LMD in Dextrose
10% LMD in 5% Dextrose Injection Rx only (Dextran 40 in Dextrose Injection, USP)
3c273512-84e2-2cea-b201-12345a6bd1fe
HUMAN PRESCRIPTION DRUG LABEL
Feb 15, 2023
Hospira, Inc.
DUNS: 141588017
Products 2
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
DEXTRAN 40
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (3)
DEXTRAN 40
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (5)
Drug Labeling Information
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 500 mL Bag Overwrap - 7419
2
HDPE
TO OPEN TEAR AT NOTCH
DO NOT REMOVE FROM OVERWRAP UNTIL READY FOR USE. AFTER REMOVING
THE OVERWRAP, CHECK FOR MINUTE LEAKS BY SQUEEZING CONTAINER
FIRMLY. IF LEAKS ARE FOUND, DISCARD SOLUTION AS STERILITY MAY BE
IMPAIRED. RECOMMENDED STORAGE: ROOM TEMPERATURE (25°C). AVOID
EXCESSIVE HEAT. PROTECT FROM FREEZING. SEE INSERT.
IM-4498
Not Made With Natural Rubber Latex
14622100

PRECAUTIONS SECTION
PRECAUTIONS
The possibility of circulatory overload should be kept in mind. Special care should be exercised in patients with impaired renal clearance of dextran. When the risk of pulmonary edema and/or congestive heart failure may be increased, dextran should be used with caution.
In patients with normal hemostasis, dosage of LMD (dextran 40) approximating 15 mL/kg of body weight may prolong bleeding time and depress platelet function. Dosages in this range also markedly decrease factor VIII, and decrease factors V and IX to a greater degree than would be expected to occur from hemodilution alone. Since these changes tend to be more pronounced following trauma or major surgery, patients should be observed for early signs of bleeding complications.
Since increased rouleaux formation may occur in the presence of dextran, it is recommended that blood samples be drawn for typing and cross-matching prior to the infusion of dextran and reserved for subsequent use if necessary. If blood is drawn after infusion of dextran, the saline agglutination and indirect antiglobulin methods may be used for typing and cross-matching. Difficulty may be encountered when proteolytic enzyme techniques are used to match blood.
Consideration should be given to withdrawal of blood for chemical laboratory tests prior to initiating therapy with dextran because of the following:
Blood sugar determinations that employ high concentrations of acid may result in hydrolysis of dextran, yielding falsely elevated glucose assay results. This has been observed both with sulfuric acid and with acetic acid.
In other laboratory tests, the presence of dextran in the blood may result in the development of turbidity, which can interfere with the assay. This has been observed in bilirubin assays in which alcohol is employed and in total protein assays employing biuret reagent.
Solutions containing dextrose should be used with caution in patients with known subclinical or overt diabetes mellitus.
Caution must be exercised in the administration of parenteral fluids, especially those containing sodium ions, to patients receiving corticosteroids or corticotropin.
Do not administer unless solution is clear and container is undamaged. Discard unused portion.
Drug Interactions. Additive medications should not be delivered via plasma volume expanders.
Pregnancy. Animal reproduction studies have not been conducted with dextran 40 in dextrose or sodium chloride. It is also not known whether dextran 40 in dextrose or sodium chloride can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. 10% LMD (dextran 40) in dextrose or sodium chloride should be given to a pregnant woman only if clearly needed.
Nursing Mothers. It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when 10% LMD (dextran 40) in dextrose or sodium chloride is administered to a nursing woman.
Pediatric Use. The safety and effectiveness of dextran 40 have not been established in neonates. Its limited use in neonates has been inadequate to fully define proper dosage and limitations for use.
DOSAGE & ADMINISTRATION SECTION
DOSAGE AND ADMINISTRATION
LMD (dextran 40) is administered by I.V. infusion only.
Dextran 1 should be administered prior to administration of clinical dextran solutions.
