERSAL PTE LTD
- Drug Type
- Therapeutic
- Forensic Classification
- Prescription Only
- Dosage Form
- TABLET, FILM COATED
- Dosage
- **DOSAGE AND ADMINISTRATION:**
**Posology**
The patient should be placed on a standard cholesterol-lowering diet before receiving Atorvastatin Tablets and should continue on this diet during treatment with Atorvastatin Tablets. The dose should be individualised according to baseline LDL-C levels, the goal of therapy, and patient response.
The usual starting dose is 10 mg once a day. Adjustment of dose should be made at intervals of 4 weeks or more. The maximum dose is 80 mg once a day.
**Primary hypercholesterolaemia and combined (mixed) hyperlipidaemia**
The majority of patients are controlled with Atorvastatin Tablets 10 mg once a day. A therapeutic response is evident within 2 weeks, and the maximum therapeutic response is usually achieved within 4 weeks. The response is maintained during chronic therapy.
**Homozygous familial hypercholesterolaemia**
In a compassionate-use study of patients with homozygous familial hypercholesterolemia, most patients responded to 80 mg of atorvastatin with a greater than 15% reduction in LDL-C (18%–45%).
**Prevention of cardiovascular disease**
For primary prevention, the recommended dose is 10 mg once daily. For secondary prevention, optimal dosing may range from 10 mg to 80 mg atorvastatin once daily, to be given at the discretion of the prescriber, taking into account the expected benefit and safety considerations relevant to the patient to be treated.
**Renal impairment**
No adjustment of dose is required.
**Hepatic impairment**
Atorvastatin Tablets should be used with caution in patients with hepatic impairment. Atorvastatin Tablets is contraindicated in patients with active liver disease.
**Use in the elderly**
Efficacy and safety in patients older than 70 using recommended doses are similar to those seen in the general population.
**Paediatric use**
Hypercholesterolaemia:
Paediatric use should only be carried out by physicians experienced in the treatment of paediatric hyperlipidaemia and patients should be re-evaluated on a regular basis to assess progress.
For patients aged 10 years and above, the recommended starting dose of atorvastatin is 10 mg per day with titration up to 20 mg per day. Titration should be conducted according to the individual response and tolerability in paediatric patients. Safety information for paediatric patients treated with doses above 20 mg, corresponding to about 0.5 mg/kg, is limited.
There is limited experience in children between 6–10 years of age. Atorvastatin is not indicated in the treatment of patients below the age of 10 years.
Treatment experience in a pediatric population is limited to doses of atorvastatin up to 80 mg/day for one year in 8 patients with homozygous FH. No clinical or biochemical abnormalities were reported in these patients.
**Use in Combination with Other Medicinal Compounds**
In cases where co-administration of atorvastatin with cyclosporine, telaprevir, or the combination tipranavir/ritonavir is necessary, the dose of atorvastatin should not exceed 10 mg. Dose of atorvastatin should not exceed 20 mg/day with concomitant use with elbasvir/grazoprevir.
Pharmacokinetic drug interactions that result in increased systemic concentration of atorvastatin have been noted with HIV protease inhibitors (lopinavir plus ritonavir, saquinavir plus ritonavir, darunavir plus ritonavir, fosamprenavir, fosamprenavir plus ritonavir and nelfinavir), Hepatitis C protease inhibitor (boceprevir), clarithromycin and itraconazole. Caution should be used when co-prescribing atorvastatin and appropriate clinical assessment is recommended to ensure that the lowest dose necessary of atorvastatin is employed.
