Regulatory Information
HSA regulatory responsibility and product classification details
Regulatory Responsibility
Product Classification
Formulation Information
TABLET, FILM COATED
**4.2 Posology and method of administration** **Posology** **Adults** - Monotherapy Adults and adolescents from 16 years of age The recommended starting dose is 250 mg twice daily which should be increased to an initial therapeutic dose of 500 mg twice daily after two weeks. The dose can be further increased by 250 mg twice daily every two weeks depending upon the clinical response. The maximum dose is 1500 mg twice daily. - Add-on therapy Adults (≥18 years) and adolescents (12 to 17 years) of 50 kg or more The initial therapeutic dose is 500 mg twice daily. This dose can be started on the first day of treatment. Depending upon the clinical response and tolerance, the daily dose can be increased up to 1,500 mg twice daily. Dose changes can be made in 500 mg twice daily increases or decreases every two to four weeks. Special populations Elderly Adjustment of the dose is recommended in elderly patients with compromised renal function. Renal impairment The daily dose must be individualised according to renal function (see section 4.4 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). For adult patients, refer to the following table and adjust the dose as indicated. To use this dosing table, an estimate of the patient’s creatinine clearance (CLcr) in ml/min is needed. The CLcr in ml/min may be estimated from serum creatinine (mg/dl) determination, for adults and adolescents weighing 50 kg or more, using the following formula: 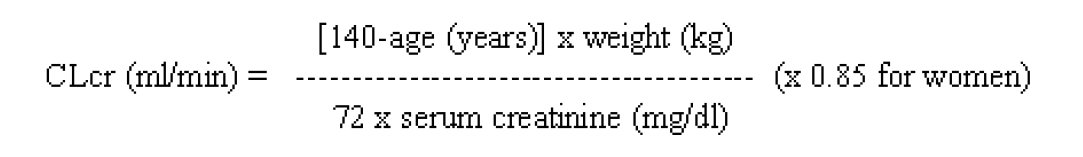 Then CLcr is adjusted for body surface area (BSA) as follows: 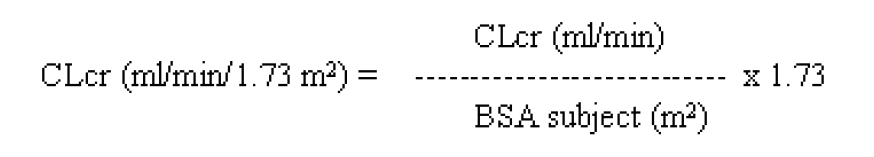 Dosing adjustment for adult and adolescent patients weighing more than 50 kg with impaired renal function  For children with renal impairment, levetiracetam dose needs to be adjusted based on the renal function as levetiracetam clearance is related to renal function. This recommendation is based on a study in adult renally impaired patients. Hepatic impairment No dose adjustment is needed in patients with mild to moderate hepatic impairment. In patients with severe hepatic impairment, the creatinine clearance may underestimate the renal insufficiency. Therefore a 50% reduction of the daily maintenance dose is recommended when the creatinine clearance is <60 ml/min/1.73m2 (see section 4.4 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). **Children** The physician should prescribe the most appropriate pharmaceutical form and strength according to age, weight and dose. The tablet formulation is not adapted for use in infants and children under the age of 6 years. Levetiracetam oral solution is the preferred formulation for use in this population. In addition, the available dose strengths of the tablets are not appropriate for initial treatment in children weighing less than 25 kg, for patients unable to swallow tablets or for the administration of doses below 250 mg. In all of the above cases, an oral solution should be used. Monotherapy The safety and efficacy of levetiracetam in children and adolescents below 16 years as monotherapy treatment have not been established. There are no data available. Add-on therapy for Children (4 to 11 years) and Adolescents (12 to 17 years) weighing less than 50 kg. The initial therapeutic dose is 10 mg/kg twice daily. Depending upon the clinical response and tolerability, the dose can be increased up to 30 mg/kg twice daily. Dose changes should not exceed increases or decreases of 10 mg/kg twice daily every two weeks. The lowest effective dose should be used. Dose in children 50 kg or greater is the same as in adults. Dose recommendations for children and adolescents: 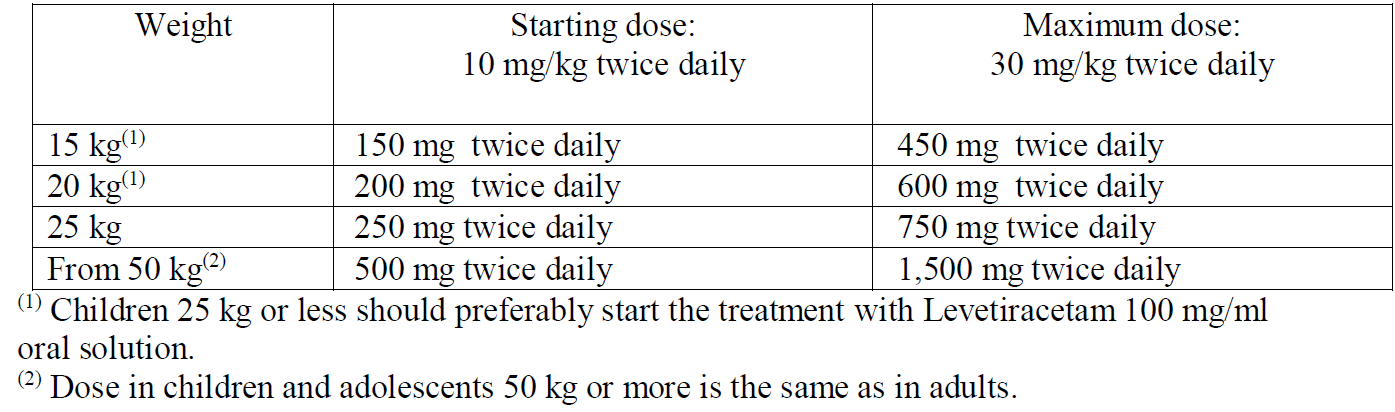 Infants and children less than 4 years There are insufficient data to recommend the use of levetiracetam in children under 4 years of age. **Method of administration** The film-coated tablets must be taken orally, swallowed with a sufficient quantity of liquid and may be taken with or without food. The daily dose is administered in two equally divided doses.
ORAL
Medical Information
**4.1 Therapeutic indications** Levetiracetam is indicated as monotherapy in the treatment of partial onset seizures with or without secondary generalisation in adults and adolescents from 16 years of age with newly diagnosed epilepsy. Levetiracetam is indicated as adjunctive therapy in the treatment of: - partial onset seizures with or without secondary generalisation in adults, adolescents and children from 4 years of age with epilepsy. - myoclonic seizures in adults and adolescents from 12 years of age with Juvenile Myoclonic Epilepsy. - primary generalised tonic-clonic seizures in adults and adolescents from 12 years of age with Idiopathic Generalised Epilepsy.
**4.3 Contraindications** Hypersensitivity to levetiracetam or other pyrrolidone derivatives or to any of the excipients listed in section 6.1 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_.
N03AX14
levetiracetam
Manufacturer Information
SANDOZ SINGAPORE PTE. LTD.
Lek Pharmaceuticals d.d.
Lek Pharmaceuticals d.d (Lendava) (Primary and Secondary Packager)
Active Ingredients
Documents
Package Inserts
Levetiracetam Sandoz FCT Tablets PI.pdf
Approved: September 10, 2020
