Regulatory Information
HSA regulatory responsibility and product classification details
Regulatory Responsibility
Product Classification
Formulation Information
TABLET, FILM COATED
**Dosage regimen and administration** Treatment with Kisqali® should be initiated by a physician experienced in the use of anticancer therapies. **Dosage regimen** **General target population** The recommended dose of Kisqali is 600 mg (3 x 200 mg film-coated tablets) taken orally, once daily for 21 consecutive days followed by 7 days off treatment resulting in a complete cycle of 28 days. Kisqali can be taken with or without food (see section Interactions – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). When co-administered with Kisqali, refer to the full prescribing information for the recommended dose of the aromatase inhibitor to be used. Patients should take their dose of Kisqali and aromatase inhibitor at approximately the same time each day, preferably in the morning. When co-administered with Kisqali, the recommended dose of fulvestrant is 500mg administered intramuscularly on days 1, 15 and 29, and once monthly thereafter. Please refer to the full prescribing information of fulvestrant. Pre/peri-menopausal women or men treated with the combination of Kisqali plus an aromatase inhibitor should also receive a luteinizing hormone-releasing hormone (LHRH) agonist according to local clinical practice standards. Men treated with the combination of Kisqali plus fulvestrant should be treated with a luteinizing hormone-releasing hormone (LHRH) agonist according to local clinical practice standards. **Dose modifications** Management of severe or intolerable adverse drug reactions (ADRs) may require temporary dose interruption, reduction, or discontinuation of Kisqali. If dose reduction is required, the recommended dose reduction guidelines for adverse drug reactions (ADRs) are listed in Table 1. 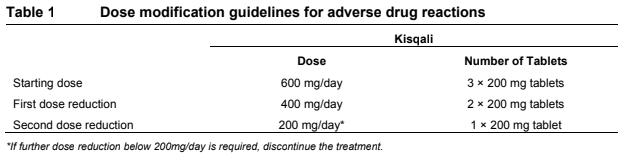 Tables 2, 3, 4, 5 and 6 summarize recommendations for dose interruption, reduction, or discontinuation of Kisqali in the management of specific ADRs. Clinical judgment of the treating physician should guide the management plan of each patient based on individual benefit/risk assessment (see sections Warnings and precautions, Adverse drug reactions – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). 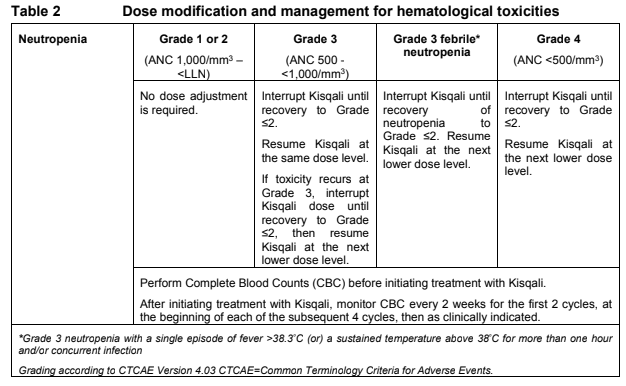 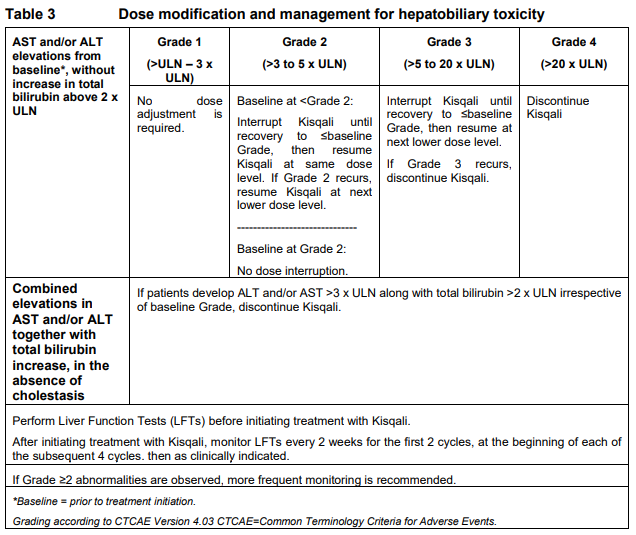  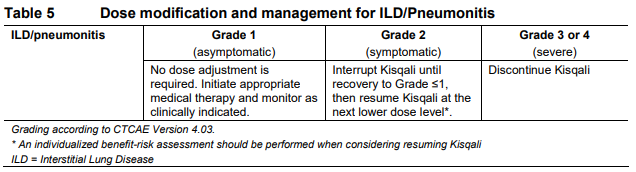  **Dose modification for use of Kisqali with strong CYP3A inhibitors** Concomitant use of Kisqali should be avoided with strong CYP3A inhibitors and an alternative concomitant medication should be considered with less potential for CYP3A inhibition. If a strong CYP3A inhibitor must be co-administered, the Kisqali dose should be reduced to 200 mg once daily If the strong inhibitor is discontinued, the Kisqali dose should be changed (after at least 5 elimination half-lives of the strong CYP3A inhibitor) to the dose used prior to the initiation of the strong CYP3A inhibitor (see sections Warnings and precautions, Interactions and Clinical