**In shock,** it is suggested that total dosage not exceed 20 mL/kg for adults and adolescents, during the first 24 hours. The first 10 mL/kg may be infused as rapidly as necessary to effect improvement. It is strongly recommended that central venous pressure be monitored frequently during the initial infusion of the drug. Should therapy continue beyond 24 hours, subsequent dosage should not exceed 10 mL/kg per day and therapy should not continue beyond five days.
**In extracorporeal perfusion,** the dosage of LMD used will vary with the volume of the pump oxygenator. LMD can serve as a sole primer or as an additive to other priming fluids. For adults and adolescents, generally 10 to 20 mL of a 10% solution (1 to 2 g) of LMD per kilogram of body weight are added to the perfusion circuit. Usually total dosage should not exceed 2 g/kg of body weight.
**In prophylaxis of venous thrombosis and thromboembolism,** the dosage of LMD for adults and adolescents, should be chosen according to the risk of thromboembolic complications, e.g., type of surgery and duration of immobilization. In general, treatment should be initiated during surgery; 500 to 1,000 mL (approximately 10 mL/kg of body weight) should be administered on the day of operation. Treatment should be continued at a dose of 500 mL daily for an additional two to three days; then, according to the risk of complications, 500 mL may be given every second or third day during the period of risk, for up to two weeks.
Infants may be given 5 mL per kg body weight and children 10 mL per kg.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. SeePRECAUTIONS.
Note: When infusing concentrated LMD, the administration set should include a filter.
Instructions for use
To Open
Tear outer wrap at notch and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect solution quality or safety. The opacity will diminish gradually.
Preparation for Administration
(Use aseptic technique)
Close flow control clamp of administration set.
Remove cover from outlet port at bottom of container.
Insert piercing pin of administration set into port with a twisting motion until the set is firmly seated.**Note**: See full directions on administration set carton.
Suspend container from hanger.
Squeeze and release drip chamber to establish proper fluid level in drip chamber.
Open flow control clamp and clear air from set. Close clamp.
Attach set to venipuncture device. If device is not indwelling, prime and make venipuncture.
Regulate rate of administration with flow control clamp.
**WARNING: Do not use flexible container in series connections.**
DESCRIPTION SECTION
DESCRIPTION
LMD (dextran 40) is a sterile, nonpyrogenic preparation of low molecular weight dextran (average mol. wt. 40,000) in 5% Dextrose Injection or 0.9% Sodium Chloride Injection. It is administered by intravenous infusion.
Also described as low viscous or low viscosity dextran, dextran 40 is prepared by acid hydrolysis and differential fractionation of a crude macromolecular polysaccharide produced from the fermentation of sucrose by the bacterium, Leuconostoc mesenteroides (strain B-512). The crude material is composed of linked glucose units. In the fraction represented by dextran 40, 80% of the molecules have a molecular weight ranging from 10,000 to 90,000 (average approximately 40,000) when measured by a light scattering method. More than 90% of the linkages are of the 1,6 alpha glucosidic, straight chain type.
Each 100 mL of 10% LMD (dextran 40) in 5% Dextrose Injection contains 10 g dextran 40 and 5 g dextrose hydrous in water for injection. Total osmolar concentration is 255 mOsmol/liter (calc.); pH is 4.4 (3.0 to 7.0).
Each 100 mL of 10% LMD (dextran 40) in 0.9% Sodium Chloride Injection contains 10 g dextran 40 and 0.9 g sodium chloride in water for injection. Total osmolar concentration is 310 mOsmol/liter (calc.); pH is 4.9 (3.5 to 7.0) (may contain sodium hydroxide and/or hydrochloric acid for pH adjustment). Electrolyte concentration per liter: Na+ 154 mEq; Cl- 154 mEq (not including ions for pH adjustment).
The solutions contain no bacteriostat, antimicrobial agent or added buffers (except for pH adjustment) and are intended only for single-dose injection. When smaller doses are required the unused portion should be discarded.
10% LMD (dextran 40) is an artificial colloid pharmacologically classified as a plasma volume expander; 5% Dextrose Injection is a fluid and nutrient replenisher; 0.9% Sodium Chloride Injection is a fluid and electrolyte replenisher.
