YAY NOVELTY ALL-IN-ONE
YAY NOVELTY ALL-IN-ONE
Approved
Approval ID
fb833158-d0d8-de7f-e053-6294a90adc5a
Product Type
HUMAN OTC DRUG LABEL
Effective Date
Aug 26, 2025
Manufacturers
FDA
YAY Novelty
DUNS: 107893510
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Titanium dioxide, Zinc Oxide
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
NDC Product Code83336-0391
Application NumberM020
Product Classification
M
Marketing Category
C200263
G
Generic Name
Titanium dioxide, Zinc Oxide
Product Specifications
Route of AdministrationTOPICAL
Effective DateAugust 26, 2025
FDA Product Classification
INGREDIENTS (39)
FERRIC OXIDE REDInactive
Code: 1K09F3G675
Classification: IACT
FERROSOFERRIC OXIDEInactive
Code: XM0M87F357
Classification: IACT
BUTYLOCTYL SALICYLATEInactive
Code: 2EH13UN8D3
Classification: IACT
POLYESTER-7Inactive
Code: 0841698D2F
Classification: IACT
GLYCERINInactive
Code: PDC6A3C0OX
Classification: IACT
NEOPENTYL GLYCOL DIHEPTANOATEInactive
Code: 5LKW3C543X
Classification: IACT
GLYCERYL STEARATE SEInactive
Code: FCZ5MH785I
Classification: IACT
ALKYL (C12-15) BENZOATEInactive
Code: A9EJ3J61HQ
Classification: IACT
ALLANTOINInactive
Code: 344S277G0Z
Classification: IACT
CAPRYLYL GLYCOLInactive
Code: 00YIU5438U
Classification: IACT
AVENA SATIVA WHOLEInactive
Code: 5P8D0Z74RG
Classification: IACT
DICRATERIA ROTUNDA OILInactive
Code: Z0DWU9R4GM
Classification: IACT
DISODIUM EDTA-COPPERInactive
Code: 6V475AX06U
Classification: IACT
CAMELLIA SINENSIS WHOLEInactive
Code: C5M4585ZBZ
Classification: IACT
TRICETEARETH-4 PHOSPHATEInactive
Code: 69534Y66NO
Classification: IACT
PULLULANInactive
Code: 8ZQ0AYU1TT
Classification: IACT
XANTHAN GUMInactive
Code: TTV12P4NEE
Classification: IACT
STEARETH-20Inactive
Code: L0Q8IK9E08
Classification: IACT
HEXYLENE GLYCOLInactive
Code: KEH0A3F75J
Classification: IACT
TRILAURETH-4 PHOSPHATEInactive
Code: M96W2OLL2V
Classification: IACT
SODIUM ASCORBATEInactive
Code: S033EH8359
Classification: IACT
OCTYLDODECYL NEOPENTANOATEInactive
