Camphor 4% Plus Menthol 10%
59088-259-07 Camphor 4% + Menthol 10%
3cab26e4-e377-f26a-e063-6394a90a17af
HUMAN OTC DRUG LABEL
Aug 20, 2025
PureTek Corporation
DUNS: 785961046
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Camphor 4% Plus Menthol 10%
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (16)
Drug Labeling Information
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Camphor 4 % + Menthol 10%
Manufactured by:
PureTek Corporation
Panorama City, CA 91402
For questions or information
call toll-free:
877-921-7873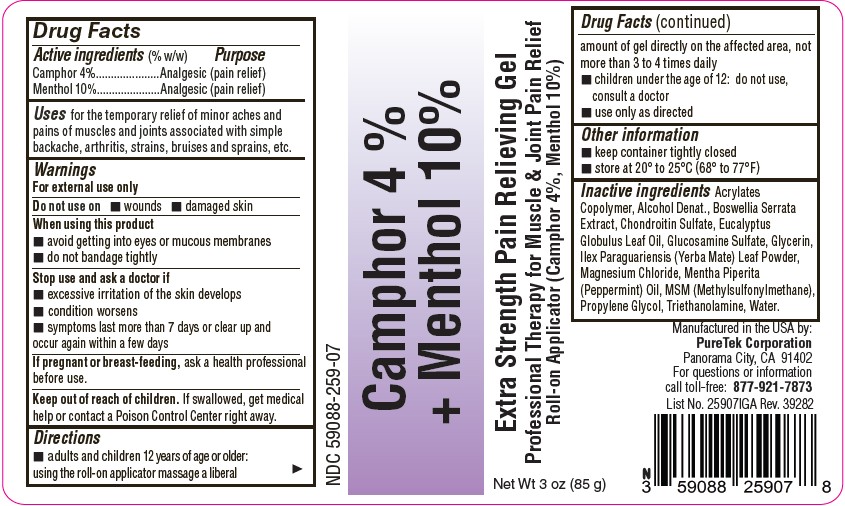
INDICATIONS & USAGE SECTION
Uses
for the temporary relief of minor aches and pains of muscles and joints associated with simple backache, arthritis, strains, bruises and sprains, etc.
OTC - ACTIVE INGREDIENT SECTION
Active Ingredients
Camphor 4%
Menthol 10%
OTC - PURPOSE SECTION
Purpose
Analgesic (pain relief)
WARNINGS SECTION
Warnings
For external use only
Do not use on
- wounds
- damaged skin
When using this product
- avoid getting into eyes or mucous membranes
- do not bandage tightly
Stop use and ask a doctor if
- excessive irritation of the skin develops
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
If pregnant or breast-feeding
ask a health professional before use.
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away.
DOSAGE & ADMINISTRATION SECTION
Directions
- adults and children 12 years of age or older: using the roll-on applicator massage a liberal amount of gel directly on the affected area, not more than 3 to 4 times daily.
- children under the age of 12: do not use, consult a doctor
- use only as directed
STORAGE AND HANDLING SECTION
Other information
- keep container tightly closed
- store at 20° to 25°C (68° to 77°F)
INACTIVE INGREDIENT SECTION
Inactive ingredients
Acrylates Copolymer, Alcohol Denat., Boswellia Serrata Extract, Chondroitin Sulfate, Eucalyptus Globulus Leaf Oil, Glucosamine Sulfate, Glycerin, Ilex Paraguariensis (Yerba Mate) Leaf Powder, Magnesium Chloride, Mentha Piperita (Peppermint) Oil, MSM (Methylsulfonylmethane), Propylene Glycol, Triethanolamine, Water.