Dextran 40 is a linear glucose polymer (polysaccharide) chemically designated (C6 H10 O5)n.
The structural formula for dextran (repeating unit) is:
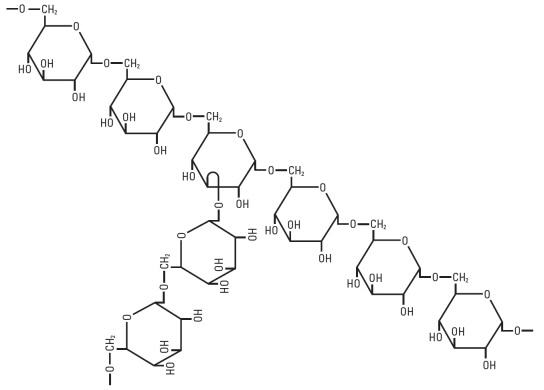
Dextrose, USP is chemically designated D-glucose monohydrate (C6 H12 O6 • H2O), a hexose sugar freely soluble in water.
Sodium Chloride, USP is chemically designated NaCl, a white crystalline powder freely soluble in water.
Water for Injection, USP is chemically designated H2O.
The flexible plastic container is fabricated from a specially formulated polyvinylchloride. Water can permeate from inside the container into the overwrap but not in amounts sufficient to affect the solution significantly. Solutions inside the plastic container also can leach out certain of the chemical components of the plastic in very small amounts before the expiration period is attained. However, safety of the plastic has been confirmed by tests in animals according to USP biological standards for plastic containers.
CLINICAL PHARMACOLOGY SECTION
CLINICAL PHARMACOLOGY
The fundamental action of LMD (dextran 40) is the enhancement of blood flow, particularly in the microcirculation. This enhancement is due to:
Its primary effect of volume expansion with resultant hemodilution;
Maintenance of the electronegativity of red blood cells;
Coating of red blood cells and platelets;
Increase in the suspension stability of blood;
Decrease in the viscosity of blood.
It should be emphasized that the above effects are not exerted separately, but conjointly they result in the enhancement of blood flow.
LMD, used in the treatment of shock, produces significant increases in blood volume, central venous pressure, cardiac output, stroke volume, blood pressure and urinary output. It reduces blood viscosity, peripheral resistance and improves peripheral blood flow with the release of sequestered blood cells, thereby increasing venous return to the heart.
When used as part of the pump prime for extracorporeal procedures, LMD, as compared to whole blood, albumin 5%, or whole blood plus 5% dextrose and water, leads to less destruction of red blood cells and platelets, reduces intravascular hemagglutination and maintains erythrocyte electronegativity.
The infusion of LMD (dextran 40) during and after surgical trauma reduces the incidence of deep venous thrombosis (DVT) and pulmonary embolism (PE) in patients subject to surgical procedures with a high incidence of thromboembolic complication. Unlike antithrombogenic agents of the anticoagulant type, LMD does not achieve its effect so much by blocking fibrinogen-fibrin conversion but acts by simultaneously inhibiting other mechanisms essential to thrombus formation such as vascular stasis and platelet adhesiveness and by altering the structure and thereby the lysability of fibrin clots.
Histopathological studies have shown that the development of a mural platelet thrombus is the first stage of thrombus formation not only in the arterial, but also in the venous system. A number of studies have further shown that many patients who develop thromboembolic complications show an abnormally high platelet adhesiveness. Infusion of LMD has been shown to reduce platelet adhesiveness as measured by various in vitro tests on blood samples obtained from humans and to inhibit the growth of a mural platelet thrombus at the site of experimental (laser beam) injury in the rabbit's ear chamber.
Studies have shown an increase in the lysability of thrombi formed in the presence of dextran. A consistent and characteristic alteration in fibrin structure has been observed when fibrin is formed in the presence of dextran, and further experiments demonstrated such fibrin to be more susceptible to plasmin digestion. Other studies have shown that dextran infused into patients during surgery increases the lysability of ex vivo thrombi. Controlled clinical trials have shown that thrombi in patients treated with dextran have a more pronounced tendency to undergo lysis as determined by phlebography.
