Atovaquone
These highlights do not include all the information needed to use ATOVAQUONE ORAL SUSPENSION safely and effectively. See full prescribing information for ATOVAQUONE ORAL SUSPENSION. ATOVAQUONE oral suspension Initial U.S. Approval: 1992
f99a8a0c-2b42-4da0-8f5a-e963fc619e8b
HUMAN PRESCRIPTION DRUG LABEL
May 12, 2025
ATLANTIC BIOLOGICALS CORP.
DUNS: 047437707
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Atovaquone
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (7)
Drug Labeling Information
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL

INDICATIONS & USAGE SECTION
1 INDICATIONS AND USAGE
1.1 Prevention of Pneumocystis jiroveciiPneumonia
Atovaquone oral suspension is indicated for the prevention of Pneumocystis jiroveciipneumonia (PCP) in adults and adolescents (aged 13 years and older) who cannot tolerate trimethoprim-sulfamethoxazole (TMP-SMX).
1.2 Treatment of Mild-to-Moderate PneumocystisjiroveciiPneumonia
Atovaquone oral suspension is indicated for the acute oral treatment of mild- to-moderate PCP in adults and adolescents (aged 13 years and older) who cannot tolerate TMP-SMX.
1.3 Limitations of Use
Clinical experience with atovaquone for the treatment of PCP has been limited to subjects with mild-to-moderate PCP (alveolar-arterial oxygen diffusion gradient [(A-a)DO 2] ≤45 mm Hg). Treatment of more severe episodes of PCP with atovaquone has not been studied. The efficacy of atovaquone in subjects who are failing therapy with TMP-SMX has also not been studied.
Atovaquone oral suspension is a quinone antimicrobial drug indicated for:
• Prevention of Pneumocystis jirovecii pneumonia (PCP) in adults and
adolescents aged 13 years and older who cannot tolerate trimethoprim-
sulfamethoxazole (TMP-SMX). ( 1.1)
•Treatment of mild-to-moderate PCP in adults and adolescents aged 13 years and
older who cannot tolerate TMP-SMX . (1.2)
Limitations of Use (1.3):
• Treatment of severe PCP (alveolar arterial oxygen diffusion gradient
[(A-a)DO 2] >45 mm Hg) with atovaquone oral suspension has not been studied.
• The efficacy of atovaquone oral suspension in subjects who are failing
therapy with TMP-SMX has also not been studied.
CONTRAINDICATIONS SECTION
4 CONTRAINDICATIONS
Atovaquone oral suspension is contraindicated in patients who develop or have a history of hypersensitivity reactions (e.g., angioedema, bronchospasm, throat tightness, urticaria) to atovaquone or any of the components of atovaquone oral suspension.
Known serious allergic/hypersensitivity reaction (e.g., angioedema, bronchospasm, throat tightness, urticaria) to atovaquone or any of the components of atovaquone oral suspension. (4)
WARNINGS AND PRECAUTIONS SECTION
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Limited Oral Absorption
Absorption of orally administered atovaquone oral suspension is limited but can be significantly increased when the drug is taken with food. Failure to administer atovaquone oral suspension with food may result in lower plasma atovaquone concentrations and may limit response to therapy. Consider therapy with other agents in patients who have difficulty taking atovaquone oral suspension with food or in patients who have gastrointestinal disorders that may limit absorption of oral medications [see Clinical Pharmacology(12.3)].
5.2 Hepatotoxicity
Cases of cholestatic hepatitis, elevated liver enzymes, and fatal liver
failure have been reported in patients treated with atovaquone [see Adverse Reactions(6.2)].
If treating patients with severe hepatic impairment, closely monitor patients
following administration of atovaquone oral suspension.
• Failure to administer atovaquone oral suspension with food may result in
lower plasma atovaquone concentrations and may limit response to therapy.
Patients with gastrointestinal disorders may have limited absorption resulting
in suboptimal atovaquone concentrations. (5.1)
• Hepatotoxicity: Elevated liver chemistry tests and cases of hepatitis and
fatal liver failure have been reported. (5.2)
ADVERSE REACTIONS SECTION
6 ADVERSE REACTIONS
The following adverse reaction is discussed in another section of the
labeling:
• Hepatotoxicity [see Warnings and Precautions(5.2)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse
reaction rates observed in the clinical trials of a drug cannot be directly
compared with rates in the clinical trials of another drug and may not reflect
the rates observed in practice.
Additionally, because many subjects who participated in clinical trials with
atovaquone had complications of advanced human immunodeficiency virus (HIV)
disease, it was often difficult to distinguish adverse reactions caused by
atovaquone from those caused by underlying medical conditions.
PCP Prevention Trials
In 2 clinical trials, atovaquone oral suspension was compared with dapsone or
aerosolized pentamidine in HIV-1-infected adolescent (13 to 18 years) and
adult subjects at risk of PCP (CD4 count <200 cells/mm 3or a prior episode of
PCP) and unable to tolerate TMP-SMX.
Dapsone Comparative Trial:In the dapsone comparative trial (n = 1,057), the
majority of subjects were white (64%), male (88%), and receiving prophylaxis
for PCP at randomization (73%); the mean age was 38 years. Subjects received
atovaquone oral suspension 1,500 mg once daily (n = 536) or dapsone 100 mg
once daily (n = 521); median durations of exposure were 6.7 and 6.5 months,
respectively. Adverse reaction data were collected only for adverse reactions
requiring discontinuation of treatment, which occurred at similar frequencies
in subjects treated with atovaquone oral suspension or dapsone (Table 1).
