Smelling Salts
85839-005 Smelling Salts
3874ec89-0f1d-87b9-e063-6394a90a300a
HUMAN OTC DRUG LABEL
Aug 3, 2025
Shenzhen Finona Information Technology Co., Ltd
DUNS: 511271543
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Ammonia
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (4)
Drug Labeling Information
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
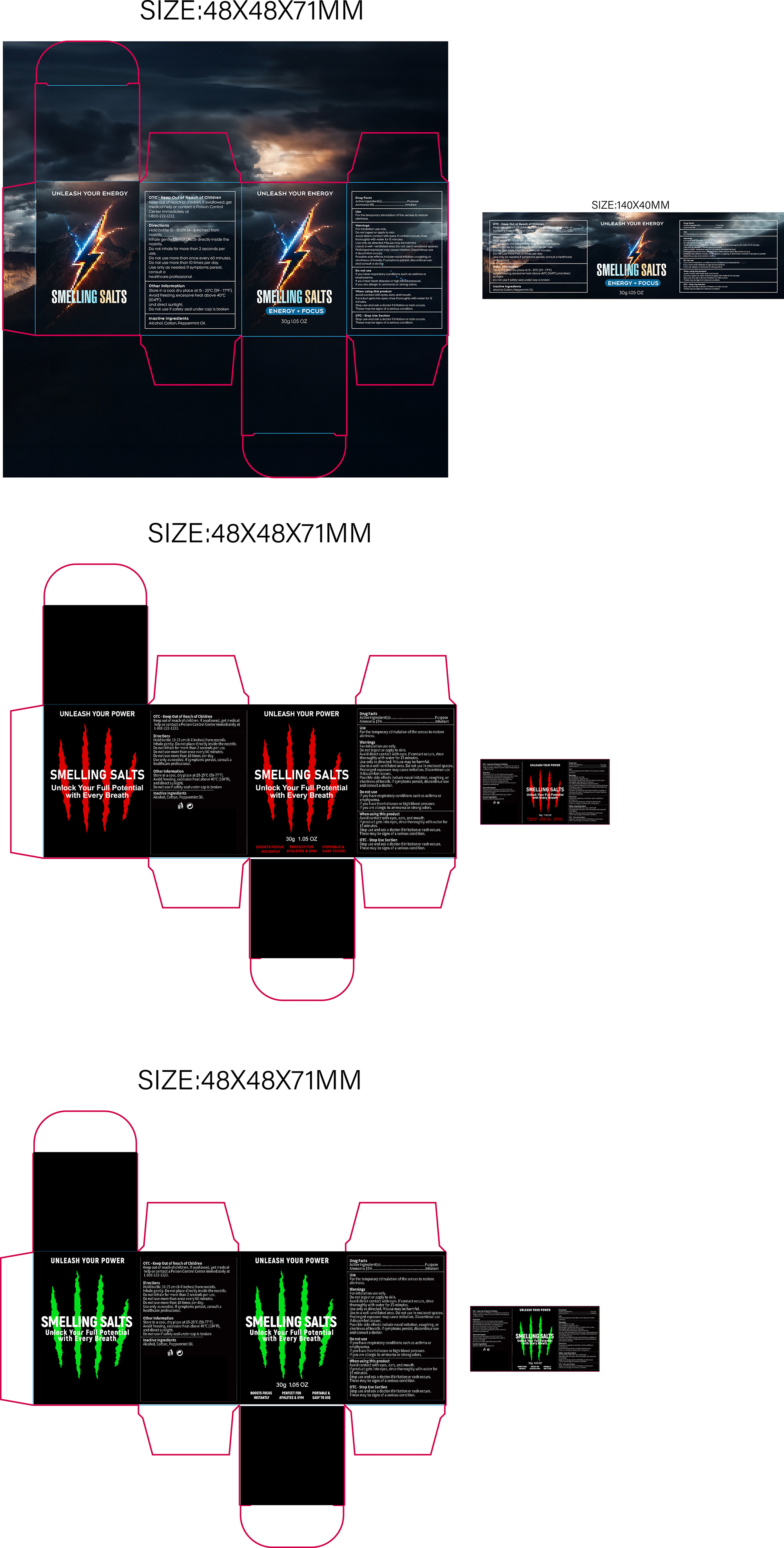
INDICATIONS & USAGE SECTION
For the temporary stimulation of the senses to restore alertness.
OTC - ACTIVE INGREDIENT SECTION
Ammonia 15%
OTC - KEEP OUT OF REACH OF CHILDREN SECTION
Keep out of reach of children. lfswallowed, get medical
help or contact a Poison Control centerimmediately at
1-800-222-1222
OTC - PURPOSE SECTION
Inhalant
OTC - DO NOT USE SECTION
It you have respiratory conditions such as asthma oremphysema.
If you have heart disease or high blood pressure.
If you are allergic to ammonia or strong odors.
WARNINGS SECTION
For inhalation use only.
Do not ingest or apply to skin.
Avoid direct contact with eyes. lf contact occurs, rinsethoroughly with water
for 15 minutes.
Use only as directed. Misuse may be harmful.
Use in a well-ventilated area. Do not use in enclosed spaces.
Prolonged exposure may cause irritation, Discontinue use if discomfort occurs.
Possible side effects include nasal irritation, coughing, or shortness of
breath. lf symptoms persist, discontinue use and consult a doctor.
OTC - WHEN USING SECTION
Avoid contact with eyes, ears, and mouth.
lf product gets into eyes, rinse thoroughly with water for 15 minutes.
Stop use and ask a doctor ifirritation or rash occurs.
These may be signs of a serious condition.
DOSAGE & ADMINISTRATION SECTION
Hold bottle 10-15 cm (4-6 inches) from nostrils.
Inhale gently. Do not place directly inside the nostrils.
Do not inhale for more than 2 seconds per use.
Do not use more than once every 60 minutes.
Do not use more than 10 times per day.
Use only as needed. lfsymptoms persist, consult ahealthcare professional
INACTIVE INGREDIENT SECTION
Alcohol
Cotton
Peppermint Oil