LMD is evenly distributed in the vascular system. Its distribution according to molecular weight shifts toward higher molecular weights as the smaller molecules are excreted by the kidney. In normovolemic subjects, approximately 50% is excreted within 3 hours, 60% is excreted within 6 hours and about 75% within 24 hours. Reabsorption of dextran by the renal tubules is negligible. The unexcreted molecules of dextran diffuse into the extravascular compartment and are temporarily taken up by the reticuloendothelial system. Some of these molecules are returned to the intravascular compartment via the lymphatics. Dextran is slowly degraded by the enzyme dextranase to glucose.
Solutions containing carbohydrate in the form of dextrose restore blood glucose levels and provide calories. Carbohydrate in the form of dextrose may aid in minimizing liver glycogen depletion and exerts a protein sparing action. Dextrose injected parenterally undergoes oxidation to carbon dioxide and water.
Sodium chloride in water dissociates to provide sodium (Na+) and chloride (Cl-) ions. Sodium (Na+) is the principal cation of the extracellular fluid and plays a large part in the therapy of fluid and electrolyte disturbances. Chloride (Cl-) has an integral role in buffering action when oxygen and carbon dioxide exchange occurs in red blood cells. The distribution and excretion of sodium (Na+) and chloride (Cl-) are largely under the control of the kidney, which maintains a balance between intake and output.
Water is an essential constituent of all body tissues and accounts for approximately 70% of total body weight. Average normal adult daily requirement ranges from two to three liters (1.0 to 1.5 liters each for insensible water loss by perspiration and urine production).
Water balance is maintained by various regulatory mechanisms. Water distribution depends primarily on the concentration of electrolytes in the body compartments and sodium (Na+) plays a major role in maintaining physiologic equilibrium.
HOW SUPPLIED SECTION
HOW SUPPLIED
10% LMD in 5% Dextrose Injection (Dextran 40 in Dextrose Injection, USP) is supplied as follows:
|
Unit of Sale |
Concentration Dextran 40 |
|
NDC 0409-7418-03 |
50 g/500 mL |
10% LMD in 0.9% Sodium Chloride Injection (Dextran 40 in Sodium Chloride Injection, USP) is supplied as follows:
|
Unit of Sale |
Concentration Dextran 40 |
|
NDC 0409-7419-03 |
50 g/500 mL |
Do not use if crystallization has occurred.
Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.] Protect from freezing.
REFERENCES SECTION
REFERENCES
Richter W. Build-in hapten inhibition of anaphylaxis by the low molecular weight fractions of a B 512 dextran fraction of MW 3400. Int Arch Allergy 1973;45:930
Lungstrom K-G, Renck H, Hedin H, et al. Prevention of dextran-induced anaphylactic reactions by hapten inhibition. I. A Scandinavian multicenter study on the effects of 10 mL dextran 1, 15%, administered before Dextran 70 or Dextran 40. Acta Chir Scand 1983;149:341-348
Renck H, Ljungstrom K-G, Rosberg B, et al. Prevention of dextran-induced anaphylactic reactions by hapten inhibition. II. A comparison of the effects of 20 mL dextran 1, 15%, administered either admixed to or before Dextran 70 or Dextran 40. Acta Chir Scand 1983;149:349-353
Renck H, Ljungstrom K-G, Hedin H, et al. Prevention of dextran-induced anaphylactic reactions by hapten inhibition. III. A Scandinavian multicenter study on the effects of 20 mL dextran 1, 15%, administered before Dextran 70 or Dextran 40. Acta Chir Scand 1983;149:355-360
Ljungstrom K-G, Renck H, Strandberg K, et al. Adverse reactions to dextran in Sweden 1970-1979. Acta Chir Scand 1983;149:253-262
Hedin H, Ljungstrom K-G. Prevention of dextran anaphylaxis. Ten years experience with hapten dextran. Int Arch Allergy Immunol 1997;113:358-359

Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
LAB-1252-4.0
Revised: 10/2018
