- Approval Id
- e4aa10705e943001
- Drug Name
- VORICONAZOLE KABI POWDER FOR SOLUTION FOR INFUSION 200MG/VIAL
- Product Name
- VORICONAZOLE KABI POWDER FOR SOLUTION FOR INFUSION 200MG/VIAL
- Approval Number
- SIN16509P
- Approval Date
- 2022-06-09
- Registrant
- FRESENIUS KABI (SINGAPORE) PTE LTD
- Licence Holder
- FRESENIUS KABI (SINGAPORE) PTE LTD
- Drug Type
- Therapeutic
- Forensic Classification
- Prescription Only
- Dosage Form
- INJECTION, POWDER, LYOPHILIZED, FOR SOLUTION
- Dosage
- **4.2 Posology and method of administration**
_Voriconazole tablet and oral suspension are unavailable in this brand however, are available in other brands. Where correct dosing requires oral formulations, refer to the specific product information for these formulations for their complete dosage and administration instructions._
Voriconazole Kabi requires reconstitution and dilution (see Section 6.6 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_) prior to administration as an intravenous infusion.
Voriconazole Kabi is **not** recommended for bolus injection.
It is recommended that Voriconazole Kabi be administered at a maximum rate of 3 mg/kg per hour over 1 to 3 hours.
**Blood products and concentrated electrolytes**
Voriconazole Kabi must not be infused concomitantly with any blood product or any short-term infusion of concentrated electrolytes, even if the two infusions are running in separate intravenous lines (or cannulas). Electrolyte disturbances such as hypokalemia, hypomagnesemia and hypocalcemia should be corrected prior to initiation of voriconazole therapy (see Section 4.4 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
**Intravenous solutions containing (non-concentrated) electrolytes**
Voriconazole Kabi can be infused at the same time as other intravenous solutions containing (non-concentrated) electrolytes, but must be infused through a separate line.
**Total parenteral nutrition (TPN)**
Voriconazole Kabi can be infused at the same time as total parenteral nutrition, but must be infused in a separate line. If infused through a multiple-lumen catheter, TPN needs to be administered using a different port from the one used for voriconazole (see Section 6.2 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
**Other intravenous products**
Voriconazole Kabi must not be infused into the same line or cannula concomitantly with other intravenous products.
**Use in adults**
Therapy must be initiated with the specified loading dose regimen of either intravenous or oral voriconazole to achieve plasma concentrations on Day 1 that are close to steady state. On the basis of the high oral bioavailability (96%; see Section 5.2 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_), switching between intravenous and oral administration is appropriate when clinically indicated.
Detailed information on dosage recommendations is provided in the following table:
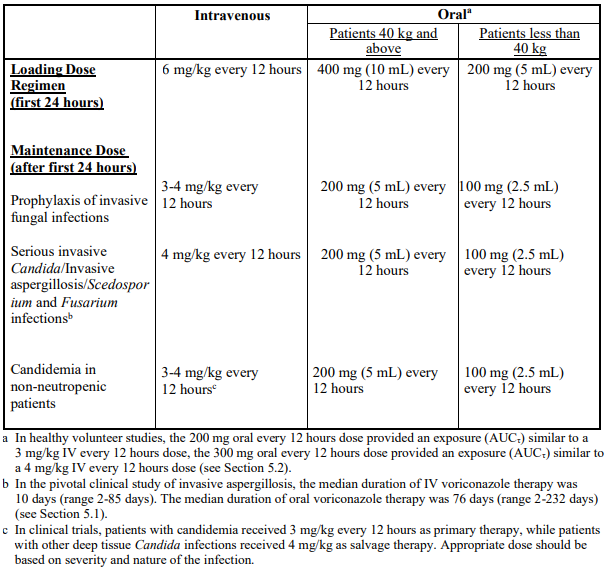
**Dosage adjustment**
If patient response at 3 mg/kg every 12 hours is inadequate, the intravenous maintenance dose may be increased to 4 mg/kg every 12 hours.
If patients are unable to tolerate 4 mg/kg every 12 hours, reduce the intravenous maintenance dose to a minimum of 3 mg/kg every 12 hours.
Phenytoin may be co-administered with voriconazole if the maintenance dose of voriconazole is increased to 5 mg/kg intravenously every 12 hours (see Sections 4.4 and 4.5 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Treatment duration depends upon patients’ clinical and mycological response. Clinical data to establish the safety of intravenously administered hydroxypropylbetadex in long term treatment are limited (see section 5.2 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
**Use in the elderly**
No dose adjustment is necessary for elderly patients.
**Use in patients with renal impairment**
In patients with moderate to severe renal dysfunction (creatinine clearance < 50 ml/min), accumulation of the intravenous vehicle, hydroxypropylbetadex, occurs. Oral voriconazole should be administered to these patients, unless an assessment of the risk benefit to the patient justifies the use of intravenous voriconazole. Serum creatinine levels should be closely monitored in these patients and, if increases occur, consideration should be given to changing to oral voriconazole therapy (see section 5.2 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). Use in patients who are not undergoing hemodialysis is not recommended.
Voriconazole is hemodialyzed with a clearance of 121 mL/min. A four-hour haemodialysis session does not remove a sufficient amount of voriconazole to warrant dose adjustment.
The intravenous vehicle, hydroxypropylbetadex, is hemodialyzed with a clearance of 37.5 ± 24 ml/min.
**Use in patients with hepatic impairment**
No dose adjustment is necessary in patients with acute hepatic injury, manifested by elevated liver function tests (ALT, AST). Continued monitoring of liver function tests for further elevations is recommended.
