- Approval Id
- 5835f84749f930d7
- Drug Name
- AVOFER INJECTION 100MG/5ML
- Product Name
- AVOFER INJECTION 100MG/5ML
- Approval Number
- SIN16513P
- Approval Date
- 2022-06-16
- Registrant
- APEX PHARMA MARKETING PTE. LTD.
- Licence Holder
- APEX PHARMA MARKETING PTE. LTD.
- Drug Type
- Therapeutic
- Forensic Classification
- Prescription Only
- Dosage Form
- INJECTION
- Dosage
- **POSOLOGY AND METHOD OF ADMINISTRATION**
**Calculation of Dosage**
Monitor carefully patients for signs and symptoms of hypersensitivity reactions during and following each administration of Avofer.
Avofer should only be administered when staff trained to evaluate and manage anaphylactic reactions is immediately available, in an environment where full resuscitation facilities can be assured. The patient should be observed for adverse effects for at least 30 minutes following each Avofer injection.
Posology
The cumulative dose of Avofer must be calculated for each patient individually and must not be exceeded.
_Calculation of dosage_
The total cumulative dose of Avofer, equivalent to the total iron deficit (mg), is determined by the haemoglobin level (Hb) and body weight (BW). The dose of Avofer must be individually calculated for each patient according to the total iron deficit calculated with the following Ganzoni formula, for example:
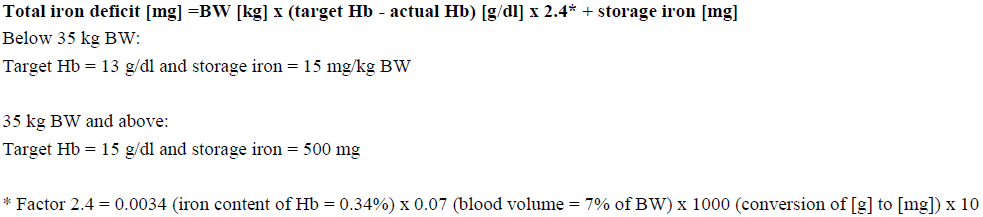
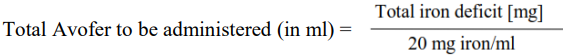
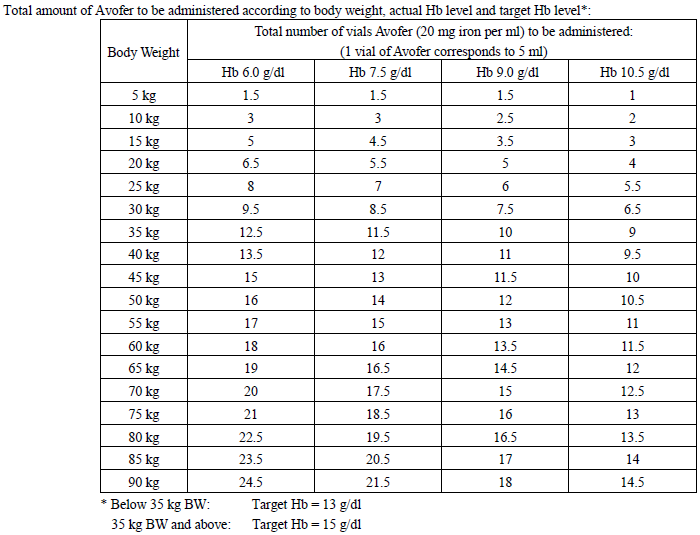
To convert Hb (mM) to Hb (g/dl), multiply the former by 1.6.
If the total necessary dose exceeds the maximum allowed single dose, then the administration must be divided. If no response of the haematological parameters is observed after 1 to 2 weeks the original diagnosis should be reconsidered.
_Calculation of dosage for iron replacement secondary to blood loss and to support autologous blood donation_
The required Avofer dose to compensate for the iron deficit may be calculated according the following formulas:
If the quantity of blood lost is known: The administration of 200 mg iron (10 ml of Avofer) should result in an increase in Hb approximately equivalent to 1 unit blood (400 ml with Hb = 15 g/dl).
