- Approval Id
- 636df41b60220a89
- Drug Name
- MEKTOVI FILM-COATED TABLET 15MG
- Product Name
- MEKTOVI FILM-COATED TABLET 15MG
- Approval Number
- SIN16826P
- Approval Date
- 2023-07-19
- Registrant
- PIERRE FABRE SINGAPORE PTE LTD
- Licence Holder
- PIERRE FABRE SINGAPORE PTE LTD
- Drug Type
- Therapeutic
- Forensic Classification
- Prescription Only
- Dosage Form
- TABLET, FILM COATED
- Dosage
- **4.2. DOSE AND METHOD OF ADMINISTRATION**
Treatment with binimetinib in combination with encorafenib should only be initiated and supervised by a physician experienced in the use of anti-cancer medicines.
**Dosage**
Patients treated with binimetinib in combination with encorafenib must have their BRAF V600 mutant melanoma status confirmed by a validated test conducted by an experienced laboratory (see 5.1 Clinical Trials – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
The recommended dose of binimetinib is 45 mg (three 15 mg tablets) twice daily (corresponding to a total dose of 90 mg), approximately 12 hours apart, when used in combination with encorafenib.
**Administration**
Binimetinib tablets should be swallowed whole with water, with or without food.
**Duration of treatment**
Treatment should continue until the patient no longer derives benefit or unacceptable toxicity develops.
**Missed dose**
If a dose of binimetinib is missed, it should not be taken if it is less than 6 hours until the next dose is due.
**Vomiting after administration**
If a patient vomits after administration of binimetinib, the patient should not take the dose again. The patient should take the next scheduled dose.
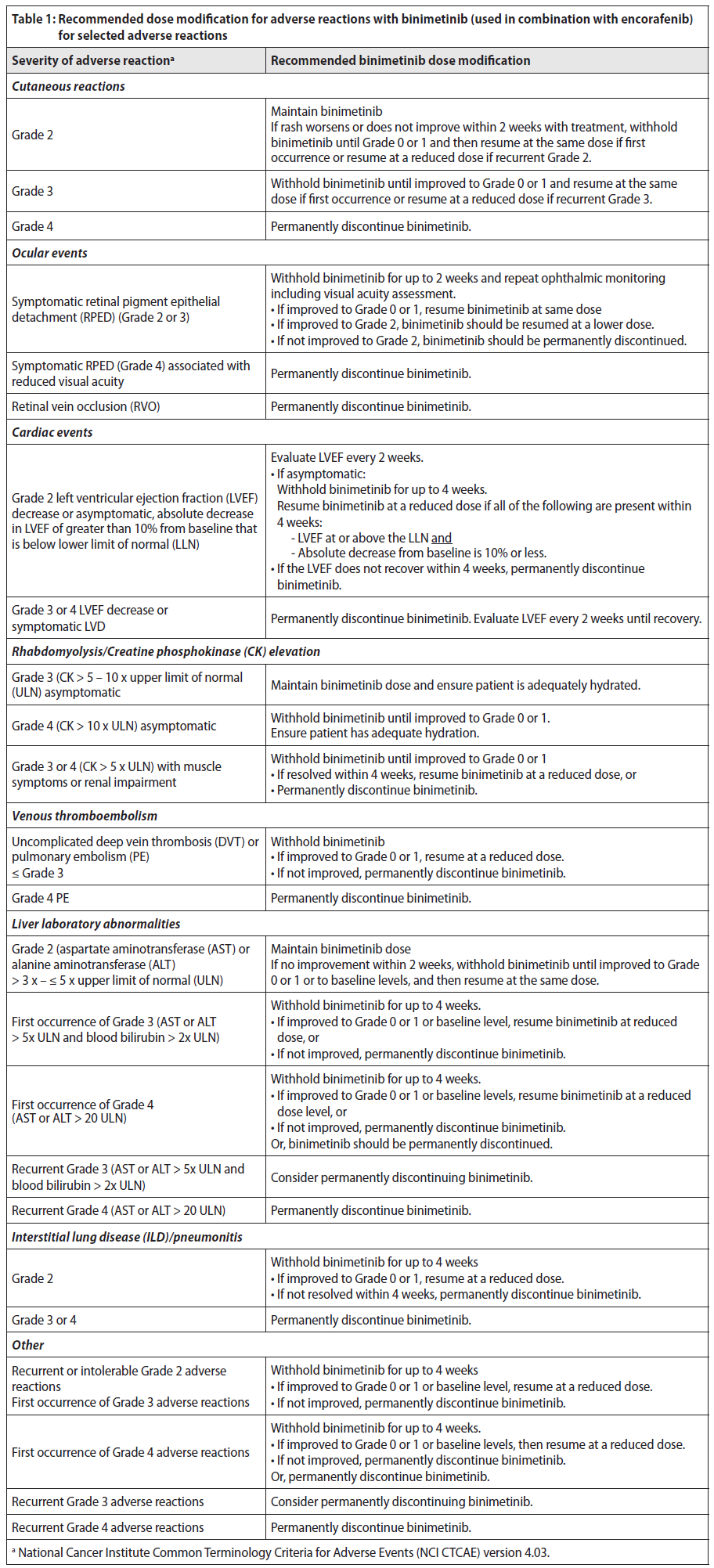
**Dose modification**
The management of adverse reactions may require dose reduction, temporary interruption or treatment discontinuation (see below and Table 1). The decision on whether to modify the dose of binimetinib should be based on the prescriber’s assessment of individual patient safety and tolerance.
