Regulatory Information
HSA regulatory responsibility and product classification details
Regulatory Responsibility
Product Classification
Formulation Information
CAPSULE
**Dosage and Administration** For optimal absorption, administer SPORANOX® capsules immediately after a full meal. The capsules must be swallowed whole. 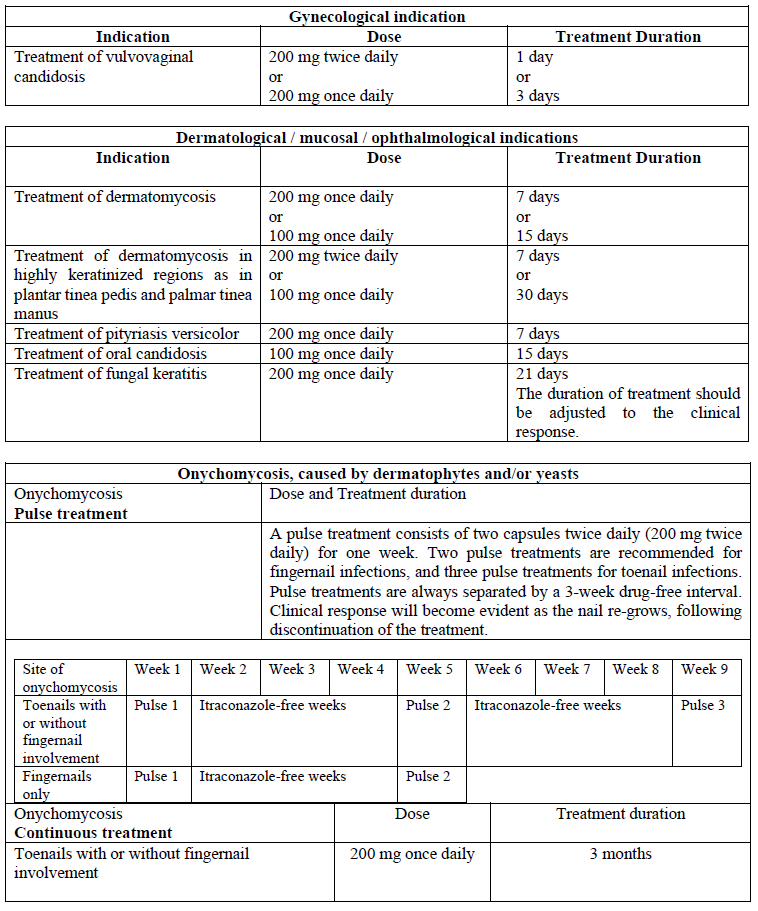 Elimination of itraconazole from skin and nail tissue is slower than from plasma. Optimal clinical and mycological response is thus reached 2 to 4 weeks after the cessation of treatment for skin infections and 6 to 9 months after the cessation of treatment for nail infections. 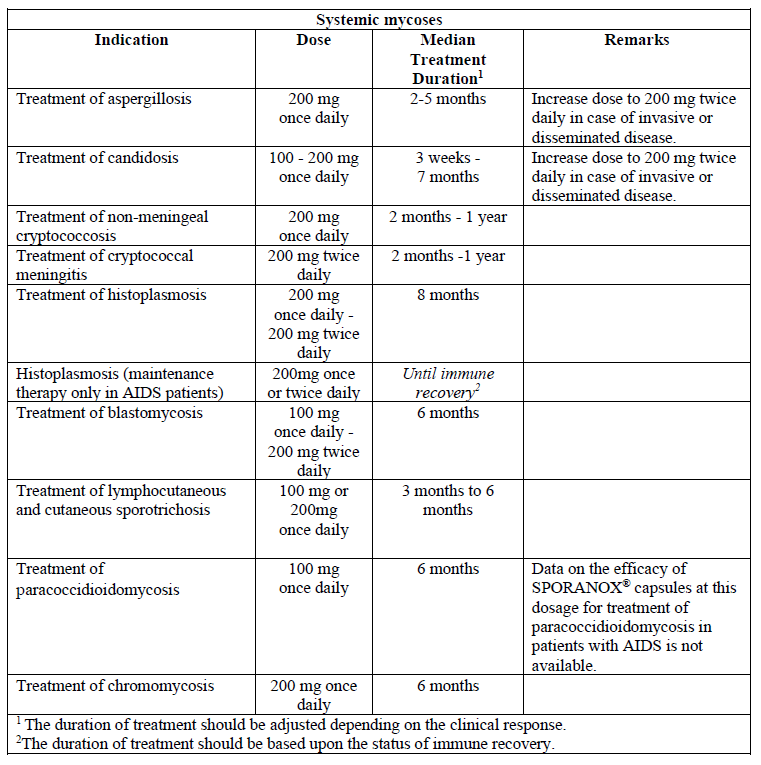 **Special populations** _**Pediatrics**_ Clinical data on the use of SPORANOX® capsules in pediatric patients are limited. The use of SPORANOX® capsules in pediatric patients is not recommended unless it is determined that the potential benefit outweighs the potential risks (see _Warnings and Precautions_ – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). _**Elderly**_ Clinical data on the use of SPORANOX® capsules in elderly patients are limited. It is advised to use SPORANOX® capsules in these patients only if it is determined that the potential benefit outweighs the potential risks. In general, it is recommended that the dose selection for an elderly patient should be taken into consideration, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy (see _Warnings and Precautions_ – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). _**Hepatic impairment**_ Limited data are available on the use of oral itraconazole in patients with hepatic impairment. Caution should be exercised when this drug is administered in this patient population (see _Pharmacokinetic properties - Special populations, Hepatic impairment_ – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). _**Renal impairment**_ Limited data are available on the use of oral itraconazole in patients with renal impairment. The exposure of itraconazole may be lower in some patients with renal insufficiency. Caution should be exercised when this drug is administered in this patient population and adjusting the dose may be considered (see _Pharmacokinetic Properties - Special populations, Renal impairment_ – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_).
ORAL
Medical Information