Among subjects taking neither dapsone nor atovaquone at enrollment (n = 487),
adverse reactions requiring discontinuation of treatment occurred in 43% of
subjects treated with dapsone and 20% of subjects treated with atovaquone oral
suspension. Gastrointestinal adverse reactions (nausea, diarrhea, and
vomiting) were more frequently reported in subjects treated with atovaquone
oral suspension (Table 1).
Table 1. Percentage (>2%) of Subjects with Selected Adverse Reactions
Requiring Discontinuation of Treatment in the Dapsone Comparative PCP
Prevention Trial
|
Adverse Reaction |
All Subjects | |
|
Atovaquone Oral Suspension |
Dapsone | |
|
Rash |
6.3 |
8.8 |
|
Nausea |
4.1 |
0.6 |
|
Diarrhea |
3.2 |
0.2 |
|
Vomiting |
2.2 |
0.6 |
Aerosolized Pentamidine Comparative Trial:In the aerosolized pentamidine
comparative trial (n = 549), the majority of subjects were white (79%), male
(92%), and were primary prophylaxis patients at enrollment (58%); the mean age
was 38 years. Subjects received atovaquone oral suspension once daily at a
dose of 750 mg (n = 188) or 1,500 mg (n = 175) or received aerosolized
pentamidine 300 mg every 4 weeks (n = 186); the median durations of exposure
were 6.2, 6.0, and 7.8 months, respectively. Table 2 summarizes the clinical
adverse reactions reported by ≥20% of the subjects receiving either the 1,500
mg dose of atovaquone oral suspension or aerosolized pentamidine.
Rash occurred more often in subjects treated with atovaquone oral suspension
(46%) than in subjects treated with aerosolized pentamidine (28%). Treatment-
limiting adverse reactions occurred in 25% of subjects treated with atovaquone
oral suspension 1,500 mg once daily and in 7% of subjects treated with
aerosolized pentamidine. The most frequent adverse reactions requiring
discontinuation of dosing in the group receiving atovaquone oral suspension
1,500 mg once daily were rash (6%), diarrhea (4%), and nausea (3%). The most
frequent adverse reaction requiring discontinuation of dosing in the group
receiving aerosolized pentamidine was bronchospasm (2%).
Table 2. Percentage (≥20%) of Subjects with Selected Adverse Reactions in
the Aerosolized Pentamidine Comparative PCP Prevention Trial
|
Adverse Reaction |
Atovaquone Oral Suspension |
Aerosolized |
|
Diarrhea |
42 |
35 |
|
Rash |
39 |
28 |
|
Headache |
28 |
22 |
|
Nausea |
26 |
23 |
|
Fever |
25 |
18 |
|
Rhinitis |
24 |
17 |
Other reactions occurring in ≥10% of subjects receiving the recommended dose
of atovaquone oral suspension (1,500 mg once daily) included vomiting,
sweating, flu syndrome, sinusitis, pruritus, insomnia, depression, and
myalgia.
PCP Treatment Trials
Safety information is presented from 2 clinical efficacy trials of the
atovaquone tablet formulation: 1) a randomized, double-blind trial comparing
atovaquone tablets with TMP-SMX in subjects with acquired immunodeficiency
syndrome (AIDS) and mild-to-moderate PCP [(A-a)DO 2] ≤45 mm Hg and PaO 2≥60 mm
Hg on room air; 2) a randomized, open-label trial comparing atovaquone tablets
with intravenous (IV) pentamidine isethionate in subjects with mild-to-
moderate PCP who could not tolerate trimethoprim or sulfa antimicrobials.
TMP-SMX Comparative Trial:In the TMP-SMX comparative trial (n = 408), the
majority of subjects were white (66%) and male (95%); the mean age was 36
years. Subjects received atovaquone 750 mg (three 250-mg tablets) 3 times
daily for 21 days or TMP 320 mg plus SMX 1,600 mg 3 times daily for 21 days;
median durations of exposure were 21 and 15 days, respectively.
Table 3 summarizes all clinical adverse reactions reported by ≥10% of the
trial population regardless of attribution. Nine percent of subjects who
received atovaquone and 24% of subjects who received TMP-SMX discontinued
therapy due to an adverse reaction. Among the subjects who discontinued, 4% of
subjects receiving atovaquone and 8% of subjects in the TMP-SMX group
discontinued therapy due to rash.
The incidence of adverse reactions with atovaquone oral suspension at the
recommended dose (750 mg twice daily) was similar to that seen with the tablet
formulation.
Table 3. Percentage (≥10%) of Subjects with Selected Adverse Reactions in
the TMP-SMX Comparative PCP Treatment Trial
|
Adverse Reaction |
Atovaquone Tablets |
TMP-SMX |
|
Rash (including maculopapular) |
23 |
34 |
|
Nausea |
21 |
44 |
|
Diarrhea |
19 |
7 |
|
Headache |
16 |
22 |
|
Vomiting |
14 |
35 |
|
Fever |
14 |
25 |
|
Insomnia |
10 |
9 |
Two percent of subjects treated with atovaquone and 7% of subjects treated
with TMP-SMX had therapy prematurely discontinued due to elevations in
ALT/AST.