**THE FOLLOWING TREATMENT GUIDELINES MAY BE USED TO ESTABLISH TREATMENT GOALS**
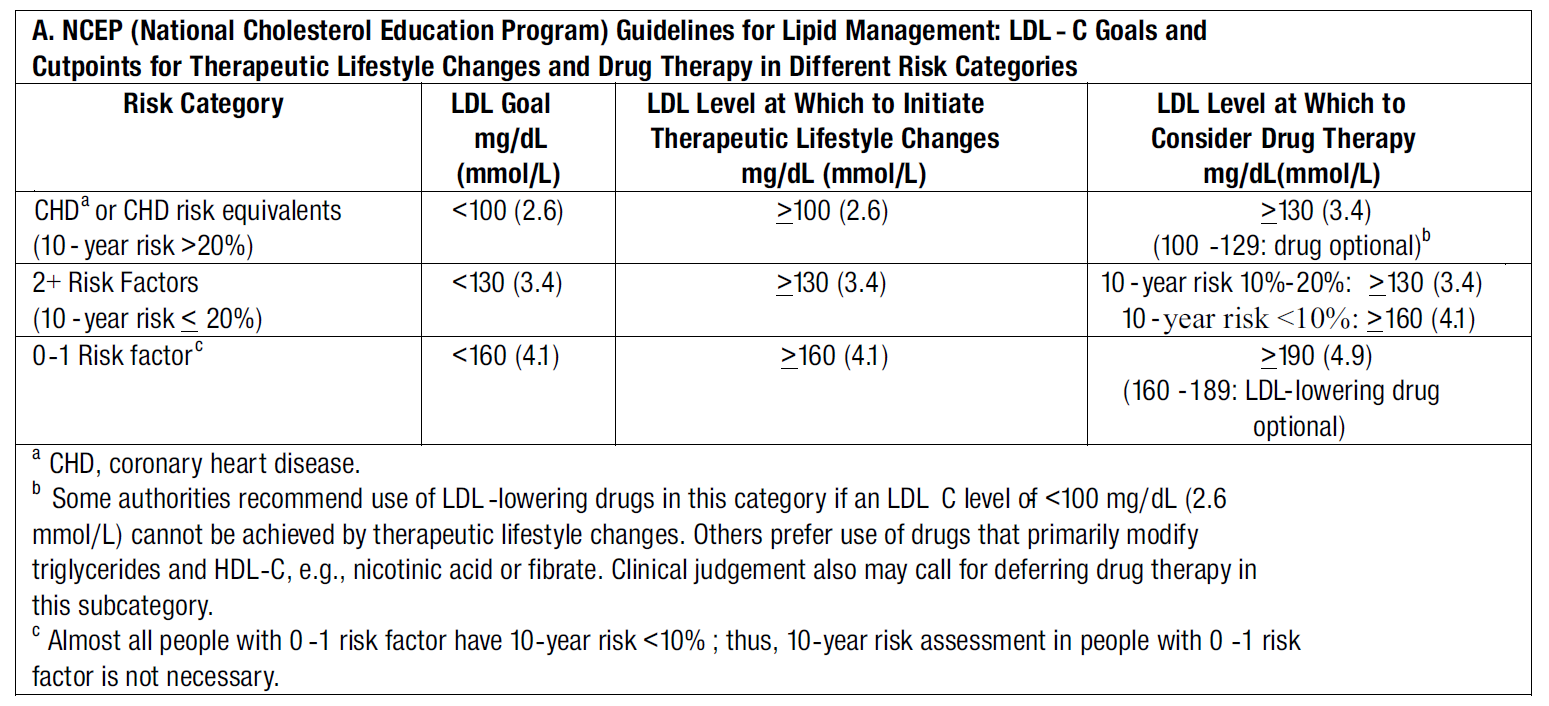
After the LDL-C goal has been achieved, if the TG is still ≥200 mg/dL (2.2 mmol/L), non-HDL-C (total-C minus HDL-C) becomes a secondary target of therapy. Non-HDL-C goals are set 30 mg/dL (0.8 mmol/L) higher than LDL-C goals for each risk category.
**Method of administration**
Atorvastatin Tablets is for oral administration. Each daily dose of atorvastatin is given all at once and may be given at any time of day with or without food.
- Route Of Administration
- ORAL
- Indication Info
- **INDICATIONS AND USAGE:**
**Hypercholesterolaemia**
Atorvastatin Tablets is indicated as an adjunct to diet for reduction of elevated total cholesterol (total-C), LDL-cholesterol (LDL-C), apolipoprotein B, and triglycerides in adults, adolescents and children aged 10 years or older with primary hypercholesterolaemia including familial hypercholesterolaemia (heterozygous variant) or combined (mixed) hyperlipidaemia (Corresponding to Types IIa and IIb of the Fredrickson classification) when response to diet and other nonpharmacological measures is inadequate.
Atorvastatin Tablets is also indicated to reduce total-C and LDL-C in adults with homozygous familial hypercholesterolaemia as an adjunct to other lipid-lowering treatments (e.g. LDL apheresis) or if such treatments are unavailable.
**Prevention of cardiovascular disease**
Atorvastatin Tablets is indicated to reduce the risk of myocardial infarction in adult hypertensive patients without clinically evident coronary heart disease, but with at least three additional risk factors for coronary heart disease such as age ≥55 years, male sex, smoking, left ventricular hypertrophy, other specified abnormalities on ECG, microalbuminia or proteinuria, ratio of plasma total cholesterol to HDL-cholesterol ≥6, or premature family history of coronary heart disease.
In adults with type 2 diabetes and without clinically evident coronary heart disease, but with multiple risk factors for coronary heart disease such as retinopathy, albuminuria, smoking or hypertension, Atorvastatin Tablet is indicated to:
- Reduce the risk of myocardial infarction
- Reduce the risk of stroke
In adults with clinically evident coronary heart disease, atorvastatin is indicated to:
- Reduce the risk of non-fatal myocardial infarction
- Reduce the risk of fatal and non-fatal stroke
- Reduce the risk for revascularization procedures
- Reduce the risk of hospitalization for CHF
- Reduce the risk of angina
- Contraindications
- **CONTRAINDICATIONS:**
Atorvastatin Tablets is contraindicated in patients:
- with hypersensitivity to the active substance or to any of the excipients of this medicinal product.
- with active liver disease or unexplained persistent elevations of serum transaminases exceeding 3 times the upper limit of normal.
- during pregnancy, while breast-feeding and in women of child-bearing potential not using appropriate contraceptive measures.
- Atorvastatin is contraindicated in patients who are concomitantly treated with glecaprevir/pibrentasvir.
- Atc Code
- C10AA05
- Atc Item Name
- atorvastatin
- Pharma Manufacturer Name
- GOLDPLUS UNIVERSAL PTE LTD
- Company Detail Path
- /organization/7cb5b69d6eb36564/goldplus-universal-pte-ltd