**Indications** SPORANOX® capsules are indicated for the following conditions: - Gynecological indications: - Treatment of vulvovaginal candidosis. - Dermatological / mucosal / ophthalmological indications: - Treatment of dermatomycosis, including highly keratinized regions as in plantar tinea pedis and palmar tinea manus; - Treatment of pityriasis versicolor; - Treatment of oral candidosis; - Treatment of fungal keratitis. - Treatment of onychomycosis, caused by dermatophytes and/or yeasts. - Systemic mycoses, only in the following fungal infections: - Treatment of systemic aspergillosis and candidosis; - Treatment of cryptococcosis (including cryptococcal meningitis): in immunocompromised patients with cryptococcosis and in patients with cryptococcosis of the central nervous system, only when first line treatment is considered inappropriate or has proven ineffective; - Histoplasmosis: - Treatment of histoplasmosis; - Maintenance therapy of histoplasmosis only in AIDS patients; - Treatment of blastomycosis; - Treatment of lymphocutaneous/cutaneous sporotrichosis; - Treatment of paracoccidioidomycosis; - Treatment of chromomycosis.
**Contraindications** - SPORANOX® capsules are contraindicated in patients with known hypersensitivity to itraconazole or to any of the excipients. - Co-administration of a number of CYP3A4 substrates is contraindicated with SPORANOX® capsules. Increased plasma concentrations of these drugs, caused by coadministration with itraconazole, may increase or prolong both therapeutic and adverse effects to such an extent that a potentially serious situation may occur. For example, increased plasma concentrations of some of these drugs can lead to QT prolongation and ventricular tachyarrhythmias including occurrences of torsade de pointes, a potentially fatal arrhythmia. Some specific examples are listed in _Interactions_ – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_. - SPORANOX® capsules should not be administered to patients with evidence of ventricular dysfunction such as congestive heart failure (CHF) or a history of CHF except for the treatment of life-threatening or other serious infections (see _Warnings and Precautions_ – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). SPORANOX® capsules must not be used during pregnancy (except for life-threatening cases) (see _Pregnancy, Breast-feeding and Fertility_ – _please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information_). Women of childbearing potential taking SPORANOX® should use contraceptive precautions. Highly effective contraception should be continued until the menstrual period following the end of SPORANOX® therapy.
J02AC02
itraconazole
Manufacturer Information
JOHNSON & JOHNSON INTERNATIONAL (SINGAPORE) PTE. LTD.
JANSSEN CILAG SPA
Active Ingredients
Documents
Package Inserts
SPORANOX CAPSULES PI.pdf
Approved: June 16, 2023