Pentamidine Comparative Trial:In the pentamidine comparative trial (n = 174),
the majority of subjects in the primary therapy trial population (n = 145)
were white (72%) and male (97%); the mean age was 37 years. Subjects received
atovaquone 750 mg (three 250 mg tablets) 3 times daily for 21 days or a 3- to
4-mg/kg single pentamidine isethionate IV infusion daily for 21 days; the
median durations of exposure were 21 and 14 days, respectively.
Table 4 summarizes the clinical adverse reactions reported by ≥10% of the
primary therapy trial population regardless of attribution. Fewer subjects who
received atovaquone reported adverse reactions than subjects who received
pentamidine (63% vs. 72%). However, only 7% of subjects discontinued treatment
with atovaquone due to adverse reactions, while 41% of subjects who received
pentamidine discontinued treatment for this reason. Of the 5 subjects who
discontinued therapy with atovaquone, 3 reported rash (4%). Rash was not
severe in any subject. The most frequently cited reasons for discontinuation
of pentamidine therapy were hypoglycemia (11%) and vomiting (9%).
Table 4. Percentage (≥10%) of Subjects with Selected Adverse Reactions in
the Pentamidine Comparative PCP Treatment Trial (Primary Therapy Group)
|
Adverse Reaction |
Atovaquone Tablets |
Pentamidine |
|
Fever |
40 |
25 |
|
Nausea |
22 |
37 |
|
Rash |
22 |
13 |
|
Diarrhea |
21 |
31 |
|
Insomnia |
19 |
14 |
|
Headache |
18 |
28 |
|
Vomiting |
14 |
17 |
|
Cough |
14 |
1 |
|
Sweat |
10 |
3 |
|
Monilia, oral |
10 |
3 |
Laboratory abnormality was reported as the reason for discontinuation of treatment in 2 of 73 subjects (3%) who received atovaquone, and in 14 of 71 subjects (20%) who received pentamidine. One subject (1%) receiving atovaquone had elevated creatinine and BUN levels and 1 subject (1%) had elevated amylase levels. In this trial, elevated levels of amylase occurred in subjects (8% versus 4%) receiving atovaquone tablets or pentamidine, respectively.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of atovaquone oral suspension. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders
Methemoglobinemia, thrombocytopenia.
Immune System Disorders
Hypersensitivity reactions including angioedema, bronchospasm, throat
tightness, and urticaria.
Eye Disorders
Vortex keratopathy.
Gastrointestinal Disorders
Pancreatitis.
Hepatobiliary Disorders
Hepatitis, fatal liver failure.
Skin and Subcutaneous Tissue Disorders
Erythema multiforme, Stevens-Johnson syndrome, and skin desquamation.
Renal and Urinary Disorders
Acute renal impairment.
• PCP Prevention: The most frequent adverse reactions (≥25% that required
discontinuation) were diarrhea, rash, headache, nausea, and fever. (6.1)
• PCP Treatment: The most frequent adverse reactions (≥14% that required
discontinuation) were rash (including maculopapular), nausea, diarrhea,
headache, vomiting, and fever. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Hetero Labs Limited at 1-866-495-1995 or FDA at 1-800-FDA-1088 orwww.fda.gov/medwatch.
DRUG INTERACTIONS SECTION
7 DRUG INTERACTIONS
7.1 Rifampin/Rifabutin
Concomitant administration of rifampin or rifabutin and atovaquone oral suspension is known to reduce atovaquone concentrations [see Clinical Pharmacology ( 12.3)]. Concomitant administration of atovaquone oral suspension and rifampin or rifabutin is not recommended.
7.2 Tetracycline
Concomitant administration of tetracycline and atovaquone oral suspension has been associated with a reduction in plasma concentrations of atovaquone [see Clinical Pharmacology ( 12.3)]. Caution should be used when prescribing tetracycline concomitantly with atovaquone oral suspension. Monitor patients for potential loss of efficacy of atovaquone if coadministration is necessary.
7.3 Metoclopramide
Metoclopramide may reduce the bioavailability of atovaquone and should be used only if other antiemetics are not available [see Clinical Pharmacology(12.3)].
7.4 Indinavir
Concomitant administration of atovaquone and indinavir did not result in any change in the steady-state AUC and C maxof indinavir but resulted in a decrease in the C troughof indinavir [see Clinical Pharmacology (12.3)]. Caution should be exercised when prescribing atovaquone oral suspension with indinavir due to the decrease in trough concentrations of indinavir. Monitor patients for potential loss of efficacy of indinavir if coadministration with atovaquone oral suspension is necessary.
• Concomitant administration of rifampin or rifabutin reduces atovaquone
concentrations; concomitant use with atovaquone oral suspension is not
recommended. (7.1)
• Concomitant administration of tetracycline reduces atovaquone
concentrations; use caution when coadministering. Monitor patients for
potential loss of efficacy of atovaquone if coadministration of tetracycline
is necessary. (7.2)
• Concomitant administration with metoclopramide reduces atovaquone
concentrations; administer concomitantly only if other antiemetics are not
available. (7.3)
• Concomitant administration of indinavir reduces indinavir trough
concentrations; use caution when coadministering. Monitor patients for
potential loss of efficacy of indinavir if coadministration is necessary.
(7.4)
DOSAGE FORMS & STRENGTHS SECTION
3 DOSAGE FORMS AND STRENGTHS
Atovaquone oral suspension, USP is a yellow homogenous suspension containing 750 mg of atovaquone USP per 5 mL. Atovaquone oral suspension, USP is supplied in 210 mL bottles.
