Bupivacaine HCl
These highlights do not include all the information needed to use BUPIVACAINE HYDROCHLORIDE INJECTION safely and effectively. See full prescribing information for BUPIVACAINE HYDROCHLORIDE INJECTION. BUPIVACAINE HYDROCHLORIDE injection, for infiltration, perineural, caudal, epidural, or retrobulbar use Initial U.S. Approval: 1972
f29c905e-9a55-446f-a89d-860ad9ade7ad
HUMAN PRESCRIPTION DRUG LABEL
Jun 9, 2023
Xellia Pharmaceuticals USA LLC
DUNS: 116768762
Products 6
Detailed information about drug products covered under this FDA approval, including NDC codes, dosage forms, ingredients, and administration routes.
Bupivacaine hydrochloride
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (1)
Bupivacaine hydrochloride
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (1)
Bupivacaine hydrochloride
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (1)
Bupivacaine hydrochloride
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (1)
Bupivacaine hydrochloride
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (1)
Bupivacaine hydrochloride
Product Details
FDA regulatory identification and product classification information
FDA Identifiers
Product Classification
Product Specifications
INGREDIENTS (1)
Drug Labeling Information
WARNINGS AND PRECAUTIONS SECTION
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Cardiac Arrest with Use of Bupivacaine Hydrochloride Injection
in Obstetrical Anesthesia
There have been reports of cardiac arrest with difficult resuscitation or death during use of bupivacaine hydrochloride injection for epidural anesthesia in obstetrical patients. In most cases, this has followed use of the 0.75% (7.5 mg/mL) concentration. Resuscitation has been difficult or impossible despite apparently adequate preparation and appropriate management. Cardiac arrest has occurred after convulsions resulting from systemic toxicity, presumably following unintentional intravascular injection. The 0.75% (7.5 mg/mL) concentration of bupivacaine hydrochloride injection is not recommended for obstetrical anesthesia and should be reserved for surgical procedures where a high degree of muscle relaxation and prolonged effect are necessary.
5.2 Dose-Related Toxicity
The safety and effectiveness of bupivacaine hydrochloride injection depend on proper dosage, correct technique, adequate precautions, and readiness for emergencies. Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient's state of consciousness should be performed after injection of bupivacaine hydrochloride injection solutions.
Possible early warning signs of central nervous system (CNS) toxicity are restlessness, anxiety, incoherent speech, lightheadedness, numbness and tingling of the mouth and lips, metallic taste, tinnitus, dizziness, blurred vision, tremors, twitching, CNS depression, or drowsiness. Delay in proper management of dose-related toxicity, underventilation from any cause, and/or altered sensitivity may lead to the development of acidosis, cardiac arrest, and, possibly, death.
During major regional nerve blocks, such as those of the brachial plexus or lower extremity, the patient should have an indwelling intravenous catheter to assure adequate intravenous access. Use the lowest dosage of bupivacaine hydrochloride injection that results in effective anesthesia to avoid high plasma levels and serious adverse effects. Avoid rapid injection of a large volume of bupivacaine hydrochloride injection solution and administer fractional (incremental) doses when feasible.
Injection of repeated doses of bupivacaine hydrochloride injection may cause significant increases in plasma levels with each repeated dose due to slow accumulation of the drug or its metabolites, or to slow metabolic degradation. Tolerance to elevated blood levels varies with the status of the patient. Debilitated, elderly patients and acutely ill patients should be given reduced doses commensurate with their age and physical status.
5.3 Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition [see Drug Interactions (7.5)]. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious CNS and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue bupivacaine hydrochloride injection and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
5.4 Antimicrobial Preservatives in Multiple-Dose Vials
Avoid use of bupivacaine hydrochloride injection solutions containing antimicrobial preservatives, i.e., those supplied in multiple-dose vials, for epidural or caudal anesthesia because safety has not been established with such use.
5.5 Chondrolysis with Intra-Articular Infusion
Intra-articular infusions of local anesthetics including bupivacaine hydrochloride injection following arthroscopic and other surgical procedures is an unapproved use, and there have been post-marketing reports of chondrolysis in patients receiving such infusions. The majority of reported cases of chondrolysis have involved the shoulder joint; cases of gleno-humeral chondrolysis have been described in pediatric and adult patients following intra-articular infusions of local anesthetics periods of 48 to 72 hours. There is insufficient information to determine whether shorter infusion periods are associated with chondrolysis. The time of onset of symptoms, such as joint pain, stiffness and loss of motion can be variable, but may begin as early as the 2nd month after surgery. Currently, there is no effective treatment for chondrolysis; patients who experienced chondrolysis have required additional diagnostic and therapeutic procedures and some required arthroplasty or shoulder replacement.
5.7 Risk of Cardiac Arrest with Intravenous Regional Anesthesia Use (Bier
Block)
There have been reports of cardiac arrest and death during the use of bupivacaine for intravenous regional anesthesia (Bier Block). Information on safe dosages and techniques of administration of bupivacaine hydrochloride injection in this procedure is lacking. Therefore, bupivacaine hydrochloride injection is contraindicated for use with this technique [see Contraindications (4)].
