Leader Restorative Formula Lubricant Eye Drops
Leader Restorative Formula Lubricant Eye Drops (PLD)
87fef7e4-e5a8-4aad-94d2-f80f34826d97
HUMAN OTC DRUG LABEL
Aug 1, 2025
Cardinal Health
DUNS: 063997360
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
propylene glycol
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (10)
Drug Labeling Information
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
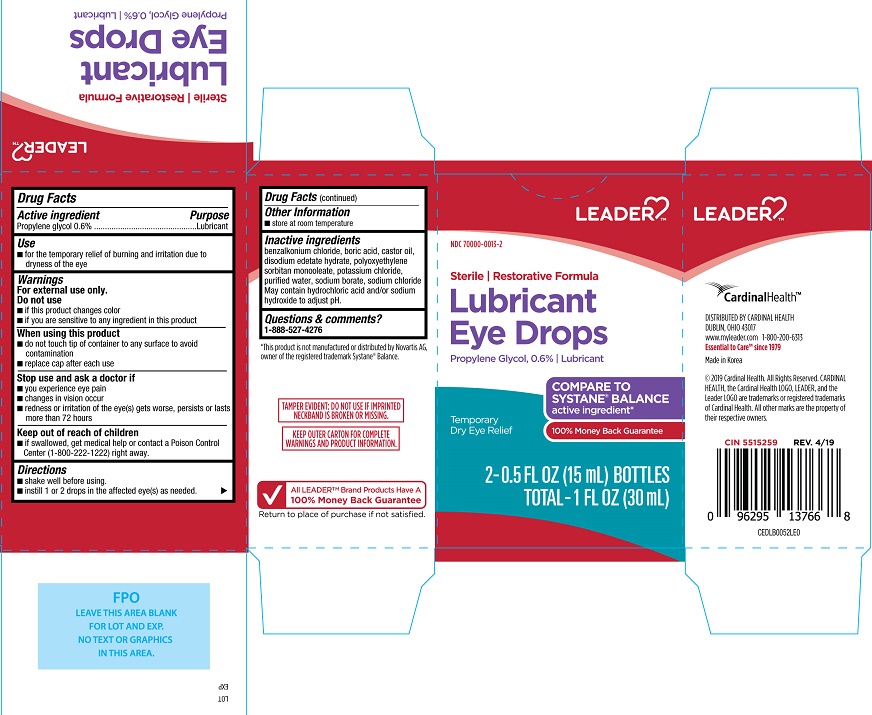
INDICATIONS & USAGE SECTION
Uses
• for the temporary relief of burning and irritation due to dryness of the eye
OTC - ACTIVE INGREDIENT SECTION
Active ingredients
Propylene glycol....0.6%
OTHER SAFETY INFORMATION
Other information
- Store at room temperature.
OTC - PURPOSE SECTION
Purpose
Propylene glycol............ Lubricant
WARNINGS SECTION
Warnings
For external use only
Do not use
• if this product changes color
• if you are sensitive to any ingredient in this product
When using this product
• do not touch tip of container to any surface to avoid contamination
• replace cap after each use
Stop use and ask a doctor if
• you experience eye pain
• changes in vision occur
• redness or irritation of the eye(s) gets worse, persists or lasts more than
72 hours
OTC - KEEP OUT OF REACH OF CHILDREN SECTION
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center
(1-800-222-1222) right away.
DOSAGE & ADMINISTRATION SECTION
Directions
- Shake well before using.
- Instill 1 or 2 drops in the affected eye(s) as needed
INACTIVE INGREDIENT SECTION
**Inactive ingredients**
Benzalkonium chloride, boric acid, castor oil, disodium edetate hydrate, polyoxyethylene sorbitan monooleate, potassium chloride, purified water, sodium borate, sodium chloride. May contain hydrochloric acid and/or sodium hydroxide to adjust pH.
OTC - QUESTIONS SECTION
Questions & comments?
1-888-527-4276