pharmacology – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). **Special populations** **Renal impairment** Based on population pharmacokinetic analysis, and data from cancer patients in clinical trials, no dose adjustment is necessary in patients with mild or moderate renal impairment (see section Clinical pharmacology – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). Based on a renal impairment study in healthy subjects and non-cancer subjects with severe renal impairment, a starting dose of 200 mg is recommended. Kisqali has not been studied in breast cancer patients with severe renal impairment. (see section Clinical pharmacology – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). **Hepatic impairment** Based on a hepatic impairment study in healthy subjects and non-cancer subjects with impaired hepatic function, no dose adjustment is necessary in patients with mild hepatic impairment (Child-Pugh class A). A dose adjustment is required in patients with moderate (Child-Pugh class B) and severe hepatic impairment (Child-Pugh class C) and the starting dose of 400mg is recommended. Kisqali has not been studied in breast cancer patients with moderate and severe hepatic impairment (see section Clinical pharmacology – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). **Pediatric patients** There are limited data in pediatric patients and the safety and efficacy of Kisqali in this population have not been established. **Geriatric patients (65 years of age or older)** No dose adjustment is required in patients over 65 years of age (see section Clinical pharmacology – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). **Method of administration** Kisqali should be taken orally once daily at the same time every day, preferably in the morning, with or without food. If the patient vomits after taking the dose or misses a dose, an additional dose should not be taken that day. The next prescribed dose should be taken at the usual time. Kisqali tablets should be swallowed whole (tablets should not be chewed, crushed or split prior to swallowing). Tablets that are broken, cracked, or otherwise not intact should not be ingested.
ORAL
Medical Information
**Indications** Kisqali is indicated for the treatment of adult patients with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer in combination with: - an aromatase inhibitor as initial endocrine-based therapy in pre/perimenopausal or postmenopausal women or in men; or - fulvestrant as initial endocrine-based therapy or following disease progression on endocrine therapy in postmenopausal women or in men.
**Contraindications** Kisqali is contraindicated in patients with hypersensitivity to the active substance or to any of the excipients.
L01EF02
ribociclib
Manufacturer Information
NOVARTIS (SINGAPORE) PTE LTD
Novartis Pharma Produktions GmbH (Primary packager and Secondary packager)
NOVARTIS SINGAPORE PHARMACEUTICAL MANUFACTURING PTE. LTD.
Novartis Pharma Stein AG (Primary packager and Secondary packager)
Active Ingredients
Documents
Package Inserts
Kisqali Film coated Tablets 200mg PI.pdf
Approved: November 9, 2022
