- Approval Id
- 67790ce79b9b45b1
- Drug Name
- SORAFENIB ALVOGEN FILM COATED TABLETS 200MG
- Product Name
- SORAFENIB ALVOGEN FILM COATED TABLETS 200MG
- Approval Number
- SIN16442P
- Approval Date
- 2022-03-09
- Registrant
- LOTUS INTERNATIONAL PTE. LTD.
- Licence Holder
- LOTUS INTERNATIONAL PTE. LTD.
- Drug Type
- Therapeutic
- Forensic Classification
- Prescription Only
- Dosage Form
- TABLET, FILM COATED
- Dosage
- **4.2 Posology and method of administration**
Sorafenib treatment should be supervised by a physician experienced in the use of anticancer therapies.
Posology
_Recommended dose_
The recommended daily dose of sorafenib is 400 mg (2 x 200 mg tablets) taken twice a day, either without food or together with a moderate fat meal.
_Method of administration_
For oral use. To be swallowed with a glass of water.
_Duration of treatment_
Treatment should be continued until the patient is no longer clinically benefiting from therapy or until unacceptable toxicity occurs.
Dose titration, dose adjustment, special monitoring advice
_Dose Reduction for Hepatocellular Carcinoma and advanced Renal cell Carcinoma_
Management of suspected adverse drug reactions may require temporary interruption and/or dose reduction of sorafenib therapy. When dose reduction is necessary during the treatment of hepatocellular carcinoma (HCC) and advanced renal cell carcinoma (RCC), the sorafenib dose should be reduced to two tablets of 200 mg once daily (see section 4.4 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
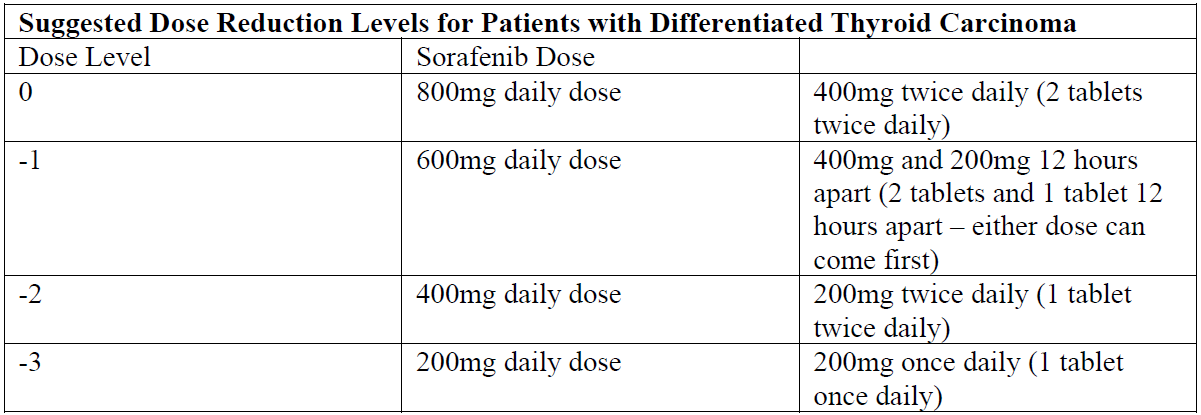
Dose Reduction for Differentiated Thyroid Carcinoma
Management of suspected adverse drug reactions may require temporary interruption and/or dose reduction of sorafenib therapy.
When dose reduction is necessary during the treatment of differentiated thyroid carcinoma, the sorafenib dose should be reduced to 600mg daily in divided doses (two tablets of 200mg and one tablet of 200mg twelve hours apart).
If additional dose reduction is necessary, sorafenib may be reduced to one tablet of 200mg twice daily, followed by one tablet of 200mg once daily. After improvement of non-hematological adverse reactions, the dose of sorafenib may be increased.
_Paediatric population_
The safety and efficacy of sorafenib in children and adolescents aged < 18 years have not yet been established. No data are available.
_Elderly population_
No dose adjustment is required on the basis of patient age (above 65 years), gender, or body weight.
_Renal impairment_
No dose adjustment is required in patients with mild, moderate, or severe renal impairment not requiring dialysis. Sorafenib has not been studied in patients undergoing dialysis (see section 5.2 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Monitoring of fluid balance and electrolytes in patients at risk of renal dysfunction is advised.
_Hepatic impairment_
No dose adjustment is required in patients with Child Pugh A or B (mild to moderate) hepatic impairment. No data is available on patients with Child Pugh C (severe) hepatic impairment (see sections 4.4 and 5.2 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Method of administration
For oral use.
It is recommended that sorafenib should be administered without food or with a low or moderate fat meal. If the patient intends to have a high-fat meal, sorafenib tablets should be taken at least 1 hour before or 2 hours after the meal. The tablets should be swallowed with a glass of water.
- Route Of Administration
- ORAL
- Indication Info
- **4.1 Therapeutic indications**
Hepatocellular carcinoma
Sorafenib is indicated for the treatment of patients with unresectable hepatocellular carcinoma (HCC) (see section 5.1 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Renal cell carcinoma
Sorafenib is indicated for the treatment of patients with advanced renal cell carcinoma who have failed prior systemic therapy or are considered unsuitable for such therapy.
Differentiated thyroid carcinoma
Sorafenib is indicated for the treatment of patients with locally advanced or metastatic, progressive, differentiated thyroid carcinoma refractory to radioactive iodine treatment.
- Contraindications
- **4.3 Contraindications**
Hypersensitivity to the active substance or to any of the excipients listed in section 6.1 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_.
- Atc Code
- L01EX02
- Atc Item Name
- sorafenib
- Pharma Manufacturer Name
- LOTUS INTERNATIONAL PTE. LTD.
- Company Detail Path
- /organization/3735e40f596a2f70/lotus-international-pte-ltd