Lidocaine Patch
Lidocaine Patch 24 Hours
0a31ba9a-8283-4bf8-b6c6-6e331465622f
HUMAN OTC DRUG LABEL
Aug 20, 2025
SLV PHARMACEUTICALS LLC DBA AUM PHARMACEUTICALS
DUNS: 081225162
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
LIDOCAINE 4%
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (4)
Drug Labeling Information
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL

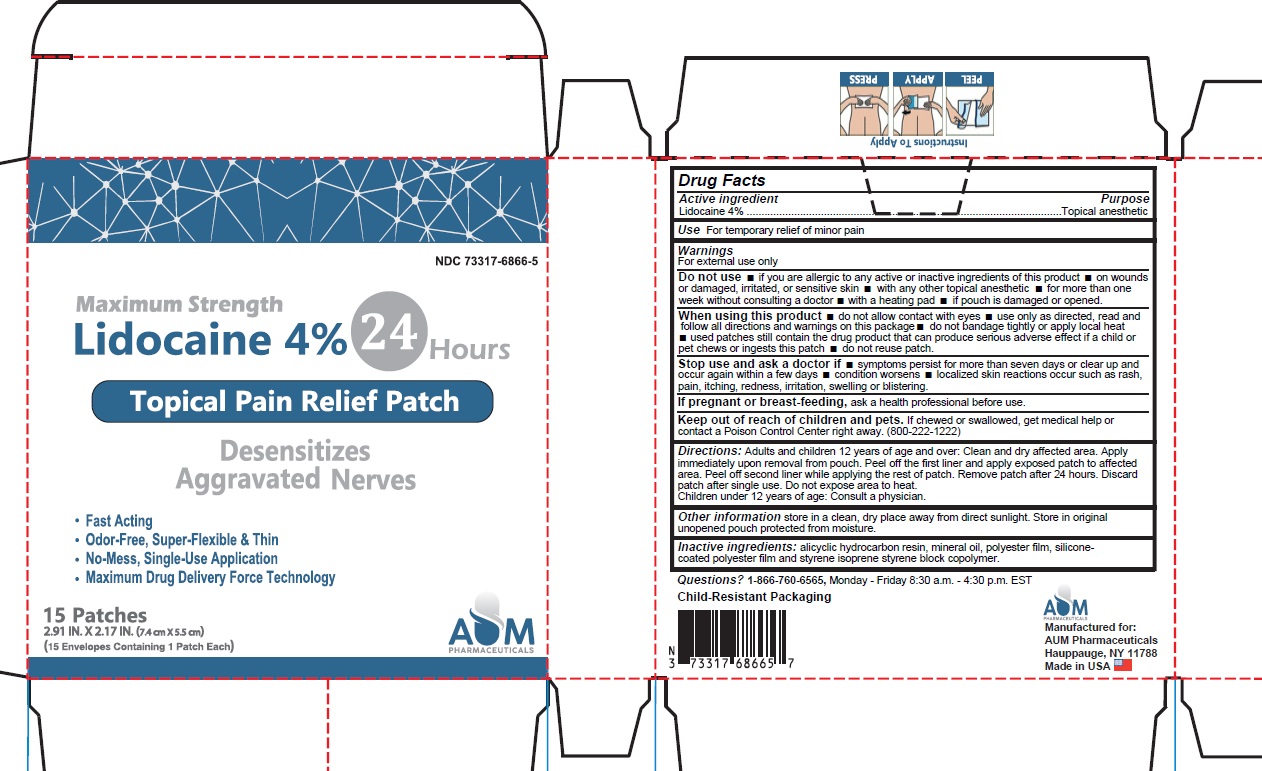
INDICATIONS & USAGE SECTION
Idications and Usage
Uses: Temporaily relieves minor pains.
OTC - ACTIVE INGREDIENT SECTION
Active Ingredients
Lidocaine 4% . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Topical Anesthetic
OTC - PURPOSE SECTION
Purpose
Temporarily relieves minor pain.
OTC - WHEN USING SECTION
When using this product
■ use only as directed
■ read and follow all directions and warnings on this carton
■ do not allow contact with the eyes
■ do not use at the same time as other topical analgesics
■ do not bandage tightly or apply local heat (such as heating pads) to the area of use
■ do not microwave
■ dispose of used patch in manner that always keeps product away from children and pets. Used patches still contain the drug product that can produce serious adverse effects if a child or pet chews or ingests this patch.
DOSAGE & ADMINISTRATION SECTION
Dosage and Administration
Directions Adults and children over 12 years:
■ clean and dry affected area
■ carefully remove backing from patch starting at corner
■ apply sticky side of patch to affected area
■ use one patch for up to 24 hours. Discard patch after single use.Children under 12 years of age: consult a physician.
WARNINGS SECTION
Warnings
For external use only.
OTC - STOP USE SECTION
Stop use and consult a doctor
■ condition worsens
■ redness is present
■ irritation develops
■ symptoms persist for more than 7 days or clear up and occur again within a few days
■ you experience signs of skin injury, such as pain, swelling or blistering where the product was applied.
OTC - PREGNANCY OR BREAST FEEDING SECTION
If pregnant or breastfeeding, ask a health professional before use.
OTC - DO NOT USE SECTION
Do not Use
■ more than one patch on your body at a time
■ on cut, irritated or swollen skin
■ on puncture wounds
■ for more than one week without consulting a doctor
■ if you are allergic to any active or inactive ingredients
■ if pouch is damaged or opened.
OTC - KEEP OUT OF REACH OF CHILDREN SECTION
Keep out of reach of children and pets.
If swallowed, get medical help or contact a Poison Control Center right away 800-222-1222
OTHER SAFETY INFORMATION
Other Safety Information
Store in a clean, dry place outside of direct sunlight. Protect from excessive moisture.
INACTIVE INGREDIENT SECTION
Inactive Ingredients
Styrene Isoprene Styrene block Copolymer, Alicyclic Saturated Hydrocarbon Resin, White Mineral Oil
OTC - QUESTIONS SECTION
Questions
Questions? 1-866-760-6565, Monday - Friday 8:30 a.m - 4:30 p.m. EST
Manufactured for:
AUM Pharmaceuticals
Hauppauge, NY 11788
Made in USA
