Regulatory Information
HSA regulatory responsibility and product classification details
Regulatory Responsibility
Product Classification
Formulation Information
INJECTION, SOLUTION
**DOSAGE AND ADMINISTRATION** **Recommended Dosing** Chemotherapy-Induced Nausea and Vomiting 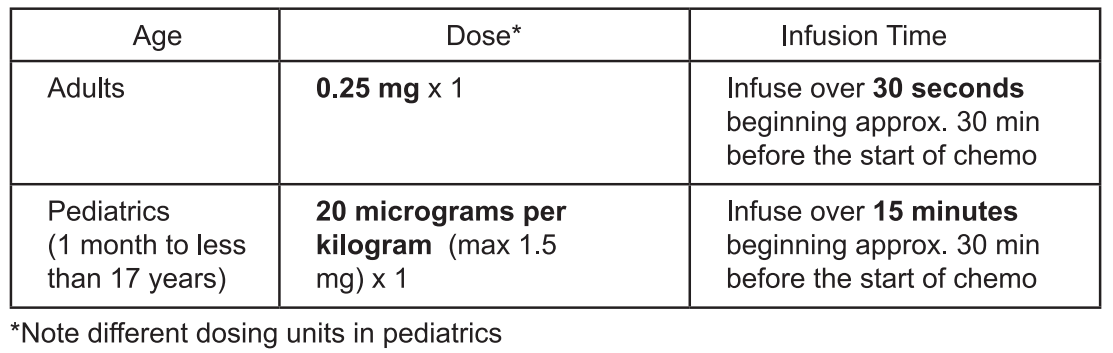 The safety and efficacy of Aloxi in children aged less than 1 month have not been established. No data are available. There is limited data on the use of Aloxi in the prevention of nausea and vomiting in children under 2 years of age. Postoperative Nausea and Vomiting Dosage for Adults – a single 0.075 mg I.V. dose administered over 10 seconds immediately before the induction of anesthesia. **Instructions for I.V. Administration** _**Aloxi**_ ® is supplied ready for intravenous administration at a concentration of 0.05 mg/mL (50 mcg/mL). _**Aloxi**_ ® should not be mixed with other drugs. Flush the infusion line with normal saline before and after administration of _**Aloxi**_ ®. Parenteral drug products should be inspected visually for particulate matter and discoloration before administration, whenever solution and container permit.
INTRAVENOUS
Medical Information
**INDICATIONS AND USAGE** **Chemotherapy-Induced Nausea and Vomiting in Adults** _**Aloxi**_ ® is indicated for: - Moderately emetogenic cancer chemotherapy -- prevention of acute and delayed nausea and vomiting associated with initial and repeat courses - Highly emetogenic cancer chemotherapy -- prevention of acute nausea and vomiting associated with initial and repeat courses **Chemotherapy-Induced Nausea and Vomiting in Pediatric Patients Aged 1 month to Less than 17 Years** _**Aloxi**_ ® is indicated for: - Prevention of acute nausea and vomiting associated with initial and repeat courses of moderately or highly emetogenic cancer chemotherapy **Postoperative Nausea and Vomiting in Adults** _**Aloxi**_ ® is indicated for: - Prevention of postoperative nausea and vomiting (PONV) for up to 24 hours following surgery. Efficacy beyond 24 hours has not been demonstrated. As with other antiemetics, routine prophylaxis is not recommended in patients in whom there is little expectation that nausea and/or vomiting will occur postoperatively. In patients where nausea and vomiting must be avoided during the postoperative period, _**Aloxi**_ ® is recommended even where the incidence of postoperative nausea and/or vomiting is low.
**CONTRAINDICATIONS** _**Aloxi**_ ® is contraindicated in patients known to have hypersensitivity to the drug or any of its components. \[ _see Adverse Reactions_ – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_\]
A04AA05
palonosetron
Manufacturer Information
juniper healthcare pte ltd
FAREVA PAU 1
Active Ingredients
Documents
Package Inserts
0201203598 SGP Juniper Aloxi PONV PIL Proof 3 28.01.22_JN_31012022 Clean.pdf
Approved: March 18, 2022