Oral suspension: 750 mg per 5 mL (3)
DOSAGE & ADMINISTRATION SECTION
2 DOSAGE AND ADMINISTRATION
2.1 Dosage for the Prevention of P. jiroveciiPneumonia
The recommended oral dosage is 1,500 mg (10 mL) once daily administered with food.
2.2 Dosage for the Treatment of Mild-to-Moderate P. jiroveciiPneumonia
The recommended oral dosage is 750 mg (5 mL) twice daily (total daily dose = 1,500 mg) administered with food for 21 days.
2.3 Important Administration Instructions
Administer atovaquone oral suspension with food to avoid low plasma atovaquone
concentrations that may limit response to therapy [see Warnings and Precautions (5.1), Clinical Pharmacology ( 12.3)].
Atovaquone Oral Suspension Bottle
Shake bottle gently before administering the recommended dosage.
• Prevention of PCP: 1,500 mg (10 mL) once daily with food (2.1)
• Treatment of PCP: 750 mg (5 mL) twice daily with food for 21 days (2.2)
• Supplied in Bottles: Shake bottle gently before use. (2.3)
USE IN SPECIFIC POPULATIONS SECTION
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data from postmarketing experience with use of atovaquone in
pregnant women are insufficient to identify a drug-associated risk for major
birth defects, miscarriage, or adverse maternal or fetal outcomes. Pregnant
women with HIV who are infected with PCP are at increased risk of adverse
pregnancy outcomes (see Clinical Considerations).Atovaquone given orally by
gavage to pregnant rats and rabbits during organogenesis did not cause fetal
malformations at plasma concentrations up to 3 times and 0.5 times,
respectively, the estimated human exposure based on steady-state plasma
concentrations (see Data).
The estimated background risk of major birth defects and miscarriage for the
indicated population is unknown. All pregnancies have a background risk of
birth defect, loss, or other adverse outcomes. In the U.S. general population,
the background risk of major birth defects and miscarriage in clinically
recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk:Pregnant women with HIV
who are infected with PCP are at increased risk of severe illness and maternal
death associated with PCP compared with non-pregnant women.
DataAnimal Data:Atovaquone administered in oral doses of 250, 500, and 1,000
mg/kg/day to pregnant rats during organogenesis (Gestation Day [GD] 6 to GD15)
did not cause maternal or embryo-fetal toxicity at doses up to 1,000 mg/kg/day
corresponding to maternal plasma concentrations approximately 3 times the
estimated human exposure during the treatment of PCP based on steady-state
plasma concentrations. In pregnant rabbits, atovaquone administered in oral
doses of 300, 600, and 1,200 mg/kg/day during organogenesis (GD6 to GD18)
caused decreased fetal body length at a maternally toxic dose of 1,200
mg/kg/day corresponding to a plasma concentration that is approximately 0.5
times the estimated human exposure based on steady-state plasma
concentrations. In a pre- and post-natal study in rats, atovaquone
administered in oral doses of 250, 500, and 1,000 mg/kg/day from GD15 until
Lactation Day (LD) 20 did not impair the growth or developmental effects in
first generation offspring at doses up to 1,000 mg/kg/day corresponding to
approximately 3 times the estimated human exposure based on steady-state
plasma concentrations during the treatment of PCP. Atovaquone crossed the
placenta and was present in fetal rat and rabbit tissue.
8.2 Lactation
Risk Summary
The Centers for Disease Control and Prevention recommend that HIV-1–infected
mothers not breastfeed their infants to avoid risking postnatal transmission
of HIV-1. There are no data on the presence of atovaquone in human milk, the
effects on the breastfed child, or the effects on milk production. Atovaquone
was detected in rat milk when lactating rats were administered oral atovaquone
(see Data).When a drug is present in animal milk, it is likely the drug will
be present in human milk. Because of the potential for HIV-1 transmission to
HIV-negative infants, instruct mothers with HIV-1 not to breastfeed if they
are taking atovaquone for the prevention or treatment of PCP.
Data
In a rat study with doses of 10 and 250 mg/kg given orally by gavage on
postpartum Day 11, atovaquone concentrations in the milk were 30% of the
concurrent atovaquone concentrations in the maternal plasma at both doses. The
concentration of drug in animal milk does not necessarily predict the
concentration of drug in human milk.
8.4 Pediatric Use
Evidence of safety and effectiveness in pediatric patients (aged 12 years and
younger) has not been established. In a trial of atovaquone oral suspension
administered once daily with food for 12 days to 27 HIV-1-infected,
asymptomatic infants and children aged between 1 month and 13 years, the
pharmacokinetics of atovaquone were age-dependent. The average steady-state
plasma atovaquone concentrations in the 24 subjects with available
concentration data are shown in Table 5.
Table 5. Average Steady-state Plasma Atovaquone Concentrations in Pediatric
Subjects
|
Age |
Dose ofAtovaquone OralSuspension | ||
|
10 mg/kg |
30 mg/kg |
45 mg/kg | |
|
Average Cssin mcg/mL (mean ± SD) | |||
|
1 to 3 months |
5.9 |
27.8 ± 5.8 |
_ |
|
5.7 ± 5.1 |
9.8 ± 3.2 |
15.4 ± 6.6 |
|
16.8 ± 6.4 |
37.1 ± 10.9 |
_ |
C ss= Concentration at steady state.
