cough and cold relief HBP
Equaline 44-689
90482e7a-d7c7-4127-972d-5ee55e55ed1c
HUMAN OTC DRUG LABEL
Aug 12, 2025
United Natural Foods, Inc. dba UNFI
DUNS: 943556183
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Chlorpheniramine maleate, Dextromethorphan HBr
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (14)
Drug Labeling Information
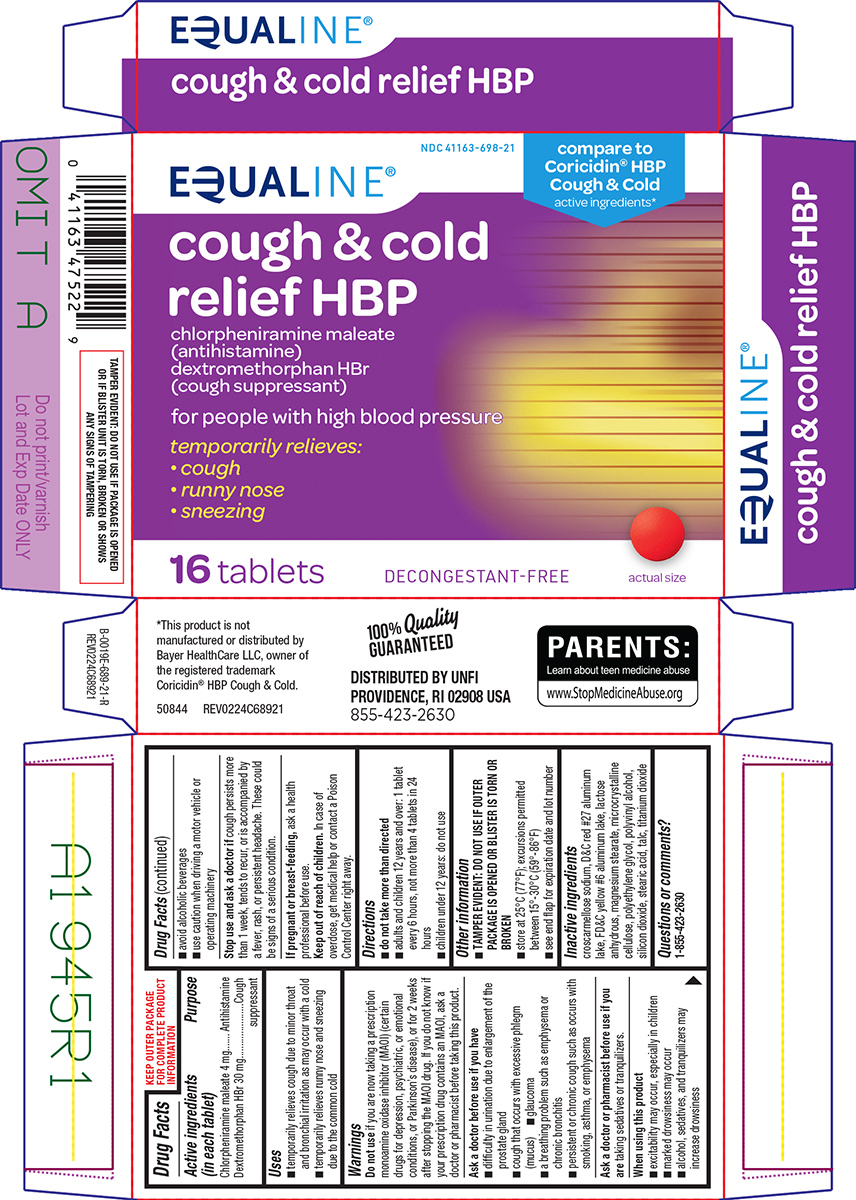
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Principal Display Panel
EQUALINE®
NDC 41163-698-21
compare to
** Coricidin****®**** HBP**
** Cough & Cold**
active ingredients*
cough & cold
** relief HBP**
chlorpheniramine maleate
(antihistamine)
dextromethorphan HBr
(cough suppressant)
for people with high blood pressure
temporarily relieves:
• cough
• runny nose
• sneezing
16 tablets
DECONGESTANT-FREE
actual size
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED
** OR IF BLISTER UNIT IS TORN, BROKEN OR SHOWS**
** ANY SIGNS OF TAMPERING**
**100%**Quality
****GUARANTEED
DISTRIBUTED BY UNFI
** PROVIDENCE, RI 02908 USA**
****855-423-2630
*This product is not
manufactured or distributed by
Bayer HealthCare LLC, owner
of the registered trademark
Coricidin® HBP Cough & Cold.
50844 REV0224C68921
PARENTS:
Learn about teen medicine abuse
www.StopMedicineAbuse.org
Equaline 44-689
INDICATIONS & USAGE SECTION
Uses
- temporarily relieves cough due to minor throat and bronchial irritation as may occur with a cold
- temporarily relieves runny nose and sneezing due to the common cold
OTC - QUESTIONS SECTION
Questions or comments?
1-855-423-2630
OTC - ACTIVE INGREDIENT SECTION
Active ingredients (in each tablet)
Chlorpheniramine maleate 4 mg
Dextromethorphan HBr 30 mg
OTC - PURPOSE SECTION
Purpose
Antihistamine
Cough suppressant
WARNINGS SECTION
Warnings
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- difficulty in urination due to enlargement of the prostate gland
- cough that occurs with excessive phlegm (mucus)
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- persistent or chronic cough such as occurs with smoking, asthma, or emphysema
Ask a doctor or pharmacist before use if you are
taking sedatives or tranquilizers.
When using this product
- excitability may occur, especially in children
- marked drowsiness may occur
- alcohol, sedatives, and tranquilizers may increase drowsiness
- avoid alcoholic beverages
- use caution when driving a motor vehicle or operating machinery
Stop use and ask a doctor if
cough persists more than 1 week, tends to recur, or is accompanied by a fever, rash, or persistent headache. These could be signs of a serious condition.
If pregnant or breast-feeding,
ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
DOSAGE & ADMINISTRATION SECTION
Directions
*do not take more than directed
- adults and children 12 years and over: 1 tablet every 6 hours, not more than 4 tablets in 24 hours.
- children under 12 years: do not use
STORAGE AND HANDLING SECTION
Other information
*TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
INACTIVE INGREDIENT SECTION
Inactive ingredients
croscarmellose sodium, D&C red #27 aluminum lake, FD&C yellow #6 aluminum lake, lactose anhydrous, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, silicon dioxide, stearic acid, talc, titanium dioxide
