Regulatory Information
HSA regulatory responsibility and product classification details
Regulatory Responsibility
Product Classification
Formulation Information
INJECTION
**DOSAGE AND ADMINISTRATION** ZADAXIN is intended for subcutaneous injection and should not be given intravenously. It should be reconstituted with 1.0 ml of the diluent provided, which consists of 1.0 ml Sterile Water for Injection, immediately prior to use. At the discretion of the physician, the patient may be taught to self-administer the medication. **Chronic Hepatitis B:** The recommended dose of ZADAXIN for chronic hepatitis B when used as a monotherapy or in combination with interferon (at the labeled dose and schedule for interferon) is 1.6 mg (900 mcg/m2) administered subcutaneously twice a week for 6 to 12 months. Patients weighing less than 40 kg should receive a ZADAXIN dose of 40 mcg/kg. **Chronic Hepatitis C:** The recommended dose of ZADAXIN for chronic hepatitis C when used in combination with interferon (at the labeled dose and schedule for interferon) is 1.6 mg (900 mcg/m2) administered subcutaneously twice a week for 12 months. Patients weighing less than 40 kg should receive a ZADAXIN dose of 40 mcg/kg.
SUBCUTANEOUS
Medical Information
**INDICATIONS AND USAGE** **Chronic Hepatitis B:** ZADAXIN thymosin alpha 1 (thymalfasin) is indicated as a monotherapy or combination therapy with interferon for the treatment of chronic hepatitis B. Pooled analysis of 3 randomized controlled trials comprising 223 patients was performed. Thymosin alpha 1 was administered twice weekly for 6 months. Follow-up assessments were performed at 12 months after completion of treatment (see table). In multiple studies, ZADAXIN was shown to have a delayed therapeutic response 12 months or longer after completion of therapy. A transient increase in ALT to more than twice baseline value (flare) can occur during ZADAXIN therapy. When ALT flare occurs, ZADAXIN should generally be continued unless signs and symptoms of liver failure are observed. 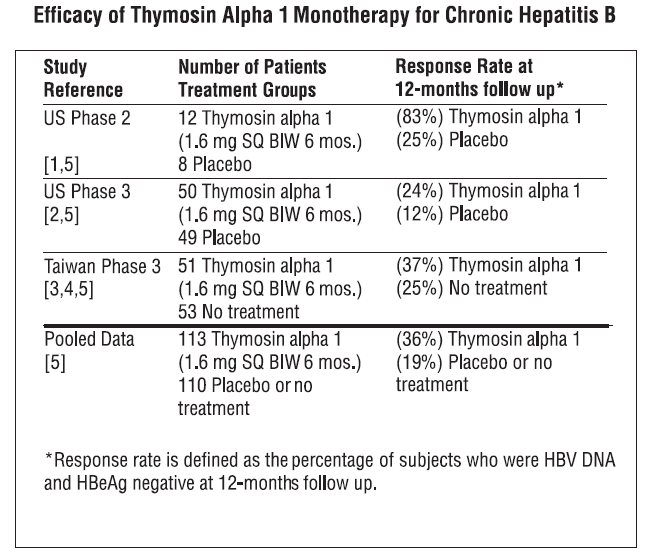 **Chronic Hepatitis C:** ZADAXIN thymosin alpha 1 (thymalfasin) is indicated as a combination therapy with interferon for the treatment of chronic hepatitis C. Pooled analysis of 2 randomized controlled trials and 1 historical controlled trial comprising 121 ZADAXIN plus interferon, or interferon treated patients, was performed. Thymosin alpha 1 was administered at least twice weekly for 6 to 12 months and interferon was administered up to three times weekly for 6 to 12 months. Follow-up assessments were performed upon completion of treatment and at 6 months after completion of treatment (see table). Pooled intent-to-treat analysis demonstrated sustained biochemical (ALT) response, defined as normal ALT 6 to 12 months after completion of treatment, observed in 22.4% of patients treated with combination therapy compared to 9.3% with interferon alone. 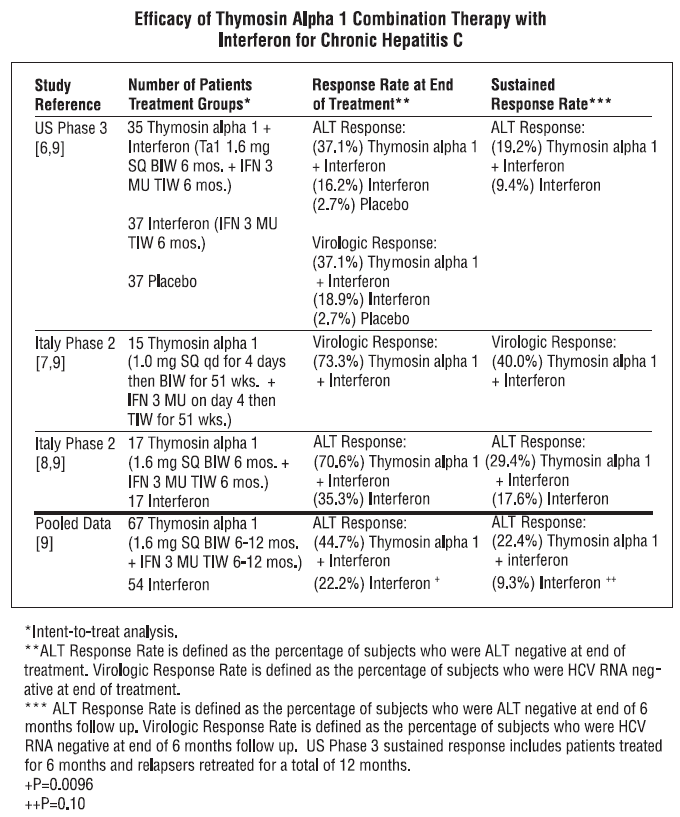
**CONTRAINDICATIONS** ZADAXIN is contraindicated in patients with a history of hypersensitivity to thymosin alpha 1 or any component of the injection. Because ZADAXIN therapy appears to work by enhancing the immune system, it should be considered contraindicated in patients who are being deliberately immunosuppressed, such as organ transplant patients, unless the potential benefits of the therapy clearly outweigh the potential risks.
L03AX
其它免疫增强剂
Manufacturer Information
ZUELLIG PHARMA PTE. LTD.
PATHEON ITALIA S.p.A
ALFA WASSERMANN S.P.A
Active Ingredients
Documents
Package Inserts
PI-Singapore_C4000-00 - Proposed PI.pdf
Approved: February 14, 2017