8.5 Geriatric Use
Clinical trials of atovaquone did not include sufficient numbers of subjects aged 65 years and older to determine whether they respond differently from younger subjects.
Lactation: Breastfeeding is not recommended in mothers with HIV-1 infection due to the potential for HIV-1 transmission. (8.2)
OVERDOSAGE SECTION
10 OVERDOSAGE
Overdoses up to 31,500 mg of atovaquone have been reported. In one such patient who also took an unspecified dose of dapsone, methemoglobinemia occurred. Rash has also been reported after overdose. There is no known antidote for atovaquone, and it is currently unknown if atovaquone is dialyzable.
DESCRIPTION SECTION
11 DESCRIPTION
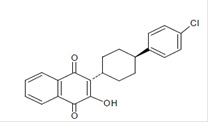
Atovaquone oral suspension is a quinone antimicrobial drug. The chemical name of atovaquone is 1,4-Naphthalenedione, 2-[4-(4-chlorophenyl)cyclohexyl]-3-hydroxy-, trans. Atovaquone USP is a yellow colored powder that is freely soluble in tetrahydrofuran, soluble in chloroform and sparingly soluble in acetone. It has a molecular weight of 366.84 and the molecular formula C 22H 19ClO 3. The compound has the following structural formula:
Atovaquone oral suspension, USP is a formulation of micro-fine particles of
atovaquone USP.
Each 5 mL of atovaquone oral suspension, USP contains 750 mg of atovaquone USP
and the inactive ingredients benzyl alcohol, flavor, hypromellose, poloxamer,
purified water, saccharin sodium, and xanthan gum.
CLINICAL PHARMACOLOGY SECTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Atovaquone is a quinone antimicrobial drug [see Microbiology (12.4)].
12.2 Pharmacodynamics
Relationship between Plasma Atovaquone Concentrations and Clinical Outcome
In a comparative clinical trial, HIV/AIDS subjects received atovaquone tablets
750 mg 3 times daily or TMP-SMX for treatment of mild-to-moderate PCP for 21
days [see Clinical Studies (14.2)]; the relationship between atovaquone plasma
concentrations and successful treatment outcome from 113 of these subjects for
whom both steady-state drug concentrations and outcome data were available is
shown in Table 6.
Table 6. Relationship between Plasma Atovaquone Concentrations and
Successful Treatment Outcome
|
Steady-State Plasma Atovaquone |
** Successful Treatment****a** |
|
0 to <5 |
0/6 (0%) |
|
5 to <10 |
18/26 (69%) |
|
10 to <15 |
30/38 (79%) |
|
15 to <20 |
18/19 (95%) |
|
≥20 |
24/24 (100%) |
a Successful treatment outcome was defined as improvement in clinical and
respiratory measures persisting at least 4 weeks after cessation of therapy.
Improvement in clinical and respiratory measures was assessed using a
composite of parameters that included oral body temperature, respiratory rate,
and severity scores for cough, dyspnea, and chest pain/tightness.
Cardiac Effects
The effect of atovaquone oral suspension on the QT interval is unknown in
humans.
12.3 Pharmacokinetics
Plasma atovaquone concentrations do not increase proportionally with dose following ascending repeat-dose administration of atovaquone oral suspension in healthy subjects. When atovaquone oral suspension was administered with food at dosage regimens of 500 mg once daily, 750 mg once daily, and 1,000 mg once daily, mean (±SD) steady-state plasma atovaquone concentrations were 11.7 ± 4.8, 12.5 ± 5.8, and 13.5 ± 5.1 mcg/mL, respectively. The corresponding mean (±SD) C maxconcentrations were 15.1 ± 6.1, 15.3 ± 7.6, and 16.8 ± 6.4 mcg/mL.
Absorption
Atovaquone is a highly lipophilic compound with low aqueous solubility. The mean (±SD) absolute bioavailability of atovaquone from a 750-mg dose of atovaquone oral suspension administered under fed conditions in 9 HIV-1–infected (CD4 >100 cells/mm 3) volunteers was 47% ± 15%.
Effect of Food:Administering atovaquone oral suspension with food enhances atovaquone bioavailability. Sixteen healthy subjects received a single 750 mg dose of atovaquone oral suspension after an overnight fast and following a meal (23 g fat: 610 kCal). The mean (±SD) atovaquone AUC under fasting and fed conditions were 324 ± 115 and 801 ± 320, h.mcg/mL, respectively, representing a 2.6 ± 1.0-fold increase.
Distribution
Following IV administration of atovaquone, the mean (±SD) volume of distribution at steady state (Vdss) was 0.60 ± 0.17 L/kg (n = 9). Atovaquone is extensively bound to plasma proteins (99.9%) over the concentration range of 1 to 90 mcg/mL. In 3 HIV-1– infected children who received 750 mg atovaquone as the tablet formulation 4 times daily for 2 weeks, the cerebrospinal fluid concentrations of atovaquone were 0.04, 0.14, and 0.26 mcg/mL, representing less than 1% of the plasma concentration.
Elimination
The mean (±SD) half-life of atovaquone was 62.5 ± 35.3 hours after IV administration and ranged from 67.0 ± 33.4 to 77.6 ± 23.1 hours following administration of atovaquone oral suspension.
Metabolism:The metabolism of atovaquone is unknown.
