Regulatory Information
HSA regulatory responsibility and product classification details
Regulatory Responsibility
Product Classification
Formulation Information
TABLET, FILM COATED
**4.2 Dose and method of administration** Dosage (dose and interval) Treatment with Otezla should be initiated by specialists experienced in the diagnosis and treatment of psoriasis or psoriatic arthritis. The recommended dose of Otezla is 30 mg twice daily taken orally approximately 12 hours apart. An initial titration schedule is required as shown below in Table 1. No re-titration is required after initial titration. 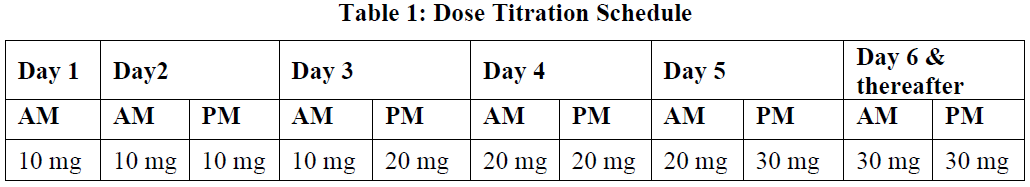 Method of administration Otezla tablets should be swallowed whole, either with or without food. The tablets should not be crushed, split or chewed. If patients miss a dose, the next dose should be taken as soon as possible. If it is close to the time for their next dose, the missed dose should not be taken and the next dose should be taken at the regular time. Dosage adjustment No dose adjustment is necessary for elderly patients (≥ 65 years of age). _Renal impairment_ No dose adjustment is needed in patients with mild renal impairment. There are limited data on moderate renal impairment. The dose of Otezla should be reduced to 30 mg once daily in patients with severe renal impairment (creatinine clearance of less than 30 mL per minute estimated by the Cockroft-Gault equation). For initial dose titration in this group, it is recommended that apremilast be titrated using only the AM schedule listed in Table 1 and the PM doses be skipped. _Hepatic impairment_ Dose adjustment is not required in patients with hepatic impairment. The safety of Otezla has not been evaluated in PsA or PSOR patients with hepatic impairment. _Monitoring advice_ In the event of intolerable adverse events, interruption or discontinuation of Otezla should be considered.
ORAL
Medical Information
**4.1 Therapeutic Indications** Otezla is indicated for: - Otezla, alone or in combination with a disease-modifying anti-rheumatic drug (DMARD), is indicated for the treatment of signs and symptoms of active psoriatic arthritis in adult patients who have had an inadequate response, intolerance, or contraindication to a prior DMARD therapy. - The treatment of adult patients with plaque psoriasis who are candidates for phototherapy or systemic therapy.
**4.3 Contraindications** Otezla is contraindicated: - In patients with known hypersensitivity to the active substance or to any of the excipients. - During pregnancy and in nursing women.
L04AA32
apremilast
Manufacturer Information
AMGEN BIOTECHNOLOGY SINGAPORE PTE. LTD.
Celgene International Sarl
Patheon Inc
Patheon Inc. (Testing only)