Code: X8725R883T
Classification: IACT
CETYL ALCOHOLInactive
Code: 936JST6JCN
Classification: IACT
SQUALANEInactive
Code: GW89575KF9
Classification: IACT
PEG-75 STEARATEInactive
Code: OT38R0N74H
Classification: IACT
PHENOXYETHANOLInactive
Code: HIE492ZZ3T
Classification: IACT
.ALPHA.-BISABOLOL, (+)-Inactive
Code: 105S6I733Z
Classification: IACT
ETHYLHEXYLGLYCERINInactive
Code: 147D247K3P
Classification: IACT
ZINC OXIDEActive
Quantity: 9.7 mg in 1 mL
Code: SOI2LOH54Z
Classification: ACTIB
TITANIUM DIOXIDEActive
Quantity: 19.4 mg in 1 mL
Code: 15FIX9V2JP
Classification: ACTIB
DIMETHICONOL/PROPYLSILSESQUIOXANE/SILICATE CROSSPOLYMER (450000000 MW)Inactive
Code: 9KB5R958PB
Classification: IACT
LECITHIN, SOYBEANInactive
Code: 1DI56QDM62
Classification: IACT
SILICON DIOXIDEInactive
Code: ETJ7Z6XBU4
Classification: IACT
ETHYLHEXYL ISONONANOATEInactive
Code: I6KB4GE3K4
Classification: IACT
CETETH-20Inactive
Code: I835H2IHHX
Classification: IACT
POTASSIUM SORBATEInactive
Code: 1VPU26JZZ4
Classification: IACT
TOCOPHEROLInactive
Code: R0ZB2556P8
Classification: IACT
RUTTNERA LAMELLOSA OILInactive
Code: 5XZ38R4SUT
Classification: IACT
FERRIC OXIDE YELLOWInactive
Code: EX438O2MRT
Classification: IACT
Drug Labeling Information
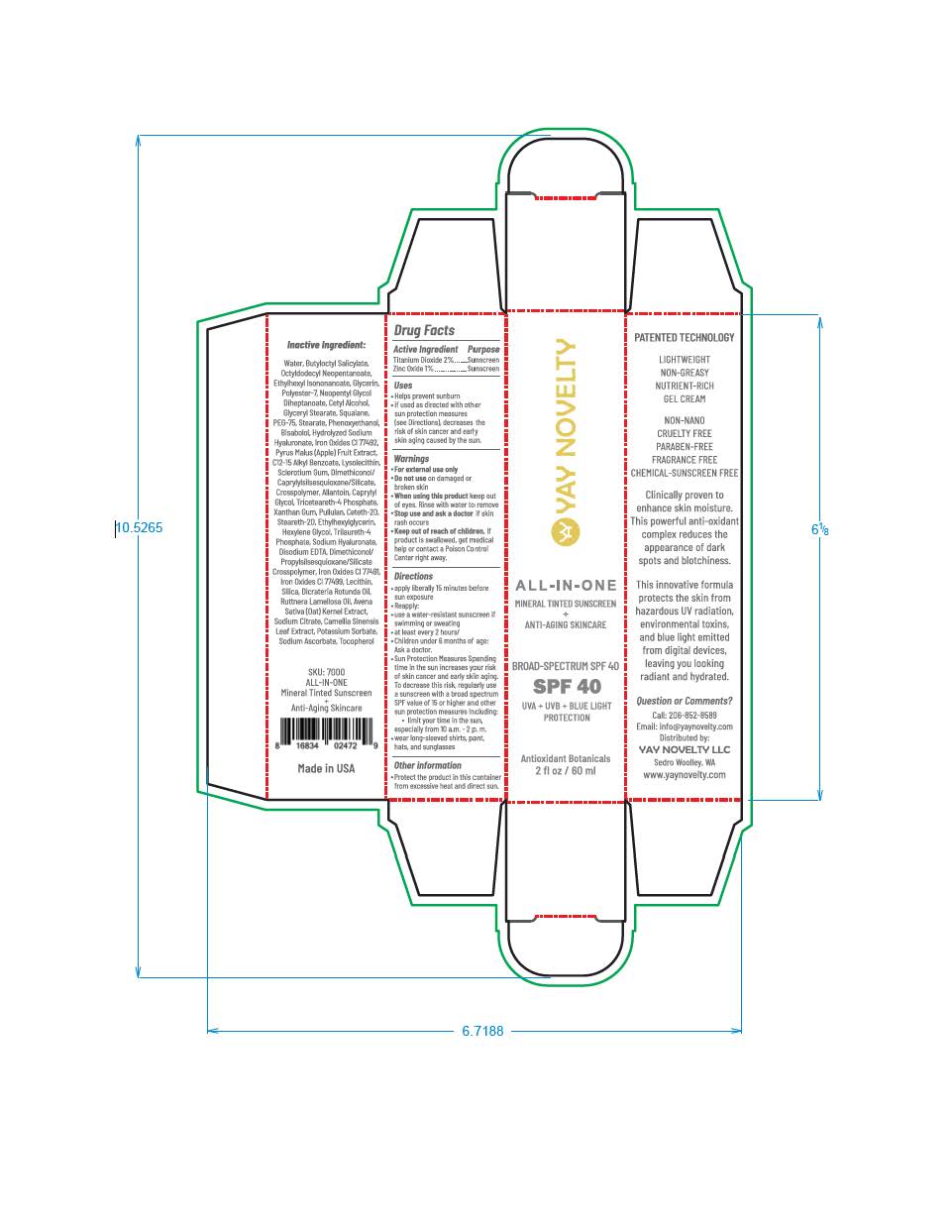
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
LOINC: 51945-4Updated: 4/24/2023
YAY NOVELTY ALL-IN-ONE MINERAL TINTED SUNSCREEN
INDICATIONS & USAGE SECTION
LOINC: 34067-9Updated: 4/24/2023
YAY Novelty All-in-one

DOSAGE & ADMINISTRATION SECTION
LOINC: 34068-7Updated: 4/24/2023
YAY Novelty All-in-one

INACTIVE INGREDIENT SECTION
LOINC: 51727-6Updated: 4/24/2023
YAY Novelty All-in-one

OTC - ACTIVE INGREDIENT SECTION
LOINC: 55106-9Updated: 4/24/2023
YAY Novelty All-in-one

OTC - KEEP OUT OF REACH OF CHILDREN SECTION
LOINC: 50565-1Updated: 4/24/2023
YAY Novelty All-in-one

OTC - PURPOSE SECTION
LOINC: 55105-1Updated: 4/24/2023
YAY Novelty All-in-one

WARNINGS SECTION
LOINC: 34071-1Updated: 4/24/2023
YAY Novelty All-in-one