Excretion:Following oral administration of 14C-labelled atovaquone to healthy subjects, greater than 94% of the dose was recovered as unchanged atovaquone in the feces over 21 days.
Specific Populations
Patients with Hepatic or Renal Impairment:The pharmacokinetics of atovaquone have not been studied in patients with hepatic or renal impairment.
HIV-Infected Subjects:When atovaquone oral suspension was administered to 5 HIV-1–infected subjects at a dose of 750 mg twice daily, the mean (±SD) steady-state plasma atovaquone concentration was 21.0 ± 4.9 mcg/mL and mean (±SD) C maxwas 24.0 ± 5.7 mcg/mL. The mean (±SD) minimum plasma atovaquone concentration (C min) associated with the 750 mg twice-daily regimen was 16.7 ± 4.6 mcg/mL.
In an open-label PCP trial in 18 HIV-1–infected subjects, administration of atovaquone oral suspension 750 mg twice daily with meals resulted in a mean (±SD) steady-state plasma atovaquone concentration of 22.0 ± 10.1 mcg/mL.
The mean (±SD) plasma clearance of atovaquone following IV administration in 9 HIV-1– infected subjects was 10.4 ± 5.5 mL/min (0.15 ± 0.09 mL/min/kg).
Drug Interaction Studies
Rifampin/Rifabutin:In a trial with 13 HIV-1–infected volunteers, the oral administration of rifampin 600 mg every 24 hours with atovaquone oral suspension 750 mg every 12 hours resulted in a 52% ± 13% decrease in the mean (±SD) steady-state plasma atovaquone concentration and a 37% ± 42% increase in the mean (±SD) steady-state plasma rifampin concentration. The half-life of atovaquone decreased from 82 ± 36 hours when administered without rifampin to 50 ± 16 hours with rifampin. In a trial of 24 healthy volunteers, the oral administration of rifabutin 300 mg once daily with atovaquone oral suspension 750 mg twice daily resulted in a 34% decrease in the mean steady-state plasma atovaquone concentration and a 19% decrease in the mean steady-state plasma rifabutin concentration.
Tetracycline:Concomitant treatment with tetracycline has been associated with a 40% reduction in plasma concentrations of atovaquone. Metoclopramide:Concomitant treatment with metoclopramide has been associated with a 50% reduction in steady-state atovaquone plasma concentrations.
Indinavir:Concomitant administration of atovaquone (750 mg twice daily with food for 14 days) and indinavir (800 mg three times daily without food for 14 days) did not result in any change in the steady-state AUC and C maxof indinavir, but resulted in a decrease in the C troughof indinavir (23% decrease [90% CI: 8%, 35%]).
Trimethoprim/Sulfamethoxazole (TMP-SMX):Concomitant administration of atovaquone oral suspension 500 mg once daily (not the approved dosage) and TMP-SMX in 6 HIV-infected adult subjects did not result in significant changes in either atovaquone or TMP- SMX exposure.
Zidovudine:The administration of atovaquone tablets 750 mg every 12 hours with zidovudine 200 mg every 8 hours to 14 HIV-1 infected subjects resulted in a 24% ± 12% decrease in zidovudine apparent oral clearance, leading to a 35% ± 23% increase in plasma zidovudine AUC. The glucuronide metabolite:parent ratio decreased from a mean of 4.5 when zidovudine was administered alone to 3.1 when zidovudine was administered with atovaquone tablets. This effect is minor and would not be expected to produce clinically significant events. Zidovudine had no effect on atovaquone pharmacokinetics.
12.4 Microbiology
Mechanism of Action
Atovaquone is a hydroxy-1,4-naphthoquinone, an analog of ubiquinone, with
antipneumocystis activity. The mechanism of action against Pneumocystis
jiroveciihas not been fully elucidated. In Plasmodiumspecies, the site of
action appears to be the cytochrome bc1complex (Complex III). Several
metabolic enzymes are linked to the mitochondrial electron transport chain via
ubiquinone. Inhibition of electron transport by atovaquone results in indirect
inhibition of these enzymes. The ultimate metabolic effects of such blockade
may include inhibition of nucleic acid and adenosine triphosphate (ATP)
synthesis.
Antimicrobial Activity
Atovaquone is active against P. jirovecii [see Clinical Studies (14)].
Resistance
Phenotypic resistance to atovaquone in vitrohas not been demonstrated for P.
jirovecii. However, in 2 subjects who developed PCP after prophylaxis with
atovaquone, DNA sequence analysis identified mutations in the predicted amino
acid sequence of P. jiroveciicytochrome b(a likely target site for
atovaquone). The clinical significance of this is unknown.
NONCLINICAL TOXICOLOGY SECTION
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies in rats were negative; 24-month studies in mice (dosed with 50, 100, or 200 mg/kg/day) showed treatment-related increases in incidence of hepatocellular adenoma and hepatocellular carcinoma at all doses tested, which correlated with 1.4 to 3.6 times the average steady-state plasma concentrations in humans during acute treatment of PCP. Atovaquone was negative with or without metabolic activation in the Ames Salmonellamutagenicity assay, the mouse lymphoma mutagenesis assay, and the cultured human lymphocyte cytogenetic assay. No evidence of genotoxicity was observed in the in vivo mouse micronucleus assay.
