Ultra Strength
Kroger Ultra Strength 72 Chewable Tablets Drug Facts
2931d427-b2be-4db9-a58d-ff89c221ff51
HUMAN OTC DRUG LABEL
Aug 1, 2025
KROGER COMPANY
DUNS: 006999528
Products 1
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Calcium Carbonate
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (7)
Drug Labeling Information
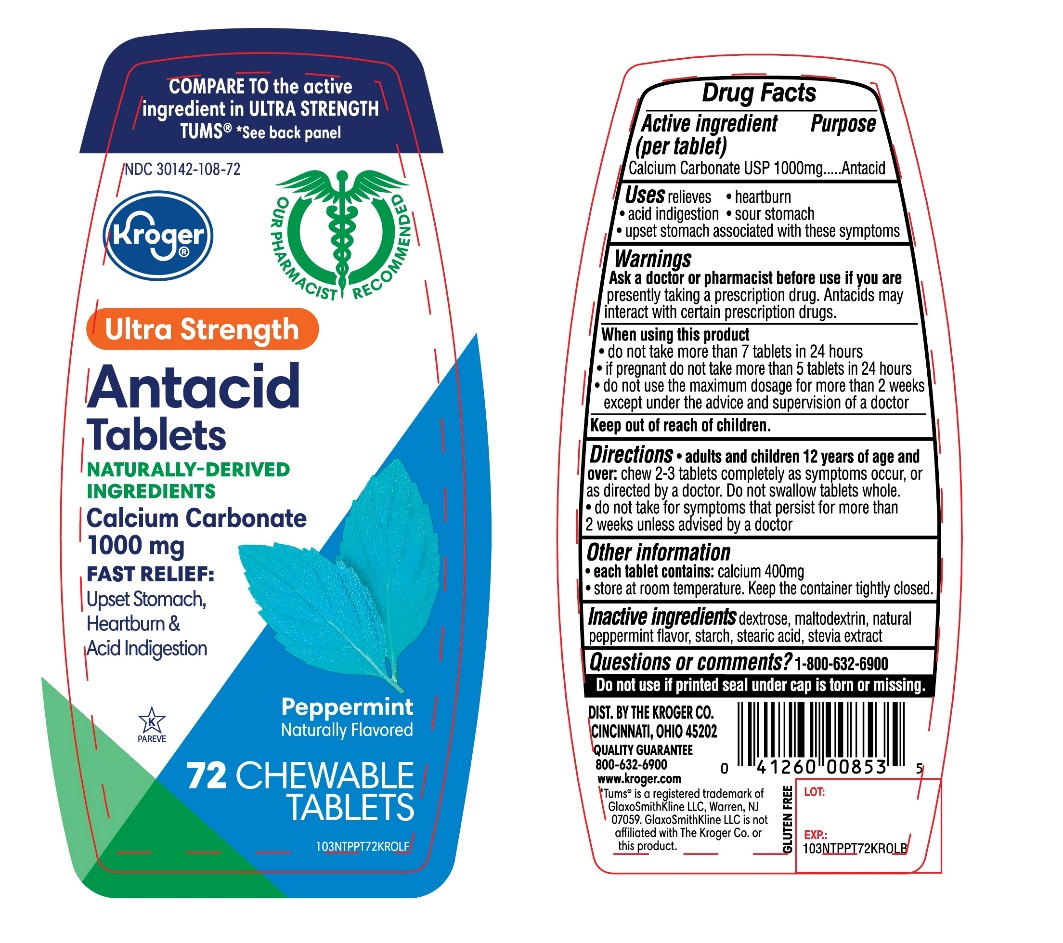
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Package/Label Principal Display Panel
Kroger**®**
**NDC#**30142-108-72
COMPARE TO the active ingredient in Ultra Strength TUMS**®**
*See back panel
Ultra Strength
Antacid Tablets
NATURALLY-DERIVED INGREDIENTS
Calcium Carbonate 1000 mg
**FAST RELIEF:Upset Stomach,**Heartburn and Acid Indigestion
Peppermint
Naturally Flavored
GLUTEN FREE
72 CHEWABLE TABLETS
Our Pharmacists Recommend
K PAREVE
DISTRIBUTED BY: THE KROGER CO. CINCINNATI, OHIO 45202
QUALITY GUARANTEE
800-632-6900
Tums® is a registered trademark of GlaxoSmithKline LLC, Warren, NJ 07059. GlaxoSmithKline LLC is not affiliated with The Kroger Co. or this product.
INDICATIONS & USAGE SECTION
Uses
relieves
- heartburn
- acid indigestion
- sour stomach
- upset stomach associated with these symptoms
OTC - ACTIVE INGREDIENT SECTION
Active ingredient (per tablet)
Calcium Carbonate USP 1000mg
OTC - PURPOSE SECTION
Purpose
Antacid
WARNINGS SECTION
Warnings
OTC - ASK DOCTOR SECTION
Ask a doctor or pharmacist before use if you are
presently taking a prescription drug. Antacids may interact with certain prescription drugs.
OTC - WHEN USING SECTION
When using this product
- do not take more than 7 tablets in 24 hours
- If pregnant do not take more than 5 tablets in 24 hours
- do not use the maximum dosage for more than 2 weeks except under the advice and supervision of a doctor
OTC - KEEP OUT OF REACH OF CHILDREN SECTION
Keep out of reach of children.
DOSAGE & ADMINISTRATION SECTION
Directions
***adults and children 12years of age and over:**chew 2-3 tablets completely as symptoms occur, or as directed by a doctor. Do not swallow tablets whole.
- do not take for symptoms that persist for more than 2 weeks unless advised by a doctor.
STORAGE AND HANDLING SECTION
Other information
***each tablets contains:**elemental calcium 400mg
- Store at room temperature. Keep the container tightly closed.
INACTIVE INGREDIENT SECTION
Inactive ingredients
dextrose, maltodextrin, natural peppermint flavor, starch, stearic acid, stevia extract.
OTC - QUESTIONS SECTION
Questions or comments?
1-866-467-2748
Safety Sealed
Do not use if printed seal under cap is torn or missing.