The recommended reduced dose of binimetinib is 30 mg twice daily. Dose reduction below 30 mg twice daily is not recommended. Therapy should be discontinued if the patient is not able to tolerate 30 mg orally twice daily.
If the adverse reaction that resulted in a dose reduction is under effective management, re-escalation to 45 mg twice daily may be considered. Dose-re-escalation to 45 mg twice daily is not recommended if the dose reduction is due to left ventricular dysfunction (LVD) or any Grade 4 toxicity.
If treatment-related toxicities occur when binimetinib is used in combination with encorafenib, then both treatments should be simultaneously dose reduced, interrupted or discontinued. Exceptions where dose modifications are necessary for encorafenib only (adverse reactions primarily related to encorafenib) are: palmar-plantar erythrodysaesthesia syndrome (PPES), uveitis including iritis and iridocyclitis, and QTc prolongation.
If one of these toxicities occurs, see section 4.2. _Dose and Method of Administration_ of encorafenib PI for dose modification instructions for encorafenib.
If binimetinib is temporarily interrupted, reduce encorafenib to 300 mg once daily during the time of binimetinib dose interruption (see Table 1) as encorafenib is not well-tolerated at the dose of 450 mg as a single agent. If binimetinib is permanently discontinued, encorafenib may be continued (at the reduced dose of 300 mg) depending on the individual clinical benefit.
If encorafenib is temporarily interrupted (see section 4.2 _Dose and Method of Administration_ of encorafenib PI), interrupt binimetinib. If encorafenib is permanently discontinued, then discontinue binimetinib.
Dose modification recommendations in case of adverse reactions are presented in Table 1. For information on the dosage and recommended dose modifications of encorafenib, refer to the encorafenib PI, section 4.2 _Dose and Method of Administration_.
**Table 1: Recommended dose modification for adverse reactions with binimetinib (used in combination with encorafenib) for selected adverse reactions**
**Hepatic impairment**
No dose adjustment is required in patients with mild hepatic impairment (Child-Pugh A). As encorafenib is not recommended in patients with moderate (Child Pugh B) or severe hepatic impairment (Child-Pugh C), administration of binimetinib is not recommended in these patients (see section 4.2 _Dose and method of administration_ of encorafenib PI).
**Renal impairment**
No dose adjustment is required for patients with renal impairment (see section 5.2 _Pharmacokinetic properties_ – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
**Elderly patients (65 years and older)**
No dose adjustment is required for elderly patients (see section 5.2 _Pharmacokinetic properties_ – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
**Children and adolescents (< 18 years)**
The safety and efficacy of binimetinib have not been established in patients below the age of 18 years. There are no data available.
- Route Of Administration
- ORAL
- Indication Info
- **4.1 THERAPEUTIC INDICATIONS**
Binimetinib in combination with encorafenib is indicated for the treatment of adult patients with unresectable or metastatic melanoma with a BRAF V600E or V600K mutation, as detected by a validated test.
- Contraindications
- **4.3. CONTRAINDICATIONS**
Hypersensitivity to the active substance binimetinib or to any of the excipients (see section 6.1 _List of excipients_ – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
- Atc Code
- L01EE03
- Atc Item Name
- binimetinib
- Pharma Manufacturer Name
- PIERRE FABRE SINGAPORE PTE. LTD.
- Company Detail Path
- /organization/96132848e73a4dd2/pierre-fabre-singapore-pte-ltd