It is recommended that the standard loading dose regimens be used but that the maintenance dose be halved in patients with mild to moderate hepatic cirrhosis (Child-Pugh A and B) receiving voriconazole.
Voriconazole has not been studied in patients with severe chronic hepatic cirrhosis (Child-Pugh C).
Voriconazole has been associated with elevations in liver function tests and clinical signs of liver damage, such as jaundice, and must only be used in patients with severe hepatic impairment if the benefit outweighs the potential risk. Patients with severe hepatic impairment must be carefully monitored for drug toxicity.
**Use in pediatrics**
Safety and effectiveness in pediatric patients below the age of 2 years has not been established (see also Section 5.1 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). Therefore, voriconazole is not recommended for children less than 2 years of age.
The recommended maintenance dosing regimen in pediatric patients 2 to <12 years is as follows:
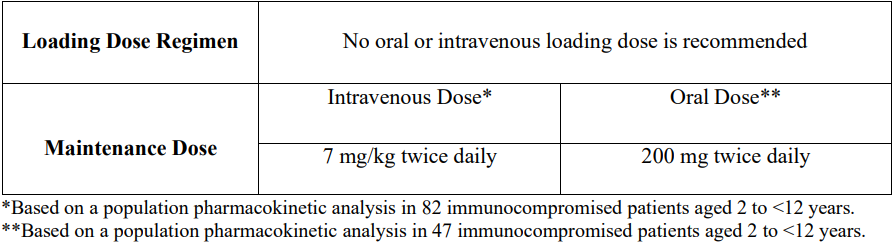
If pediatric patients are unable to tolerate an intravenous dose of 7 mg/kg twice daily, a dose reduction from 7 mg/kg to 4 mg/kg twice daily may be considered based on the population pharmacokinetic analysis and previous clinical experience. This provides equivalent exposure to 3 mg/kg twice daily in adults (see Section 4.2, Use in adults).
Clinical data to establish the safety of intravenously administered hydroxypropylbetadex in the pediatric population are limited.
Use in pediatric patients aged 2 to <12 years with hepatic or renal insufficiency has not been studied (see Sections 4.8 and 5.2 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
**Adolescents** (12 to 16 years of age) should be dosed as adults.
- Route Of Administration
- INTRAVENOUS
- Indication Info
- **4.1 Therapeutic indications**
Voriconazole is a broad spectrum, triazole antifungal agent and is indicated as follows:
Treatment of invasive aspergillosis;
Treatment of candidemia in non-neutropenic patients;
Treatment of fluconazole-resistant serious invasive _Candida_ infections (including _C. krusei_);
Treatment of serious fungal infections caused by _Scedosporium_ spp. and _Fusarium_ spp.;
Prophylaxis in patients ≥12 years old who are at high risk of developing invasive fungal infections. The indication is based on a study which includes patients ≥12 years old undergoing allogeneic haematopoietic stem cell transplantation (see Section on 'Clinical Experience' – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
- Contraindications
- **4.3 Contraindications**
Voriconazole is contraindicated in patients with known hypersensitivity to voriconazole or to any of the excipients.
Co-administration of the CYP3A4 substrates, terfenadine, astemizole, cisapride, pimozide, quinidine or ivabradine with voriconazole is contraindicated since increased plasma concentrations of these medicinal products can lead to QTc prolongation and rare occurrences of _torsades de pointes_ (see Section 4.5 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Co-administration of voriconazole and sirolimus is contraindicated, since voriconazole has been shown to significantly increase plasma concentrations of sirolimus in healthy subjects (see Section 4.5 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Co-administration of voriconazole with rifabutin, rifampicin, carbamazepine, long-acting barbiturates (e.g., phenobarbital) and St John’s Wort is contraindicated since these medicinal products are likely to decrease plasma voriconazole concentrations significantly (see Section 4.5 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Co-administration of standard doses of voriconazole with efavirenz doses of 400 mg QD or higher is contraindicated because efavirenz significantly decreases plasma voriconazole concentrations in healthy subjects at these doses. Voriconazole also significantly increases efavirenz plasma concentrations (see Section 4.5, for lower doses see Section 4.4 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Co-administration of ergot alkaloids (ergotamine, dihydroergotamine), which are CYP3A4 substrates, is contraindicated since increased plasma concentrations of these medicinal products can lead to ergotism (see Section 4.5 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Co-administration of voriconazole with high-dose ritonavir (400 mg and above twice daily) is contraindicated because ritonavir significantly decreases plasma voriconazole concentrations in healthy subjects at this dose (see Section 4.5, for lower doses see Section 4.4 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Co-administration of voriconazole with naloxegol is contraindicated because voriconazole may significantly increase plasma concentrations of naloxegol which may precipitate opioid withdrawal symptoms (see Section 4.5 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Co-administration of voriconazole with tolvaptan is contraindicated because voriconazole may significantly increase plasma concentrations of tolvaptan (see Section 4.5 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Co-administration of voriconazole with venetoclax at initiation and during the venetoclax dose titration phase since voriconazole is likely to significantly increase plasma concentrations of venetoclax and increase risk of tumour lysis syndrome (see Section 4.5 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
Co-administration of voriconazole with lurasidone is contraindicated since it may result in significant increases in lurasidone exposure and the potential for serious adverse reactions (see Section 4.5 – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
- Atc Code
- J02AC03
- Atc Item Name
- voriconazole
- Pharma Manufacturer Name
- FRESENIUS KABI (SINGAPORE) PTE LTD
- Company Detail Path
- /organization/15bc26a40571352c/fresenius-kabi-singapore-pte-ltd