5.9 Risk of Systemic Toxicities with Unintended Intravascular or
Intrathecal Injection
Unintended intravascular or intrathecal injection of bupivacaine hydrochloride injection may be associated with systemic toxicities, including CNS or cardiorespiratory depression and coma, progressing ultimately to respiratory arrest. Unintentional intrathecal injection during the intended performance of caudal or lumbar epidural block or nerve blocks near the vertebral column has resulted in underventilation or apnea ("Total or High Spinal"). A high spinal has been characterized by paralysis of the legs, loss of consciousness, respiratory paralysis, and bradycardia [see Adverse Reactions (6)].
Aspirate for blood or cerebrospinal fluid (where applicable) before injecting bupivacaine hydrochloride injection, the initial dose and all subsequent doses, to avoid intravascular or intrathecal injection. However, a negative aspiration for blood or cerebrospinal fluid does not ensure against an intravascular or intrathecal injection.
5.10 Risk of Toxicity in Patients with Hepatic Impairment
Because amide local anesthetics such as bupivacaine are metabolized by the liver, consider reduced dosing and increased monitoring for bupivacaine systemic toxicity in patients with moderate to severe hepatic impairment who are treated with bupivacaine hydrochloride injection, especially with repeat doses [see Use in Specific Populations (8.6)].
5.11 Risk of Use in Patients with Impaired Cardiovascular Function
Bupivacaine hydrochloride injection, should be given in reduced doses in patients with impaired cardiovascular function (e.g., hypotension, heartblock) because they may be less able to compensate for functional changes associated with the prolongation of AV conduction produced by bupivacaine hydrochloride injection. Monitor patients closely for blood pressure, heart rate, and ECG changes.
5.14 Risk of Adverse Reactions with Use in Head and Neck Area
Small doses of local anesthetics (e.g., bupivacaine hydrochloride injection) injected into the head and neck area, including retrobulbar and stellate ganglion blocks, may produce adverse reactions similar to systemic toxicity seen with unintentional intravascular injections of larger doses. The injection procedures require the utmost care.
Confusion, convulsions, respiratory depression, and/or respiratory arrest, and cardiovascular stimulation or depression have been reported. These reactions may be due to intra-arterial injection of the local anesthetic with retrograde flow to the cerebral circulation. They may also be due to puncture of the dural sheath of the optic nerve during retrobulbar block with diffusion of any local anesthetic along the subdural space to the midbrain. Monitor circulation and respiration and constantly observe patients receiving bupivacaine hydrochloride injection blocks. Resuscitative equipment and drugs, and personnel for treating adverse reactions should be immediately available. Dosage recommendations should not be exceeded [see Dosage and Administration (2.2)].
5.15 Risk of Respiratory Arrest with Use in Ophthalmic Surgery
Clinicians who perform retrobulbar blocks should be aware that there have been reports of respiratory arrest following local anesthetic injection. Prior to retrobulbar block (e.g., with bupivacaine hydrochloride injection), as with all other regional procedures, resuscitative equipment and drugs, and personnel to manage respiratory arrest or depression, convulsions, and cardiac stimulation or depression should be immediately available [see Warnings and Precautions (5.14)]. As with other anesthetic procedures, patients should be constantly monitored following ophthalmic blocks for signs of these adverse reactions, which may occur following relatively low total doses.
A concentration of 0.75% bupivacaine is indicated for retrobulbar block; however, this concentration is not indicated for any other peripheral nerve block, including the facial nerve, and not indicated for local infiltration, including the conjunctiva [see Indications and Usage (1)].
- Dose-Related Toxicity: Monitor cardiovascular and respiratory vital signs and patient's state of consciousness after injection of bupivacaine hydro- chloride injection. (5.2)
- Methemoglobinemia: Cases of methemoglobinemia have been reported in association with local anesthetic use. See full prescribing information for more detail on managing these risks. (5.3)
- Chondrolysis with Intra-Articular Infusion: Intra-articular infusions of local anesthetics including bupivacaine hydrochloride injection following ar- throscopic and other surgical procedures is an unapproved use, and there have been post-marketing reports of chondrolysis in patients receiving such infusions. (5.5)
- Risk of Cardiac Arrest with Intravenous Regional Anesthesia Use (Bier Block): There have been reports of cardiac arrest and death during the use of bupivacaine for intravenous regional anesthesia (Bier Block). (5.7)
- Risk of Systemic Toxicities with Unintended Intravascular or Intrathecal Injection: Unintended intravascular or intrathecal injection may be associated with systemic toxicities, including CNS or cardiorespiratory depression and coma, progressing ultimately to respiratory arrest. Aspirate for blood or cerebrospinal fluid (where applicable) prior to each dose. (5.9)
DRUG INTERACTIONS SECTION
7 DRUG INTERACTIONS
7.1 Local Anesthetics
The toxic effects of local anesthetics are additive. If coadministration of other local anesthetics with bupivacaine hydrochloride injection cannot be avoided, monitor patients for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Dosage and Administration (2.1), Warnings and Precautions (5.2)].