Impairment of Fertility
Atovaquone administered by oral gavage in doses of 100, 300, or 1,000
mg/kg/day to adult male rats from 73 days prior to mating until 20 days after
mating and to adult female rats from 14 days prior to mating until LD20 did
not impair male or female fertility or early embryonic development at doses up
to 1,000 mg/kg/day corresponding to plasma exposures of approximately 3 times
the estimated human exposure based on steady-state plasma concentrations.
CLINICAL STUDIES SECTION
14 CLINICAL STUDIES
14.1 Prevention of PCP
The indication for prevention of PCP is based on the results of 2 clinical
trials comparing atovaquone oral suspension with dapsone or aerosolized
pentamidine in HIV-1-infected adolescent (aged 13 to 18 years) and adult
subjects at risk of PCP (CD4 count <200 cells/mm 3or a prior episode of PCP)
and unable to tolerate TMP-SMX.
Dapsone Comparative Trial
This open-label trial enrolled 1,057 subjects, randomized to receive
atovaquone oral suspension 1,500 mg once daily (n = 536) or dapsone 100 mg
once daily (n = 521). The majority of subjects were white (64%), male (88%),
and receiving prophylaxis for PCP at randomization (73%); the mean age was 38
years. Median follow-up was 24 months. Subjects randomized to the dapsone arm
who were seropositive for Toxoplasma gondiiand had a CD4 count <100 cells/mm
3also received pyrimethamine and folinic acid. PCP event rates are shown in
Table 7. Mortality rates were similar.
Aerosolized Pentamidine Comparative Trial
This open-label trial enrolled 549 subjects, randomized to receive atovaquone
oral suspension 1,500 mg once daily (n = 175), atovaquone oral suspension 750
mg once daily (n = 188), or aerosolized pentamidine 300 mg once monthly (n =
186). The majority of subjects were white (79%), male (92%), and were primary
prophylaxis patients at enrollment (58%); the mean age was 38 years. Median
follow-up was 11.3 months. The results of the PCP event rates appear in Table
7. Mortality rates were similar among the groups.
**Table 7. Confirmed or Presumed/Probable PCP Events (As-Treated Analysis)
**a
|
Assessment |
Trial 1 |
Trial 2 | |||
|
Atovaquone Oral****Suspension |
Dapsone |
Atovaquone Oral****Suspension 750 mg/day |
Atovaquone Oral****Suspension 1,500 mg/day (n = 172) |
Aerosolized Pentamidine 300 mg/month (n = 169) | |
|
% |
15 |
19 |
23 |
18 |
17 |
|
Relative Risk b |
0.77 |
1.47 |
1.14 |
a Those events occurring during or within 30 days of stopping assigned
treatment.
b Relative risk <1 favors atovaquone and values >1 favor comparator. Trial
results did not show superiority of atovaquone to the comparator.
c The confidence level of the interval for the dapsone comparative trial was
95% and for the pentamidine comparative trial was 97.5%.
An analysis of all PCP events (intent-to-treat analysis) for both trials
showed results similar to those shown in Table 7.
14.2 Treatment of PCP
The indication for treatment of mild-to-moderate PCP is based on the results of 2 efficacy trials: a randomized, double-blind trial comparing atovaquone tablets with TMP-SMX in subjects with HIV/AIDS and mild-to-moderate PCP (defined in the protocol as [(A-a)DO 2] ≤45 mm Hg and PaO 2≥60 mm Hg on room air) and a randomized open-label trial comparing atovaquone tablets with IV pentamidine isethionate in subjects with mild-to-moderate PCP who could not tolerate trimethoprim or sulfa antimicrobials. Both trials were conducted with the tablet formulation using 750 mg 3 times daily. Results from these efficacy trials established a relationship between plasma atovaquone concentrations and successful outcome. Successful outcome was defined as improvement in clinical and respiratory measures persisting at least 4 weeks after cessation of therapy [see Clinical Pharmacology (12.2)].
TMP-SMX Comparative Trial
This double-blind, randomized trial compared the safety and efficacy of
atovaquone tablets with that of TMP-SMX for the treatment of subjects with
HIV/AIDS and histologically confirmed PCP. Only subjects with mild-to-moderate
PCP were eligible for enrollment.
A total of 408 subjects were enrolled into the trial. The majority of subjects
were white (66%) and male (95%); the mean age was 36 years. Eighty-six
subjects without histologic confirmation of PCP were excluded from the
efficacy analyses. Of the 322 subjects with histologically confirmed PCP, 160
were randomized to receive 750 mg atovaquone (three 250 mg tablets) 3 times
daily for 21 days and 162 were randomized to receive 320 mg TMP plus 1,600 mg
SMX 3 times daily for 21 days. Therapy success was defined as improvement in
clinical and respiratory measures persisting at least 4 weeks after cessation
of therapy. Improvement in clinical and respiratory measures was assessed
using a composite of parameters that included oral body temperature,
respiratory rate, severity scores for cough, dyspnea, and chest
pain/tightness. Therapy failures included lack of response, treatment
discontinuation due to an adverse experience, and unevaluable.
There was a significant difference ( P= 0.03) in mortality rates between the
treatment groups favoring TMP-SMX. Among the 322 subjects with confirmed PCP,
13 of 160 (8%) subjects treated with atovaquone and 4 of 162 (2.5%) subjects
receiving TMP-SMX died during the 21-day treatment course or 8-week follow-up
period. In the intent-to-treat analysis for all 408 randomized subjects, there
were 16 (8%) deaths among subjects treated with atovaquone and 7 (3.4%) deaths
among subjects treated with TMP-SMX ( P= 0.051). Of the 13 subjects with
confirmed PCP and treated with atovaquone who died, 4 died of PCP and 5 died
with a combination of bacterial infections and PCP; bacterial infections did
not appear to be a factor in any of the 4 deaths among TMP-SMX-treated
subjects.