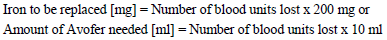
If the Hb level is less than desired: Formula assumes that the storage iron does not need to be restored. Iron to be replaced \[mg\] = BW \[kg\] x 2.4 x (target Hb – actual Hb) \[g/dl\]
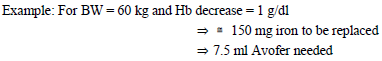
For the maximum tolerated single and weekly dose, see “Normal posology” and “Maximum tolerated single and weekly doses”.
_Normal Posology_
_Adult:_
5–10ml of Avofer (100–200 mg iron) 1 to 3 times a week.
For administration time and dilution ratio see “Method of administration”.
_Paediatric population_
There is moderate amount of data in children under study conditions. If there is a clinical need, it is recommended not to exceed 0.15 ml of Avofer (3 mg iron) per kg body weight not more than three times per week.
For administration time and dilution ratio see “Method of administration”.
_Maximum tolerated single and weekly doses_
_Adults_
As an injection, maximum tolerated dose per day given not more than 3 times per week:
➢ 10 ml of Avofer (200 mg iron) injected over at least 10 minutes
As an infusion, maximum tolerated dose per day given not more than once per week:
➢ Patients above 70 kg body weight: 500 mg iron (25 ml of Avofer) over at least 3 ½ hours
➢ Patients of 70 kg body weight and below: 7 mg iron/kg body weight over at least 3 ½ hours
The infusion times given in “Method of administration” should be strictly adhered to, even if the patient does not receive the maximum tolerated single dose.
**Method of Administration**
Avofer must only be administered by the intravenous route. This may be by drip infusion, slow injection or directly into the venous line of the dialysis machine.
1. Intravenous drip infusion
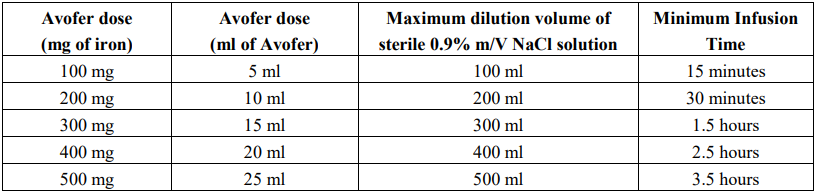
Avofer must only be diluted in sterile 0.9% m/V sodium chloride (NaCl) solution. Dilution must take place immediately prior to infusion and the solution should be administered as follows:
2. Intravenous Injection
Avofer may be administered by slow intravenous injection at a rate of 1 ml undiluted solution per minute and not exceeding 10 ml (200 mg iron) per injection.
3. Injection into venous line of dialysis machine
Avofer may be administered during haemodialysis session directly into the venous line of the dialysis machine under the same conditions as for intravenous injection.
- Route Of Administration
- INTRAVENOUS
- Indication Info
- **INDICATIONS**
Avofer is indicated in the treatment of iron deficiency in the following indications:
➢ Where there is a clinical need for a rapid iron supply
➢ In patients who cannot tolerate oral iron therapy or who are non-compliant
➢ Where oral iron preparations are ineffective (e.g., in active inflammatory bowel disease).
Avofer should only be administered where the indication is confirmed by appropriate investigations.
- Contraindications
- **CONTRAINDICATIONS**
The use of Avofer is contraindicated in the following conditions:
- Hypersensitivity to iron sucrose, Avofer or to any of its excipients listed in section _List of excipients_ – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_
- Anaemia not caused by iron deficiency
- Evidence of iron overload or disturbances in utilisation of iron
- Pregnancy first Trimester
- Atc Code
- B03AC
- Atc Item Name
- 非肠道用药的三价铁制剂
- Pharma Manufacturer Name
- APEX PHARMA MARKETING PTE. LTD.
- Company Detail Path
- /organization/f3877d86bd1d513a/apex-pharma-marketing-pte-ltd