7.5 Drugs Associated with Methemoglobinemia
Patients who are administered bupivacaine hydrochloride injection are at increased risk of developing methemoglobinemia when concurrently exposed to following drugs, which could include other local anesthetics [see Warnings and Precautions (5.3)].
Examples of Drugs Associated with Methemoglobinemia:|
Class |
Examples |
|---|---|
|
Nitrates/Nitrites |
nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
|
Local anesthetics |
articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
|
Antineoplastic agents |
cyclophosphamide, flutamide, hydroxyurea, isofamide, rasburicase |
|
Antibiotics |
dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
|
Antimalarials |
chloroquine, primaquine |
|
Anticonvulsants |
phenobarbital, phenytoin, sodium valproate |
|
Other drugs |
acetaminophen, metoclopramide, quinine, sulfasalazine |
- Local Anesthetics: The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects when additional local anesthetics are administered. (7.1)
- Drugs Associated with Methemoglobinemia: Patients are at increased risk of developing methemoglobinemia when concurrently exposed to nitrates, nitrites, local anesthetics, antineoplastic agents, antibiotics, antimalarials, anticonvulsants, and other drugs. (7.5)
DESCRIPTION SECTION
11 DESCRIPTION
Bupivacaine hydrochloride injection contains bupivacaine hydrochloride, an amide local anesthetic, as the active pharmaceutical ingredient. The route of administration for bupivacaine hydrochloride injection is by injection, for infiltration, perineural, caudal, epidural, or retrobulbar use. [see Warnings and Precautions (5.4)].
Bupivacaine hydrochloride is 2-piperidinecarboxamide, 1-butyl-N-(2,6-dimethylphenyl)-, monohydrochloride, monohydrate. It is a white crystalline powder that is freely soluble in 95 percent ethanol, soluble in water, and slightly soluble in chloroform or acetone. It has the following structural formula:
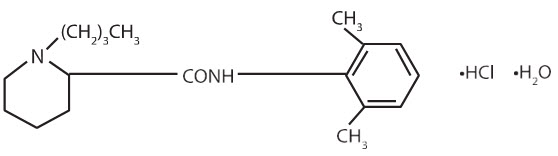
Bupivacaine hydrochloride injection is a clear and colorless sterile isotonic solution. Each mL of single-dose vial contains 2.5 mg, 5 mg or 7.5 mg of bupivacaine hydrochloride (equivalent to 2.22 mg, 4.44 mg or 6.66 mg of bupivacaine, respectively), sodium chloride for isotonicity, sodium hydroxide or hydrochloric acid to adjust the pH between 4 and 6.5, in water for injection.
For the Multiple-dose vials, each mL also contains 1 mg methylparaben as preservative.
NONCLINICAL TOXICOLOGY SECTION
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long-term studies in animals to evaluate the carcinogenic potential of bupivacaine hydrochloride have not been conducted.
Mutagenesis
The mutagenic potential of bupivacaine hydrochloride has not been determined.
Impairment of Fertility
The effect of bupivacaine on fertility has not been determined.
HOW SUPPLIED SECTION
16 HOW SUPPLIED/STORAGE AND HANDLING
Store at 20 °C to 25 °C (68 °F to 77 °F); excursions permitted between 15 °C to 30 °C (59 ° F to 86 °F). [See USP Controlled Room Temperature.]
Protect from light. This product is clear and colorless. Do not use the solution if it is discolored or if it contains a precipitate.
|
Unit of Sale |
Concentration |
Each |
|---|---|---|
|
0.25% Contains 2.5 mg bupivacaine hydrochloride per mL. | ||
|
NDC 70594-114-02 |
25 mg/10 mL (2.5 mg/mL) |
NDC 70594-114-01 |
|
NDC 70594-115-01 |
75 mg/30 mL (2.5 mg/mL) |
NDC 70594-115-01 |
|
0.5% Contains 5 mg bupivacaine hydrochloride per mL. | ||
|
NDC 70594-117-02 |
50 mg/10 mL (5 mg/mL) |
NDC 70594-117-01 |
|
NDC 70594-118-01 |
150 mg/30 mL (5 mg/mL) |
NDC 70594-118-01 |
|
0.75% Contains 7.5 mg bupivacaine hydrochloride per mL. | ||
|
NDC 70594-120-02 |
75 mg/10 mL (7.5 mg/mL) |
NDC 70594-120-01 |
|
NDC 70594-121-01 |
225 mg/30 mL (7.5 mg/mL) |
NDC 70594-121-01 |
For single-dose vials: Discard unused portion.
INFORMATION FOR PATIENTS SECTION
17 PATIENT COUNSELING INFORMATION
Allergic-Type Reactions
Assess if the patient has had allergic-type reactions to amide-type local anesthetics or to other formulation ingredients, such as the antimicrobial preservative methylparaben contained in multiple-dose vials [see Contraindications (4), Adverse Reactions (6)].
Temporary Loss of Sensation and Motor Activity After Caudal or Epidural Anesthesia
When appropriate, patients should be informed in advance that they may experience temporary loss of sensation and motor activity, usually in the lower half of the body, following proper administration of caudal or epidural anesthesia.
Methemoglobinemia
Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue [see Warnings and Precautions (5.3)].