A correlation between plasma atovaquone concentrations and death demonstrated
that subjects with lower plasma concentrations were more likely to die. For
those subjects for whom Day 4 plasma atovaquone concentration data are
available, 5 (63%) of 8 subjects with concentrations <5 mcg/mL died during
participation in the trial. However, only 1 (2%) of the 49 subjects with Day 4
plasma atovaquone concentrations ≥5 mcg/mL died.
Sixty-two percent of subjects on atovaquone and 64% of subjects on TMP-SMX
were classified as protocol-defined therapy successes (Table 8).
Table 8. Outcome of Treatment for PCP-Positive Subjects Enrolled in the TMP-
SMX Comparative Trial
|
Outcome of Therapy****a |
Number of Subjects (%) | |||
|
Atovaquone****Tablets |
TMP-SMX | |||
|
Therapy success |
99 |
62% |
103 |
64% |
|
Therapy failure due to: |
28 |
17% |
10 |
6% |
|
Required alternate PCP therapy during trial |
55 |
34% |
55 |
34% |
aAs defined by the protocol and described in trial description above.
The failure rate due to lack of response was significantly higher for subjects
receiving atovaquone, while the failure rate due to an adverse reaction was
significantly higher for subjects receiving TMP-SMX.
Pentamidine Comparative Trial
This unblinded, randomized trial was designed to compare the safety and
efficacy of atovaquone with that of pentamidine for the treatment of
histologically-confirmed mild or moderate PCP in subjects with HIV/AIDS.
Approximately 80% of the subjects either had a history of intolerance to
trimethoprim or sulfa antimicrobials (the primary therapy group) or were
experiencing intolerance to TMP-SMX with treatment of an episode of PCP at the
time of enrollment in the trial (the salvage treatment group). A total of 174
subjects were enrolled into the trial. Subjects were randomized to receive
atovaquone 750 mg (three 250 mg tablets) 3 times daily for 21 days or
pentamidine isethionate 3- to 4-mg/kg single IV infusion daily for 21 days.
The majority of subjects were white (72%) and male (97%); the mean age was
approximately 37 years. Thirty-nine subjects without histologic confirmation
of PCP were excluded from the efficacy analyses. Of the 135 subjects with
histologically-confirmed PCP, 70 were randomized to receive atovaquone and 65
to pentamidine. One hundred and ten (110) of these were in the primary therapy
group and 25 were in the salvage therapy group. One subject in the primary
therapy group randomized to receive pentamidine did not receive trial
medication.
There was no difference in mortality rates between the treatment groups. Among
the 135 subjects with confirmed PCP, 10 of 70 (14%) subjects receiving
atovaquone and 9 of 65 (14%) subjects receiving pentamidine died during the
21-day treatment course or 8-week follow-up period. In the intent-to-treat
analysis for all subjects, there were 11 (12.5%) deaths among those treated
with atovaquone and 12 (14%) deaths among those treated with pentamidine.
Among subjects for whom Day 4 plasma atovaquone concentrations were available,
3 of 5 (60%) subjects with concentrations <5 mcg/mL died during participation
in the trial. However, only 2 of 21 (9%) subjects with Day 4 plasma
concentrations ≥5 mcg/mL died. The therapeutic outcomes for the 134 subjects
who received trial medication in this trial are presented in Table 9.
Table 9. Outcome of Treatment for PCP-Positive Subjects (%) Enrolled in the
Pentamidine Comparative Trial
|
Outcome of Therapy |
Primary Treatment |
Salvage Treatment | ||||||
|
Atovaquone**(n = 56)** |
Pentamidine |
Atovaquone**(n = 14)** |
Pentamidine | |||||
|
Therapy success |
32 |
57% |
21 |
40% |
13 |
93% |
7 |
64% |
|
Therapy failure due to: |
16 |
29% |
9 |
17% |
0 |
7% |
0 |
27% |
|
Required alternate PCP therapy during trial |
19 |
34% |
29 |
55% |
0 |
4 |
36% |
HOW SUPPLIED SECTION
16 HOW SUPPLIED/STORAGE AND HANDLING
Atovaquone oral suspension, USP is a yellow homogenous suspension containing
750 mg atovaquone USP per 5 mL.
• Bottle of 210 mL with child-resistant cap (NDC 31722-629-21). Store at 15°
to 25°C (59° to 77°F). Do not freeze. Dispense in tight container as defined
in USP.
INFORMATION FOR PATIENTS SECTION
17 PATIENT COUNSELING INFORMATION
Administration Instructions
Instruct patients to:
• Ensure the prescribed dose of atovaquone oral suspension is taken as
directed.
• Take their daily doses of atovaquone oral suspension with food, as food will
significantly improve the absorption of the drug.
• Shake atovaquone oral suspension gently before use each time.
Lactation
Instruct mothers with HIV-1 infection not to breastfeed because HIV-1 can be
passed to the baby in the breast milk [see Use in Specific Populations (8.2)].
Trademarks are owned or licensed to the GSK group of companies.
DISTRIBUTED BY:
ATLANTIC BIOLOGICALS CORP.
MIAMI, FL 33179
